葡萄(Vitis spp.)是较易受病毒侵染的果树树种。全球已鉴定的葡萄病毒多达95种,我国已报道了22种[1-2]。由于长期进行无性繁殖及新品种推广,导致病毒在植株体内逐年积累和传播,病毒病发生日趋严重。因此,培育和栽培无病毒苗木对葡萄生产具有重要意义。热处理结合茎尖培养是目前应用广泛的脱毒方法[3-6],经热处理后茎尖无毒区域增加,接种1~2 mm茎尖即可获得脱毒苗,操作方便,且茎尖成活率和脱毒率高。Skiada等[7]利用此方法脱除了圣乔治葡萄试管苗中的葡萄卷叶相关病毒1(GLRaV-1)和沙地葡萄茎痘相关病毒(GRSPaV),脱毒率分别为91.2%、67.6%。杜易静等[8]通过热处理与茎尖培养,对携带葡萄灰比诺病毒(GPGV)、葡萄卷叶相关病毒3(GLRaV-3)、葡萄病毒E(GVE)、蚕豆萎蔫病毒(GFabV)和GRSPaV的阳光玫瑰葡萄试管苗进行脱毒处理,在检测的164个无性系中,68.3%的无性系脱除了原株携带的全部病毒,93.3%的无性系仅存在GRSPaV。Gambino等[9]、张尊平等[10]和Hu等[11]的研究也相继表明,利用热处理结合茎尖培养进行葡萄脱毒时,GRSPaV较难脱除。
器官再生是指将植物的茎、叶等器官从母体分离,在无菌条件下进行离体培养,形成新组织和器官的过程。在器官再生过程中,细胞分裂迅速,容易获得部分无毒的组织、细胞,由此诱导不定芽再生,获得无病毒植株[12]。利用器官再生进行脱毒已有成功报道,徐品三等[13]通过百合鳞片诱导不定芽再生,成功获得无病毒的卡萨布兰卡百合植株;周美玲等[14]利用福建百香果3号枝条茎段诱导愈伤组织,由愈伤组织再生不定芽,成苗后脱毒率为45.0%。
葡萄是较难离体再生的果树树种,多数品种再生率较低。常新等[15]诱导黑香蕉葡萄试管苗不同器官再生不定芽,结果显示,叶柄和叶片的再生率分别仅为16.9%、4.9%。不同基因型间的再生能力差异较大,Maletich等[16]以霞多丽等22个葡萄品种的试管苗离体叶片和叶柄为材料,诱导不定芽再生,仅有Ruta和Podarok Magaracha两个品种的再生率较高,分别为87.0%和100.0%;8个品种再生率在1.7%~3.5%之间;其余5个品种未实现再生。为解决葡萄常规脱毒方法难以脱除GRSPaV等病毒的问题,笔者利用器官再生途径探究脱除病毒的可行性。选用具有优良性状的红斯威特和阳光玫瑰葡萄试管苗,优化再生培养基配方提高再生效率,通过诱导离体叶片直接再生不定芽或通过愈伤组织再生不定芽两种途径脱除病毒,以期为葡萄无病毒苗木的培育提供理论依据与技术支撑。
1 材料和方法
1.1 材料
试验于2021年10月—2024年8月在河北农业大学园艺学院生物技术实验室进行。以感染GRSPaV的红斯威特葡萄以及感染GVE、GRSPaV和GFabV的阳光玫瑰葡萄试管苗为试材(两个品种均对葡萄病毒A、葡萄病毒B、GVE、卷叶相关病毒1、卷叶相关病毒2、卷叶相关病毒3、卷叶相关病毒4、葡萄浆果内坏死病毒、葡萄灰比诺病毒、葡萄扇叶病毒、GRSPaV和GFabV共12种病毒进行了检测)。红斯威特葡萄试管苗继代培养基为B5+0.5 mg·L-1 IAA +25 g·L-1蔗糖+6 g·L-1琼脂;阳光玫瑰葡萄试管苗继代培养基为B5+0.3 mg·L-1 IBA +0.5 mg·L-1 IAA +25 g·L-1葡萄糖+6 g·L-1琼脂,pH=5.8~6.0。试管苗每隔30~40 d继代一次。
1.2 方法
1.2.1 细胞分裂素与生长素对葡萄试管苗叶片离体再生的影响 选取继代培养30~40 d的红斯威特和阳光玫瑰葡萄试管苗顶部1~3叶位的幼嫩叶片,切分成长宽为0.5~1 cm的叶块,接种时远轴面向上。基本培养基为B5+15 g·L-1葡萄糖+6 g·L-1 琼脂,在此基础上分别附加不同浓度的细胞分裂素(TDZ/BA)与生长素(IAA/NAA),共12个处理组合(表1)。每个处理设置9瓶,每瓶接种5~7个叶块,采用完全随机试验设计,3次重复。接种后,先黑暗培养14 d,然后转移至光下培养,日光灯强度为2000 lx;光周期为14 h 光照/10 h黑暗,培养温度为(25±2) ℃(下同)。培养50 d后统计以下指标:不定芽再生率/%=再生不定芽叶块数/接种总叶块数×100;平均出芽数=出芽数/接种总叶块数;愈伤组织再生率/%=再生愈伤组织叶块数/接种总叶块数×100。
表1 培养基的细胞分裂素与生长素处理组合
Table 1 The combination of cytokinin and auxin treatments on medium composition(mg·L-1)
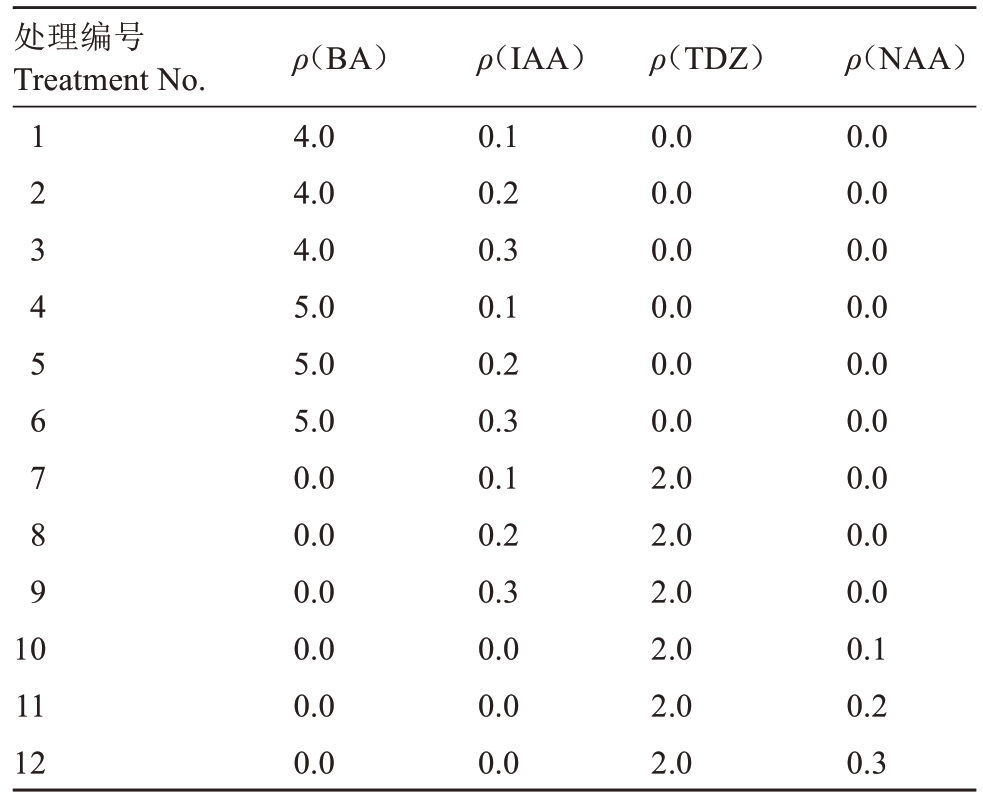
处理编号Treatment No.1 2 3 4 5 6 7 8 9 10 11 12 ρ(BA)4.0 4.0 4.0 5.0 5.0 5.0 0.0 0.0 0.0 0.0 0.0 0.0 ρ(IAA)0.1 0.2 0.3 0.1 0.2 0.3 0.1 0.2 0.3 0.0 0.0 0.0 ρ(TDZ)0.0 0.0 0.0 0.0 0.0 0.0 2.0 2.0 2.0 2.0 2.0 2.0 ρ(NAA)0.0 0.0 0.0 0.0 0.0 0.0 0.0 0.0 0.0 0.1 0.2 0.3
1.2.2 不同TDZ浓度对葡萄试管苗叶片离体再生的影响 将红斯威特和阳光玫瑰葡萄试管苗离体叶片接种于B5+0.1 mg·L-1 IAA+15 g·L-1葡萄糖+6 g·L-1琼脂+(0.5、1.0、2.0、2.5、3.0 mg·L-1)TDZ培养基中。培养50 d后统计不定芽再生率、平均出芽数和愈伤组织再生率。
1.2.3 硝普钠(SNP)对葡萄试管苗叶片离体再生不定芽的影响 将红斯威特和阳光玫瑰葡萄试管苗叶片接种于MS或B5+1.0 mg·L-1 TDZ+0.1 mg·L-1 IAA+15 g·L-1 葡萄糖+6 g·L-1 琼脂+(0、4、6、8、10、12 mg·L-1)SNP培养基中。培养50 d后统计不定芽再生率、平均出芽数。
1.2.4 红斯威特葡萄试管苗叶片离体再生不定芽病毒脱除 将红斯威特葡萄试管苗离体叶片再生的不定芽,接种于B5+0.5 mg·L-1 IAA+25 g·L-1蔗糖培养基中,培养成试管苗并扩繁。同一个不定芽繁殖的试管苗为一个无性系,选取7个无性系试管苗利用RT-PCR技术进行病毒检测。
1.2.5 阳光玫瑰葡萄试管苗叶片离体再生愈伤组织和不定芽病毒脱除 将阳光玫瑰葡萄试管苗离体叶片再生的愈伤组织,接种于B5+1.0 mg·L-1 BA+0.1 mg·L-1 NAA+30 g·L-1葡萄糖培养基中,每20~30 d继代一次,从每代愈伤组织中选取6个样品进行病毒检测。
将无病毒愈伤组织转接于B5+2.0 mg·L-1 TDZ+0.2 mg·L-1 NAA+15 g·L-1葡萄糖培养基中,诱导不定芽再生。将不定芽接种于B5+0.3 mg·L-1 IBA+0.5 mg·L-1 IAA+25 g·L-1葡萄糖培养基中,培养成试管苗并扩繁,对获得的试管苗进行病毒检测。
1.2.6 红斯威特和阳光玫瑰葡萄试管苗和愈伤组织病毒检测 红斯威特葡萄试管苗检测GRSPaV,阳光玫瑰葡萄愈伤组织和试管苗检测GVE、GRSPaV和GFabV。参照全能型植物RNA试剂盒(DNase I)说明书(CWBIO,Beijing,China)提取总RNA。红斯威特每个无性系试管苗样品随机选取9株,每3株试管苗为一组,取其上、中、下部叶片混合取样50~100 mg,共设置3次重复;阳光玫瑰愈伤组织每个样品选取6瓶,每2瓶愈伤组织混合取样50~100 mg,共设置3次重复。阴性对照为无病毒的葡萄试管苗;阳性对照为感染相应病毒的葡萄试管苗。
利用EasyScript® One-Step gDNA Removal and cDNA Synthesis SuperMix试剂盒(TRAN,Beijing,China)进行cDNA合成和gDNA去除,反转录反应程序:42 ℃孵育30 min;80 ℃加热5 s。用于RTPCR的引物序列信息见表2[17];反应体系(20 µL):上、下游引物各1 μL、cDNA 1 μL、2×EasyTaq®PCR SuperMix 10 μL、无菌超纯水7 μL;反应程序:94 ℃5 min;94 ℃ 30 s,各引物适宜退火温度退火30 s,72 ℃延伸(延伸时间见表2),35个循环;72 ℃ 5 min。
表2 RT-PCR检测的引物序列及反应条件
Table 2 The primer sequences and program used for RT-PCR analysis

病毒Virus GRSPaV GVE GFabV引物序列Primer sequence F:TGAAGGCTTTAGGGGTTAG;R:CTTAACCCAGCCTTGAAAT F:TGTGGGGTGCATAGTCATAGGTTT;R:GCTTTTGACTCCATTGGCTTTCTC F:GGAAATAACATTCCCAGGAAG;R:TCCTGCTGTCAAATTTCATTC片段大小Fragment size/bp 905 478 325退火温度Annealing temperature/℃57.8 56.0 50.0延伸时间Extension time/s 60 40 40
1.3 数据统计分析
利用Excel 2019软件对数据的平均值和标准差进行整理和计算,利用DPS 7.05软件对数据进行差异显著性测验,其中百分数数据进行反正弦平方根转换后进行方差分析(Duncan新复极差测验),显著水平P<0.05。
2 结果与分析
2.1 不同细胞分裂素与生长素种类对葡萄试管苗叶片离体再生的影响
培养基中添加不同种类的细胞分裂素(BA、TDZ)对红斯威特和阳光玫瑰葡萄试管苗叶片离体再生具有显著影响。如表3所示,仅处理7(2.0 mg·L-1 TDZ+0.1 mg·L-1 IAA)可诱导红斯威特葡萄叶片再生不定芽,不定芽再生率为13.10%,平均出芽数0.29个,显著高于其他处理(P<0.05),而BA处理组均未诱导出不定芽;在愈伤组织再生方面,红斯威特和阳光玫瑰葡萄的TDZ处理组愈伤组织再生率均显著高于BA处理组,表明TDZ与生长素的组合更利于诱导红斯威特葡萄不定芽再生及两个品种愈伤组织形成,而BA处理对不定芽分化和愈伤组织形成的效果较差。添加不同种类和浓度的生长素(IAA与NAA)对两个葡萄品种叶片离体再生的影响不大,除处理7(2.0 mg·L-1 TDZ+0.1 mg·L-1 IAA)以及处理11(2.0 mg·L-1 TDZ+0.2 mg·L-1 NAA)外,其余生长素处理之间无显著差异。
表3 细胞分裂素与生长素对葡萄试管苗叶片离体再生的影响
Table 3 Effects of cytokinin and auxin on regeneration from the leaves of grape plantlets in vitro
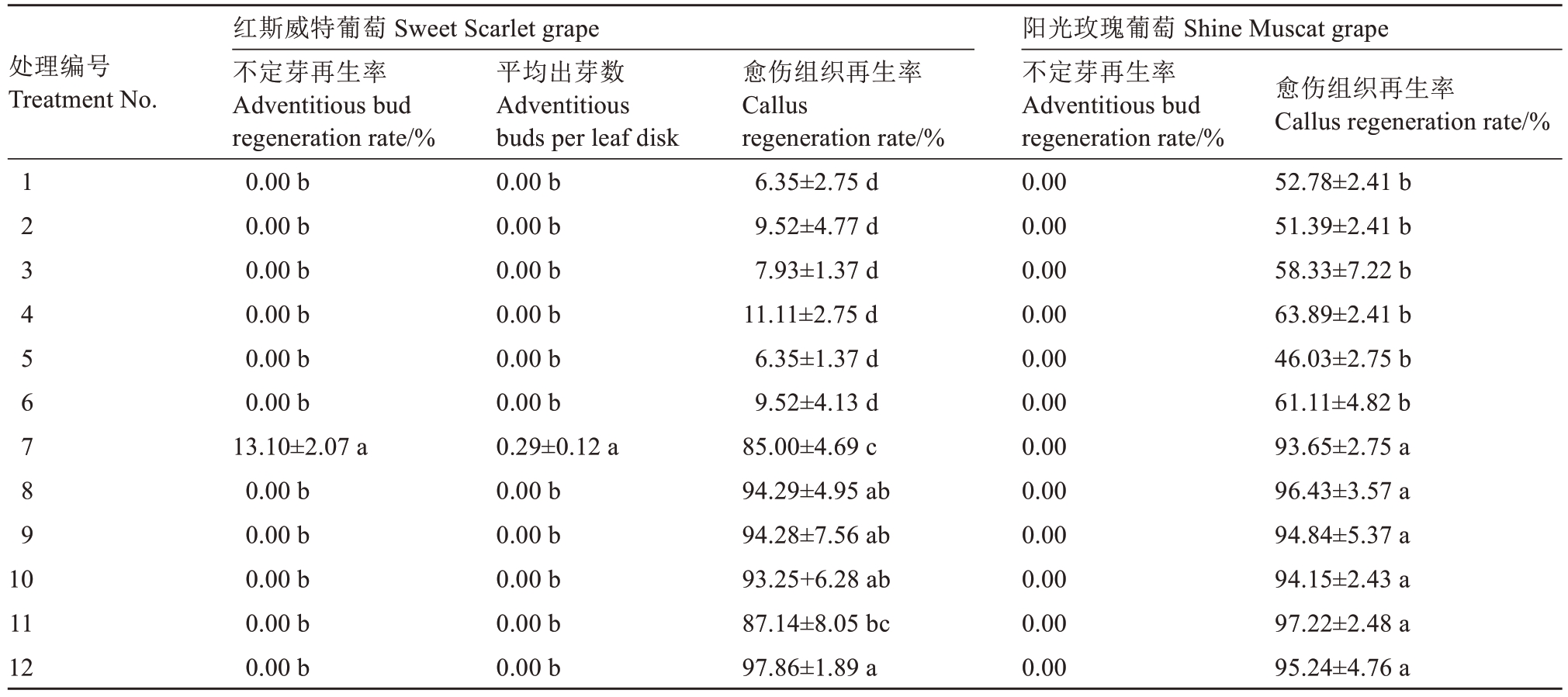
注:同列不同小写字母表示差异显著(P<0.05)。下同。
Note:Different small letters in the same column indicate significant difference at P<0.05. The same below.
处理编号Treatment No.1 2 3 4 5 6 7 8 9 10 11 12红斯威特葡萄 Sweet Scarlet grape不定芽再生率Adventitious bud regeneration rate/%0.00 b 0.00 b 0.00 b 0.00 b 0.00 b 0.00 b 13.10±2.07 a 0.00 b 0.00 b 0.00 b 0.00 b 0.00 b平均出芽数Adventitious buds per leaf disk 0.00 b 0.00 b 0.00 b 0.00 b 0.00 b 0.00 b 0.29±0.12 a 0.00 b 0.00 b 0.00 b 0.00 b 0.00 b愈伤组织再生率Callus regeneration rate/%6.35±2.75 d 9.52±4.77 d 7.93±1.37 d 11.11±2.75 d 6.35±1.37 d 9.52±4.13 d 85.00±4.69 c 94.29±4.95 ab 94.28±7.56 ab 93.25+6.28 ab 87.14±8.05 bc 97.86±1.89 a阳光玫瑰葡萄 Shine Muscat grape不定芽再生率Adventitious bud regeneration rate/%0.00 0.00 0.00 0.00 0.00 0.00 0.00 0.00 0.00 0.00 0.00 0.00愈伤组织再生率Callus regeneration rate/%52.78±2.41 b 51.39±2.41 b 58.33±7.22 b 63.89±2.41 b 46.03±2.75 b 61.11±4.82 b 93.65±2.75 a 96.43±3.57 a 94.84±5.37 a 94.15±2.43 a 97.22±2.48 a 95.24±4.76 a
2.2 TDZ浓度对葡萄试管苗叶片离体再生的影响
将红斯威特和阳光玫瑰葡萄试管苗离体叶片分别接种于不同浓度TDZ的培养基中,结果如表4所示:随着TDZ浓度增加,红斯威特葡萄的不定芽再生率和平均出芽数呈“升-降-升-降”的变化趋势,其中1.0 mg·L-1 TDZ处理下的不定芽再生率达18.06%,平均出芽数为0.96个,显著高于其他处理(P<0.05),而2.0、2.5、3.0 mg·L-1 TDZ处理下的愈伤组织再生率显著高于低浓度处理(P<0.05)。阳光玫瑰葡萄在所有TDZ浓度处理下均未诱导出不定芽,2.0和2.5 mg·L-1 TDZ处理的愈伤组织再生率分别达89.95%和86.57%,显著高于低浓度处理(P<0.05)。这表明TDZ浓度对两个葡萄品种的叶片再生途径影响显著,红斯威特葡萄诱导不定芽的最适TDZ浓度为1.0 mg·L-1,而阳光玫瑰葡萄诱导愈伤组织的适宜TDZ浓度为2.0~2.5 mg·L-1。
表4 TDZ浓度对葡萄试管苗叶片离体再生的影响
Table 4 Effects of TDZ concentration on regeneration from the leaves of grape plantlets in vitro
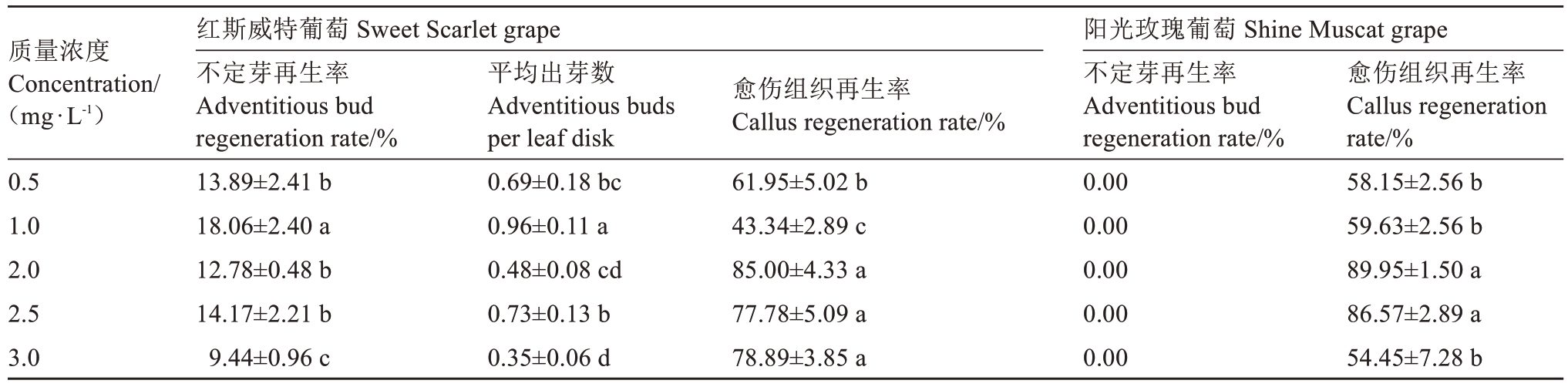
质量浓度Concentration/(mg·L-1)0.5 1.0 2.0 2.5 3.0红斯威特葡萄 Sweet Scarlet grape不定芽再生率Adventitious bud regeneration rate/%13.89±2.41 b 18.06±2.40 a 12.78±0.48 b 14.17±2.21 b 9.44±0.96 c平均出芽数Adventitious buds per leaf disk 0.69±0.18 bc 0.96±0.11 a 0.48±0.08 cd 0.73±0.13 b 0.35±0.06 d愈伤组织再生率Callus regeneration rate/%61.95±5.02 b 43.34±2.89 c 85.00±4.33 a 77.78±5.09 a 78.89±3.85 a阳光玫瑰葡萄 Shine Muscat grape不定芽再生率Adventitious bud regeneration rate/%0.00 0.00 0.00 0.00 0.00愈伤组织再生率Callus regeneration rate/%58.15±2.56 b 59.63±2.56 b 89.95±1.50 a 86.57±2.89 a 54.45±7.28 b
2.3 不同基本培养基及SNP浓度对葡萄试管苗叶片离体再生不定芽的影响
在MS与B5基本培养基中添加不同浓度SNP的试验表明,培养基类型及SNP浓度对红斯威特和阳光玫瑰葡萄试管苗叶片离体再生不定芽具有显著影响(表5)。红斯威特葡萄在B5培养基中的不定芽再生效果总体上优于MS培养基,当添加10 mg·L-1 SNP时,不定芽再生率达25.46%,平均出芽数达1.82个,较不含SNP的B5对照组(不定芽再生率20.57%,平均出芽数0.85个)明显提升,且平均出芽数显著高于其他浓度处理(P<0.05)。在B5+1.0 mg·L-1 TDZ+0.1 mg·L-1 IAA+15 m·L-1 葡萄糖+10 mg·L-1 SNP培养基中,离体叶片边缘及表面可见簇生的鲜绿饱满不定芽(图1-A)。阳光玫瑰葡萄离体叶片在不含SNP的培养基中无法再生不定芽,只在附加了SNP的培养基中才能诱导出芽,综合不定芽再生率和平均出芽数的结果,以B5+8 mg·L-1 SNP培养基表现最佳,不定芽再生率为17.03%,平均出芽数1.80个,总出芽数显著高于其他处理(P<0.05)。形态学观察显示(图1-B),在B5+1.0 mg·L-1 TDZ+0.1 mg·L-1 IAA+15 g·L-1葡萄糖+8 mg·L-1 SNP培养基中,阳光玫瑰葡萄不定芽多着生于叶脉附近,且排列整齐,与表5中17.03%的不定芽再生率统计数据相互印证。综上所述,在培养基中添加适宜浓度的SNP对两个葡萄品种叶片不定芽再生均有显著促进效应,红斯威特葡萄和阳光玫瑰葡萄最适SNP浓度分别为10 mg·L-1和8 mg·L-1。
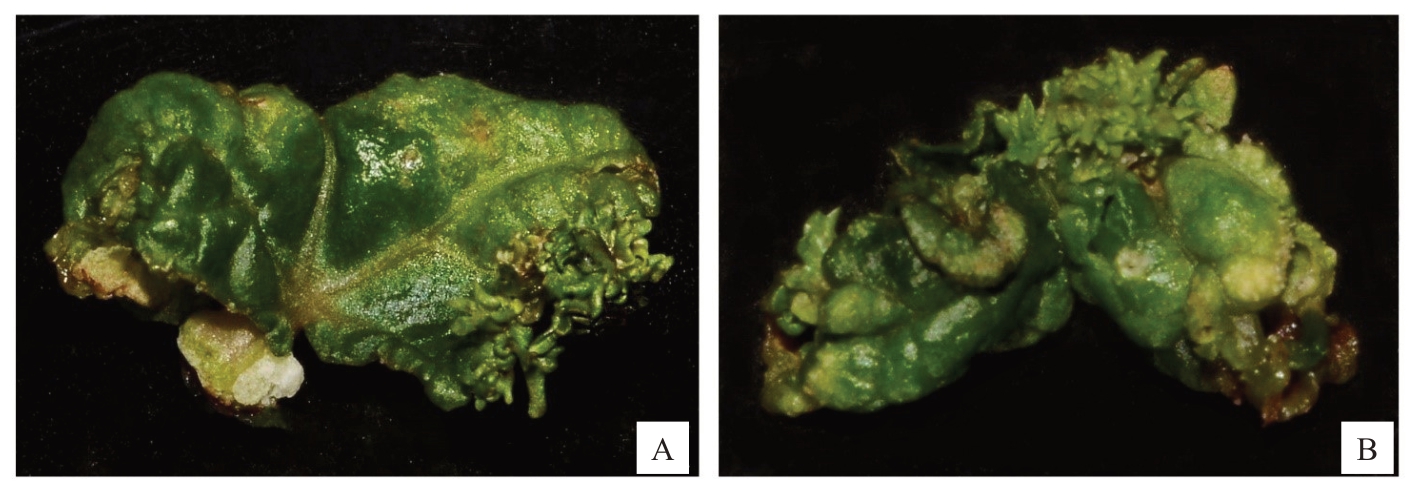
图1 红斯威特(A)和阳光玫瑰(B)葡萄试管苗离体叶片再生不定芽
Fig. 1 The adventitious buds that regenerated from the leaves of Sweet Scarlet (A) and Shine Muscat (B) plantlets in vitro
表5 不同基本培养基及SNP浓度对葡萄试管苗叶片离体再生不定芽的影响
Table 5 Effects of different basic medium and SNP concentration on regeneration from the leaves of grape plantlets in vitro
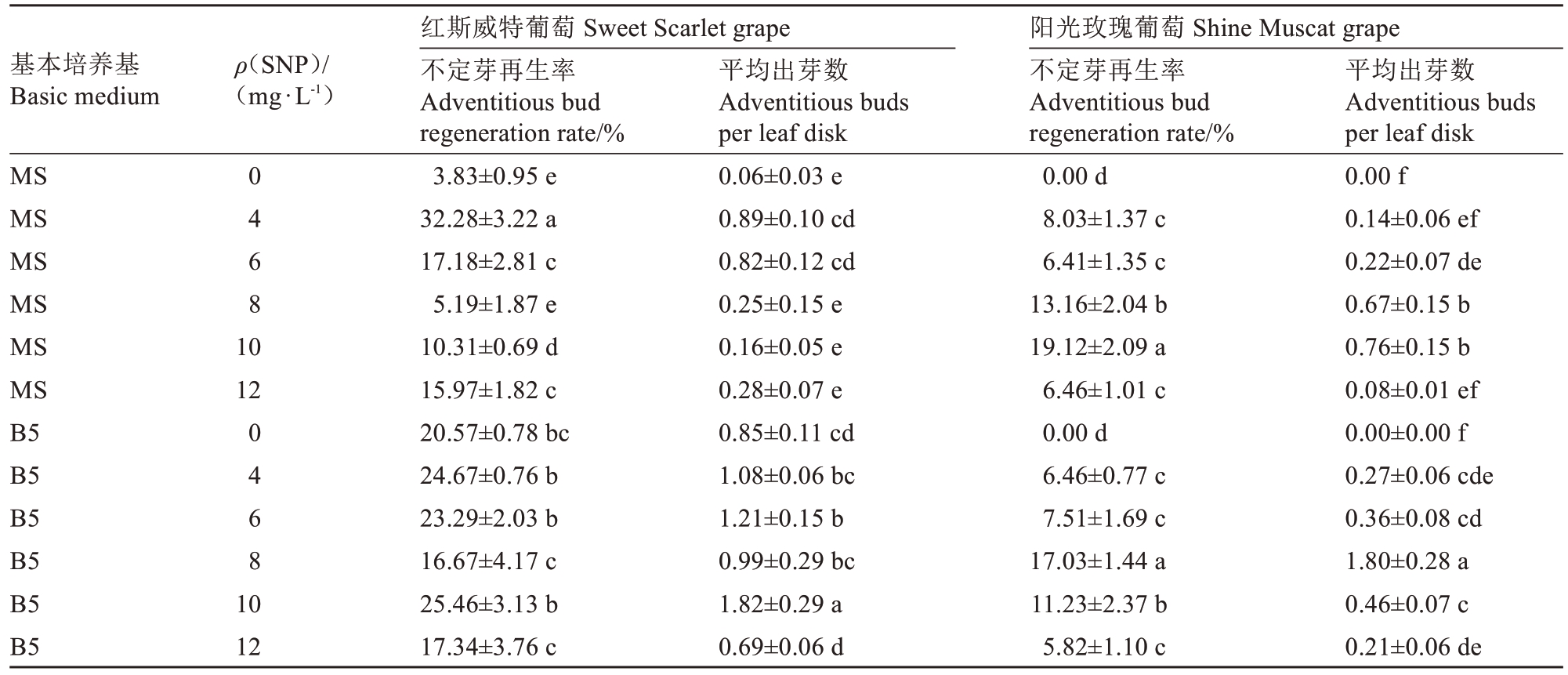
基本培养基Basic medium ρ(SNP)/(mg·L-1)MS MS MS MS MS MS B5 B5 B5 B5 B5 B5 0 4 6 8 10 12 0 4 6 8 10 12红斯威特葡萄 Sweet Scarlet grape不定芽再生率Adventitious bud regeneration rate/%3.83±0.95 e 32.28±3.22 a 17.18±2.81 c 5.19±1.87 e 10.31±0.69 d 15.97±1.82 c 20.57±0.78 bc 24.67±0.76 b 23.29±2.03 b 16.67±4.17 c 25.46±3.13 b 17.34±3.76 c平均出芽数Adventitious buds per leaf disk 0.06±0.03 e 0.89±0.10 cd 0.82±0.12 cd 0.25±0.15 e 0.16±0.05 e 0.28±0.07 e 0.85±0.11 cd 1.08±0.06 bc 1.21±0.15 b 0.99±0.29 bc 1.82±0.29 a 0.69±0.06 d阳光玫瑰葡萄 Shine Muscat grape不定芽再生率Adventitious bud regeneration rate/%0.00 d 8.03±1.37 c 6.41±1.35 c 13.16±2.04 b 19.12±2.09 a 6.46±1.01 c 0.00 d 6.46±0.77 c 7.51±1.69 c 17.03±1.44 a 11.23±2.37 b 5.82±1.10 c平均出芽数Adventitious buds per leaf disk 0.00 f 0.14±0.06 ef 0.22±0.07 de 0.67±0.15 b 0.76±0.15 b 0.08±0.01 ef 0.00±0.00 f 0.27±0.06 cde 0.36±0.08 cd 1.80±0.28 a 0.46±0.07 c 0.21±0.06 de
2.4 红斯威特葡萄试管苗通过叶片离体再生不定芽途径进行病毒脱除
将感染GRSPaV的红斯威特葡萄试管苗离体叶片接种于优化后的B5+1.0 mg·L-1 TDZ+0.1 mg·L-1 IAA+15 g·L-1 葡萄糖+10 mg·L-1 SNP培养基中诱导不定芽再生,然后将不定芽经转接至B5+0.5 mg·L-1 IAA+25 g·L-1 蔗糖培养基中,培养成试管苗(图2)。对7个再生无性系试管苗进行RT-PCR检测,结果显示,5个无性系已成功脱除GRSPaV,脱毒率达71.4%(图3)。其中,无性系1、2、4、5、7的检测结果与阴性对照一致,均未出现特异性条带,表明其已脱除病毒;而无性系3、6则与阳性对照相同,在905 bp处出现特异性扩增条带,表明其仍携带病毒。该结果证实,通过离体叶片直接再生不定芽途径可有效脱除红斯威特葡萄中的GRSPaV。

图2 红斯威特葡萄试管苗叶片离体再生的不定芽培养成苗
Fig. 2 The plantlets came from the adventitious buds that regenerated from the leaves of Sweet Scarlet grape plantlets in vitro
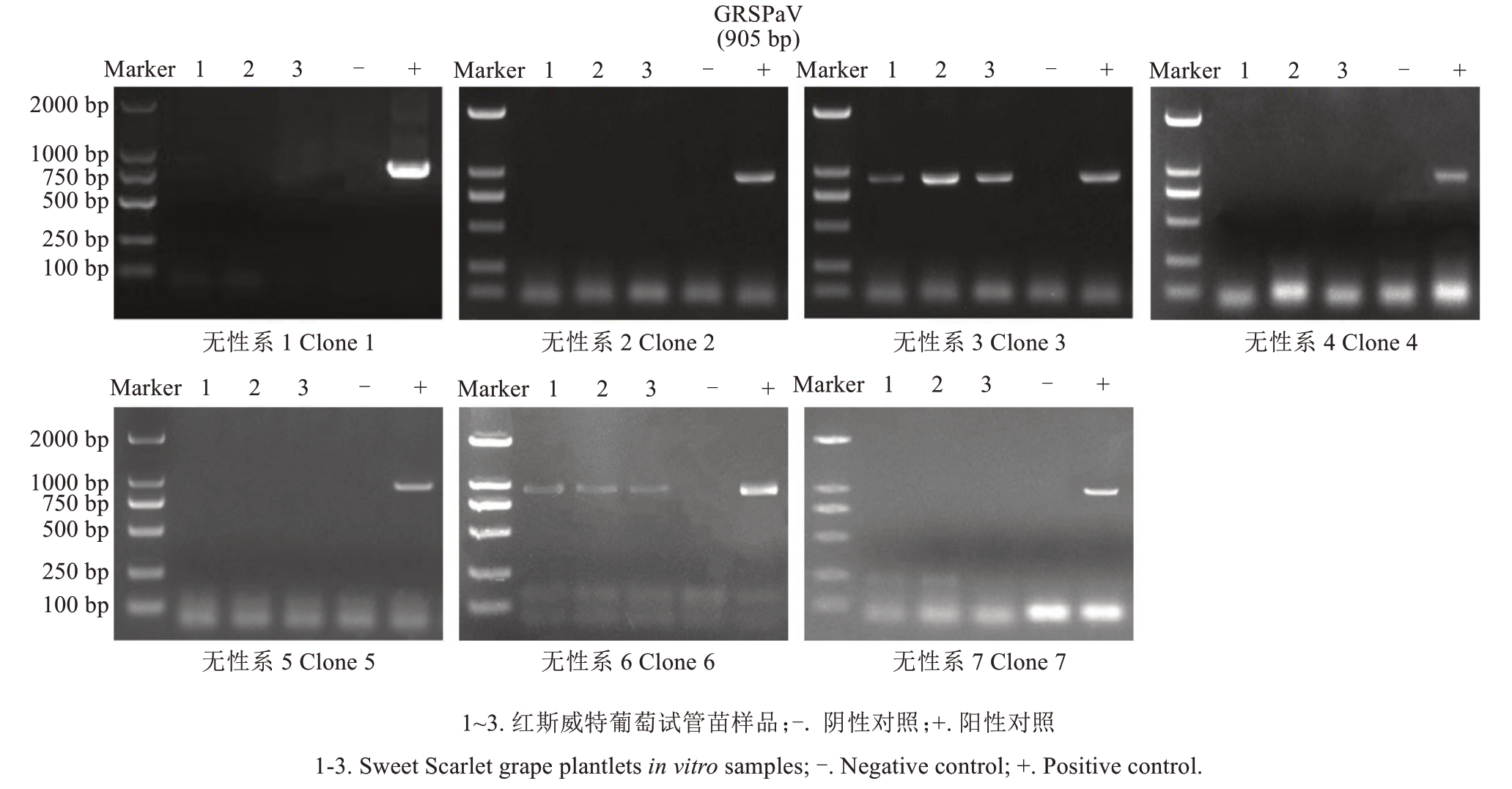
图3 红斯威特葡萄叶片离体再生植株病毒检测
Fig. 3 Virus detection for the plantlets in vitro that regenerated from the leaves of Sweet Scarlet grape
2.5 阳光玫瑰葡萄离体叶片再生愈伤组织和不定芽病毒脱除
将感染GVE、GRSPaV和GFabV的阳光玫瑰葡萄试管苗离体叶片诱导形成愈伤组织,转接至B5+1.0 mg·L-1 BA+0.1 mg·L-1 NAA+30 g·L-1葡萄糖培养基继代培养后,对不同代数愈伤组织进行RT-PCR检测(图4)。结果显示:第1代愈伤组织仍携带全部3种病毒;第2代脱除GFabV和GVE,仅检测到GRSPaV;第3代则完全脱除所有病毒,且第4代及后续继代培养的愈伤组织均保持无病毒状态。将无病毒愈伤组织转接至B5+2.0 mg·L-1 TDZ+0.2 mg·L-1 NAA+15 g·L-1 葡萄糖培养基中,成功诱导不定芽再生(图5),对不定芽培养成的试管苗进行GVE、GRSPaV和GFabV检测,结果均呈阴性,表明再生植株能够保持无病毒状态。
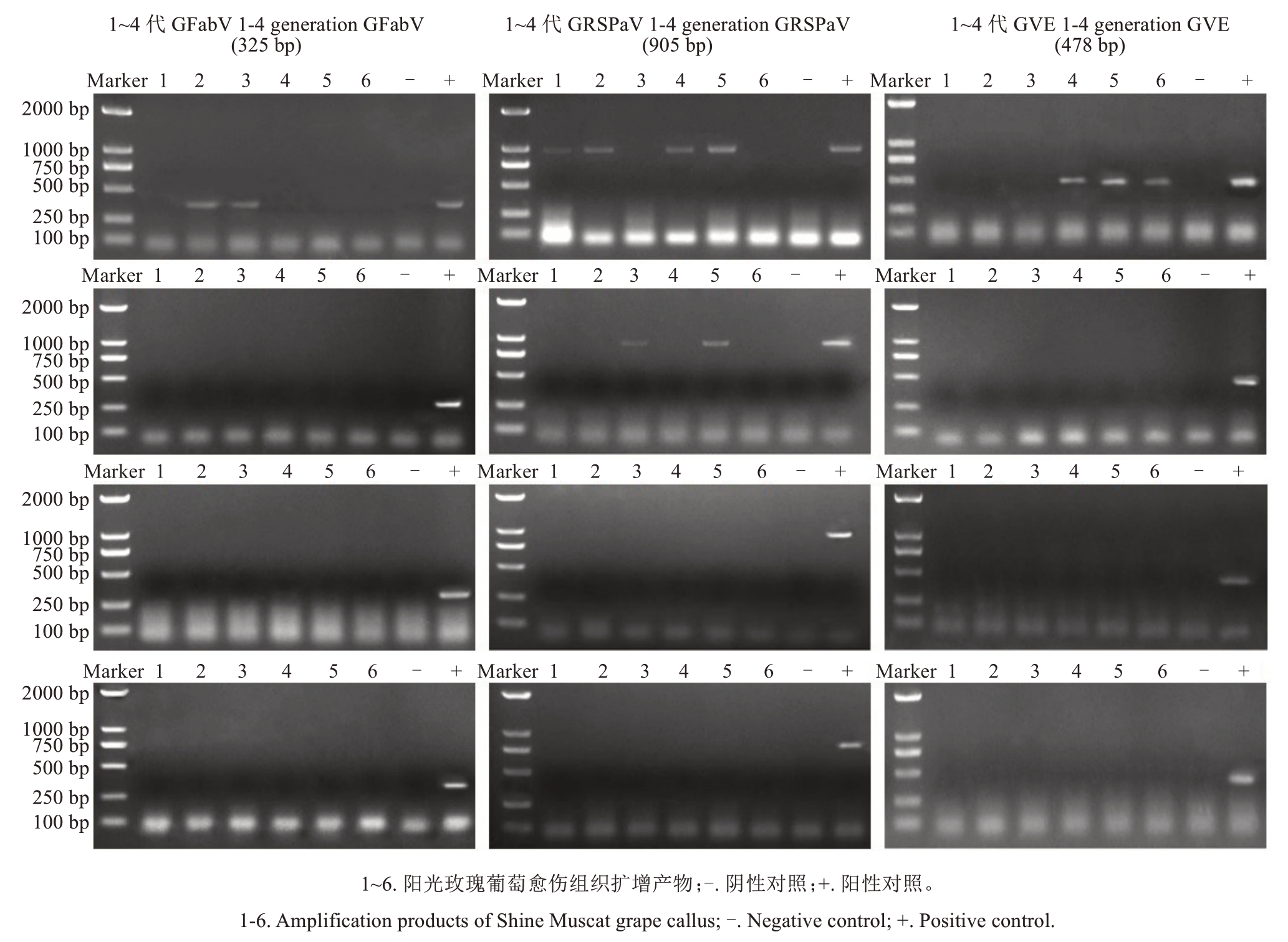
图4 阳光玫瑰葡萄试管苗叶片愈伤组织病毒检测
Fig. 4 Virus detection for the callus that generated from the leaves of Shine Muscat grape plantlets in vitro

图5 无病毒阳光玫瑰葡萄愈伤组织再生不定芽
Fig. 5 Regeneration of adventitious buds from the virus-free callus of Shine Muscat grape
3 讨 论
培育栽培无病毒苗对葡萄产业的健康可持续发展具有积极作用。不同葡萄品种携带的病毒种类不同,单独选择某一种方法进行脱毒存在一定的局限性,很难获得理想效果。最常见的热处理结合茎尖培养脱毒法虽然可以发挥两种技术的优势,降低了茎尖操作难度,提高了脱毒效率,但众多学者[7-11]在实际应用中发现,其对GRSPaV的脱除效果不佳。热处理仅对球状和类似纹状病毒有效,而对线状病毒(如GRSPaV)或杆状病毒不敏感[18]。胡国君等[19]研究表明,苹果病毒的脱除效率与病毒数量没有明显相关性,但与病毒种类关系密切。因此,针对不同病毒种类采用差异化的脱毒策略并研究多种脱毒途径是必要的。
在众多脱毒方法中,利用植物叶片、花药等器官再生实现脱毒的应用和报道较少。张春雨等[20]以0.3~0.4 mm珠葱茎尖为试材,通过诱导愈伤组织分化不定芽,成功脱除了葱潜隐病毒(SLV)、洋葱黄矮病毒(OYDV),脱毒率可达92%。李松等[21]以甘蔗茎尖愈伤组织为试材进行脱毒,发现经5次继代培养后完全脱除了花叶病毒。在本试验中,红斯威特葡萄试管苗的离体叶片可直接再生不定芽,并成功脱除了原有病毒;而阳光玫瑰葡萄试管苗的离体叶片直接再生不定芽较难,则对其采用了先脱分化产生愈伤组织,再分化成苗的二步诱导法。由于愈伤组织结构松散,薄壁细胞间联系不紧密,细胞分裂旺盛,增殖生长速度快于病毒繁殖速度,因此新增殖细胞常不携带病毒[12,22]。本试验通过对阳光玫瑰葡萄愈伤组织进行病毒检测,证实了脱毒效果。第1代愈伤组织仍携带全部3种病毒,第2代脱除了GVE和GFabV,仅检测到GRSPaV;第3代则完全脱除了所有病毒。将无病毒愈伤组织诱导不定芽再生,成功获得无病毒原种。这表明葡萄试管苗通过器官直接再生或愈伤组织再生途径脱除病毒具有可行性。
提高离体器官再生效率是实现葡萄器官再生脱毒的基础。再生过程中,植物激素起关键作用。细胞分裂素主要促进植物细胞分裂和不定芽分化,生长素则有助于诱导植物细胞分裂、增殖和愈伤组织的形成。因此,选择合适的细胞分裂素与生长素配比对葡萄离体再生至关重要[23]。本试验与多数文献报道相似,即细胞分裂素TDZ促进再生的效果显著优于BA,且与不同种类生长素(IAA、IBA、NAA)组合均可获得较好的不定芽再生效果[24-25]。然而,由于TDZ过强的细胞分裂作用,导致芽丛密集,甚至畸形,成苗非常困难。在本试验中,阳光玫瑰葡萄离体叶片诱导的不定芽转入继代培养基中数月都难以生长成苗,这可能与品种对细胞分裂素的敏感性有关。未来还需进一步研究,解决部分品种不定芽的成苗难题。
本研究也证实了SNP对葡萄器官再生的重要作用。Tun等[26]发现NO参与细胞分裂素的信号转导,并促进芽分化。在离体叶片再生过程中,SNP作为外源NO的供体,添加在培养基中能显著促进叶片与子叶不定芽分化[27]。韩小娇等[28]以平邑甜茶苹果叶片为试材,在培养基中添加6 mg·L-1 SNP,不定芽再生率可达67.8%。田春英等[29]研究表明,红富士苹果试管苗离体叶片在接种初期及后期不定芽形成时,NO含量处于较高水平,表明叶片不定芽再生过程与NO有关。在培养基中添加SNP促进红斯威特、阳光玫瑰葡萄试管苗离体叶片再生不定芽的效果显著,尤其是阳光玫瑰试管苗,其离体叶片仅在添加SNP的培养基中诱导出不定芽。不同基因型植株的再生能力不同,诱导分化所需SNP浓度不同。红斯威特和阳光玫瑰葡萄试管苗离体叶片不定芽再生的最适SNP浓度分别为10 mg·L-1和8 mg·L-1。此外NO作为重要的抗病信号分子,在植株抗病反应中发挥关键作用[30]。Kazemi等[31]将阿巴特、考西亚等5个梨品种的茎尖接种在添加SNP的培养基中,诱导成苗后进行病毒检测,发现ASPV脱毒率达100.0%,表明SNP作为抗病毒药剂可增强植株对病原体的抗性,显著提高脱毒率。本试验在葡萄试管苗离体叶片再生培养基中添加适宜浓度的SNP,除了可有效促进不定芽再生外,还可能对再生植株病毒脱除有一定作用。
4 结 论
TDZ促进红斯威特和阳光玫瑰葡萄试管苗离体叶片再生不定芽及愈伤组织形成的效果优于BA;在培养基中添加SNP可显著促进葡萄离体叶片不定芽再生,红斯威特葡萄适宜添加10 mg·L-1 SNP,阳光玫瑰葡萄适宜添加8 mg·L-1 SNP;红斯威特葡萄通过离体叶片直接再生不定芽,成功脱除GRSPaV;阳光玫瑰葡萄经愈伤组织继代培养,至第3代后脱除了原株所有病毒。这两种再生途径均能获得无病毒原种,表明器官再生途径可以用于葡萄试管苗的病毒脱除。
[1] TALQUENCA S G,ALONSO R,LUNA F,VOLPE M L,BUSCEMA F. Occurrence of nine grapevine viruses in commercial vineyards of Mendozɑ,Argentina[J] . Viruses,2023,15(1):177.
[2] 王澍,刘保军,张李娅,单佳祁,白剑宇,李克梅. 吐鲁番葡萄病毒病的发生情况及病原鉴定[J] . 植物检疫,2024,38(3):40-46.WANG Shu,LIU Baojun,ZHANG Liya,SHAN Jiaqi,BAI Jianyu,LI Kemei. Occurrence and pathogen identification of grape virus disease in Turpan[J] . Plant Quarantine,2024,38(3):40-46.
[3] 顾沛雯. 葡萄卷叶病毒的脱毒技术研究[J] . 西北农林科技大学学报(自然科学版),2008,36(5):85-91.GU Peiwen. Study on techniques of elimination of grapevine roll-leaf virtus[J] . Journal of Northwest A&F University (Natural Science Edition),2008,36(5):85-91.
[4] 胡国君,董雅凤,张尊平,范旭东,任芳,朱红娟. 葡萄病毒脱除技术研究进展[J] . 果树学报,2013,30(2):304-310.HU Guojun,DONG Yafeng,ZHANG Zunping,FAN Xudong,REN Fang,ZHU Hongjuan. Research progress on virus elimination techniques of grapevine[J] . Journal of Fruit Science,2013,30(2):304-310.
[5] 林茜,高营营,覃换玲,黄天琨,赵宇,王钟霞,陈淑媛. '阳光玫瑰'葡萄组培脱毒快繁技术研究[J] . 果树学报,2021,38(3):435-443.LIN Qian,GAO Yingying,QIN Huanling,HUANG Tiankun,ZHAO Yu,WANG Zhongxia,CHEN Shuyuan. Study on rapid propagation technology of virus-free seedlings by tissue culture in Shine Muscat grape[J] . Journal of Fruit Science,2021,38(3):435-443.
[6] ALJUAID B S,ATTIA A O,ISMAIL I A,DESSOKY E S,SADIK A S,KHALIFA M E. Production of virus-free rose plants using meristem-tip culture and in vitro thermotherapy[J] . Pakistan Journal of Biological Sciences,2022,25(2):160-167.
[7] SKIADA F G,GRIGORIADOU K,MALIOGKA V I,KATIS N I,ELEFTHERIOU E P. Elimination of grɑpevine leɑfroll-ɑssociɑted virus 1 and grɑpevine rupestrs stem pitting-ɑssociɑted virus from grapevine cv. Agiorgitiko,and a micropropagation protocol for mass production of virus-free plantlets[J] . Journal of Plant Pathology,2009,91(1):177-184.
[8] 杜易静,刘文林,乔月莲,王莉,安德志,杜国强,师校欣. ‘阳光玫瑰’葡萄试管苗热处理结合茎尖和腋芽培养脱毒技术研究[J] . 园艺学报,2024,51(4):893-902.DU Yijing,LIU Wenlin,QIAO Yuelian,WANG Li,AN Dezhi,DU Guoqiang,SHI Xiaoxin. Virus elimination from ‘Shine Muscat’ grape plantlets in vitro via heat treatment combined with shoot tip and axillary bud culture[J] . Acta Horticulturae Sinica,2024,51(4):893-902.
[9] GAMBINO G,CUOZZO D,FASOLI M,PAGLIARANI C,VITALI M,BOCCACCI P,PEZZOTTI M,MANNINI F. Co-evolution between grapevine rupestris stem pitting-associated virus and Vitis viniferɑ L. leads to decreased defence responses and increased transcription of genes related to photosynthesis[J] . Journal of Experimental Botany,2012,63(16):5919-5933.
[10] 张尊平,范旭东,胡国君,任芳,朱红娟,董雅凤. 葡萄试管苗热处理脱毒技术研究[J] . 中国果树,2013(1):39-41.ZHANG Zunping,FAN Xudong,HU Guojun,REN Fang,ZHU Hongjuan,DONG Yafeng. Studied on technics of virus elimination via heat treatment in grape in vitro[J] . China Fruits,2013(1):39-41.
[11] HU G J,DONG Y F,ZHANG Z P,FAN X D,REN F,LI Z N,ZHANG S N. Elimination of Grɑpevine rupestris stem pittingɑssociɑted virus from Vitis viniferɑ‘Kyoho’ by an antiviral agent combined with shoot tip culture[J] . Scientia Horticulturae,2018,229:99-106.
[12] 符国芳,李青. 植物组织培养脱毒方法综述[J] . 福建林业科技,2007,34(3):255-258.FU Guofang,LI Qing. Summarization on the methods of virus elimination by plant tissue culture[J] . Journal of Fujian Forestry Science and Technology,2007,34(3):255-258.
[13] 徐品三,栾雨时,刘纪文,新美芳二. 百合不定芽培养脱毒种球生产的研究[J] . 植物学通报,2003,20(3):313-318.XU Pinsan,LUAN Yushi,LIU Jiwen,XINMEI fanger. Study on production of virus-free bulblets by adventitious bud culture in Lilium spp.[J] . Chinese Bulletin of Botany,2003,20(3):313-318.
[14] 周美玲,曾军,张志勇,蔡建荣,江巍,张文斌. 黄果百香果茎段愈伤组织培养脱毒及脱毒苗快繁体系建立[J] . 江西农业学报,2022,34(9):97-101.ZHOU Meiling,ZENG Jun,ZHANG Zhiyong,CAI Jianrong,JIANG Wei,ZHANG Wenbin. Establishment of virus-free callus culture and rapid propagation system of virus-free seedlings from stem segments of yellow passion fruit[J] . Acta Agriculturae Jiangxi,2022,34(9):97-101.
[15] 常新,梁晋军,刘振华,肖坤,王燕,崔海洋,胡建芳. 黑香蕉葡萄不同器官发生途径再生体系的优化[J] . 中国农业大学学报,2018,23(9):43-52.CHANG Xin,LIANG Jinjun,LIU Zhenhua,XIAO Kun,WANG Yan,CUI Haiyang,HU Jianfang. Optimization of Vitis viniferɑ × V. lɑbruscɑ ‘Hei Xiangjiao’ adventitious buds regeneration viɑ organogenesis[J] . Journal of China Agricultural University,2018,23(9):43-52.
[16] MALETICH G,PUSHIN A,RYBALKIN E,PLUGATAR Y,DOLGOV S,KHVATKOV P. Organogenesis in a broad spectrum of grape genotypes and Agrobɑcterium-mediated transformation of the podarok magaracha grapevine cultivar[J] . Plants,2024,13(19):2779.
[17] 范旭东,董雅凤,张尊平,张梦妍,任芳,胡国君. ‘阳光玫瑰’葡萄病毒小RNA测序鉴定及RT-PCR检测[J] . 植物病理学报,2019,49(6):749-755.FAN Xudong,DONG Yafeng,ZHANG Zunping,ZHANG Mengyan,REN Fang,HU Guojun. Small RNA sequencing and RT-PCR detection of viruses infecting ‘Shine Muscat’ grapevines[J] . Acta Phytopathologica Sinica,2019,49(6):749-755.
[18] 陈冲,曹贵寿,王国平,刘伟,樊新平,霍辰思,史华平. 果树脱毒技术研究进展[J] . 果树资源学报,2021,2(1):72-75.CHEN Chong,CAO Guishou,WANG Guoping,LIU Wei,FAN Xinping,HUO Chensi,SHI Huaping. Research progress on virus free technique of fruit tree[J] . Journal of Fruit Resources,2021,2(1):72-75.
[19] 胡国君,张尊平,范旭东,任芳,翟秀丽,时晓燕,董雅凤. 苹果试管苗携带病毒种类数量与脱除效率的关系[J] . 中国果树,2022(5):57-61.HU Guojun,ZHANG Zunping,FAN Xudong,REN Fang,ZHAI Xiuli,SHI Xiaoyan,DONG Yafeng. Relationship between the number of virus species infected in in vitro apple plantlets and elimination efficiency[J] . China Fruits,2022(5):57-61.
[20] 张春雨,王晨,刘建青,战笑蕾,李小宇,王永志. 珠葱组培脱毒技术研究[J] . 园艺学报,2024,51(6):1426-1434.ZHANG Chunyu,WANG Chen,LIU Jianqing,ZHAN Xiaolei,LI Xiaoyu,WANG Yongzhi. Establishment of tissue culture and virus eliminating technology for shallot[J] . Acta Horticulturae Sinica,2024,51(6):1426-1434.
[21] 李松,余坤兴,刘丽敏,淡明,刘红坚,杨柳,谭芳,游建华,戴友铭. 甘蔗茎尖胚状体脱毒苗快繁技术研究[J] . 江苏农业科学,2011,39(2):83-87.LI Song,YU Kunxing,LIU Limin,DAN Ming,LIU Hongjian,YANG Liu,TAN Fang,YOU Jianhua,DAI Youming. Sugarcane micropropagation through shoot tip embryogenesis[J] . Jiangsu Agricultural Sciences,2011,39(2):83-87.
[22] 覃兰英,邓世秀,李青,杨满昌,陈巽祯,付仓生,李振兰. 培育草莓脱毒苗方法的研究[J] . 园艺学报,1988,15(3):175-179.QIN Lanying,DENG Shixiu,LI Qing,YANG Manchang,CHEN Xunzhen,FU Cangsheng,LI Zhenlan. Studies on the culture for the production of virus-free strawberry plant[J] . Acta Horticulturae Sinica,1988,15(3):175-179.
[23] TORREGROSA L,BOUQUET A,GOUSSARD P G. In vitro culture and propagation of grapevine[M] //ROUBELAKIS-ANGELAKIS K. Molecular Biology & Biotechnology of the Grapevine. Dordrecht:Springer Netherlands,2001:281-326.
[24] 代培红,徐明,尤扬子,刘超,罗淑萍,李月. 木纳格葡萄组织培养和再生体系的建立[J] . 分子植物育种,2022,20(1):246-251.DAI Peihong,XU Ming,YOU Yangzi,LIU Chao,LUO Shuping,LI Yue. Tissue culture and establishment of regeneration system of Vitis viniferɑ cv. Munage[J] . Molecular Plant Breeding,2022,20(1):246-251.
[25] WADA M,NISHITANI C,KOMORI S. Stable and efficient transformation of apple[J] . Plant Biotechnology,2020,37(2):163-170.
[26] TUN N N,HOLK A,SCHERER G F E. Rapid increase of NO release in plant cell cultures induced by cytokinin[J] . FEBS Letters,2001,509(2):174-176.
[27] MOREAU M,LINDERMAYR C,DURNER J,KLESSIG D F.NO synthesis and signaling in plants:Where do we stand?[J] .Physiologia Plantarum,2010,138(4):372-383.
[28] 韩小娇,杨洪强,由淑贞,段凯旋,张鑫荣,赵海洲. 平邑甜茶叶片不定芽再生及NO的效应[J] . 园艺学报,2008,35(3):419-422.HAN Xiaojiao,YANG Hongqiang,YOU Shuzhen,DUAN Kaixuan,ZHANG Xinrong,ZHAO Haizhou. Adventitious shoot regeneration from leaves of Mɑlus hupehensis and effects of nitric oxide[J] . Acta Horticulturae Sinica,2008,35(3):419-422.
[29] 田春英,邵建柱,刘莹,徐继忠. 红富士苹果叶片不定芽再生中激素、多胺和NO含量的变化[J] . 园艺学报,2010,37(9):1403-1408.TIAN Chunying,SHAO Jianzhu,LIU Ying,XU Jizhong. Changes of hormones,polyamines and NO content during regeneration of adventitious buds from in vitro leaves of Red Fuji apple[J] .Acta Horticulturae Sinica,2010,37(9):1403-1408.
[30] 王晨. NO信号分子及其在植物抗病反应中的作用研究综述[J] . 现代园艺,2019(11):42-44.WANG Chen. A review of research on NO signaling molecules and their role in plant disease resistance responses[J] . Xiandai Horticulture,2019(11):42-44.
[31] KAZEMI N,NAHANDI F Z,HABASHI A A,MASOOMI-ALADIZGEH F. Comparing the efficiency of conventional and novel methods of virus elimination using molecular techniques[J] . European Journal of Plant Pathology,2020,157(4):887-897.