石榴(Punicɑ grɑnɑtum L.)为千屈菜科(Lythraceae)石榴属(Punicɑ L.)落叶灌木或小乔木[1],是世界上重要的经济林树种之一。果实富含维生素C等多种营养成分,具有抗氧化、抗炎、抗癌和预防心血管疾病等重要功能[2]。石榴在中国栽培历史悠久,2023年全国的栽培面积11万hm2,年产量200万t[3]。山东省是全国石榴主产区之一,目前栽培面积达1万hm2,年产量10万t,石榴种植是农民增收致富的重要产业[4]。石榴为异花授粉果树,由于栽培过程中自发突变、不同地域间的引种、品种交换等造成亲缘关系模糊,谱系混乱现象突显,严重影响了种质资源的高效利用。传统上对石榴种质间的遗传关系、品种鉴定经常依据花色、瓣型、果色等表型性状,但这些性状极易受环境影响,导致结果不准确。市场上“假树苗”以次充好现象层出不穷,造成新品种权得不到保护,极大地影响了育种单位的积极性和果农的经济利益。随着商业化育种进程加速,石榴种质资源同质化现象加剧,传统形态学鉴定方法难以满足精准分类需求,分子标记技术的发展为解决这类问题提供了新途径。
采用DNA标记技术鉴定石榴种质能从分子水平解析种质间的遗传差异,比形态学、细胞学更准确[5-7]。目前,多种分子标记已应用于研究石榴种质资源的遗传关系分析,其中RAPD标记使用最广泛但不稳定。因此,AFLP、SSR等分子标记更多地应用于石榴属植物间的遗传关系研究[8]。基于SSR技术建立的ISSR分子标记无需预知物种基因组信息,多态性高、重复性好、成本较低,在植物遗传多样性、品种鉴定、遗传图谱构建等研究中应用广泛,目前已用于樱桃[9]、枇杷[10]、番石榴[11]、桃[12]、匙羹藤[13]等植物种质资源的研究。然而,目前大多数研究多聚焦于局部种质资源,缺乏系统性分析。本研究采用ISSR技术,系统分析山东产区石榴种质资源的遗传多样性、亲缘关系并构建指纹图谱数据库,为石榴的科学分类、品种鉴定和遗传改良提供理论基础和科学依据,对今后优异种质的挖掘及高效利用具有重要意义。
1 材料和方法
1.1 材料来源
供试76份山东石榴种质资源均来自中国石榴种质资源圃(位于山东省枣庄市),石榴种质资源信息见表1。
表1 76份石榴种质资源信息
Table 1 Information on 76 pomegranate germplasm accessions
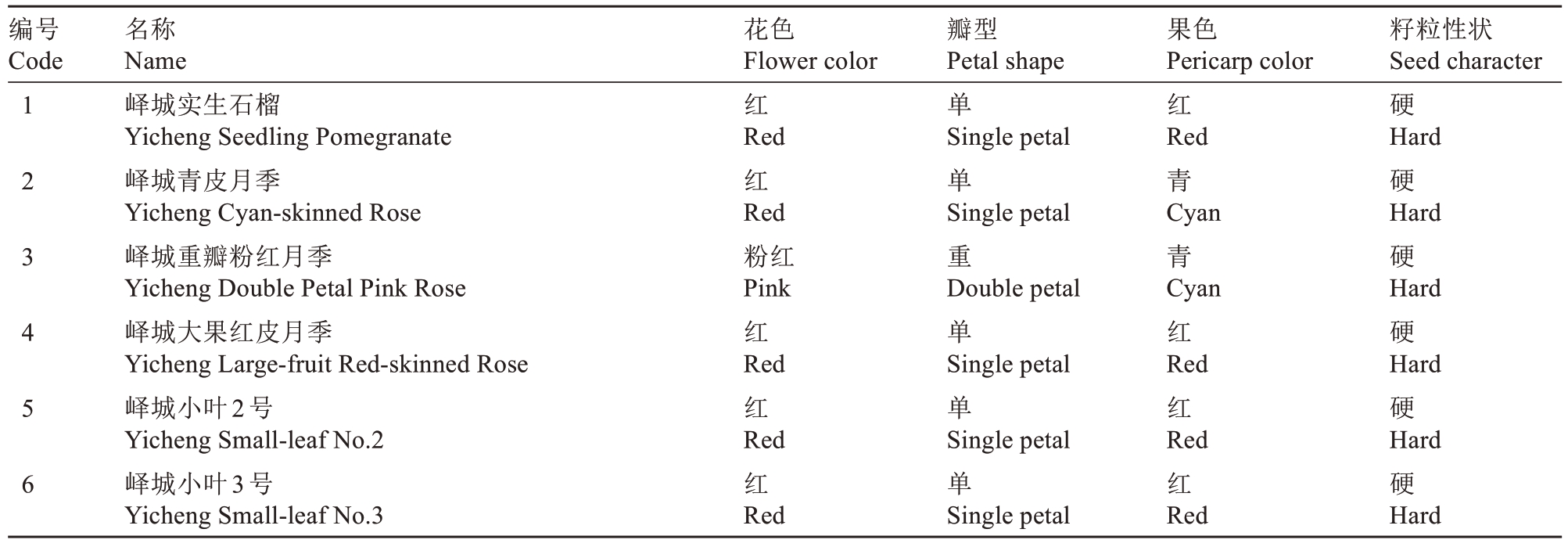
编号Code名称Name花色Flower color瓣型Petal shape果色Pericarp color籽粒性状Seed character 1 2 3 4 5 6峄城实生石榴Yicheng Seedling Pomegranate峄城青皮月季Yicheng Cyan-skinned Rose峄城重瓣粉红月季Yicheng Double Petal Pink Rose峄城大果红皮月季Yicheng Large-fruit Red-skinned Rose峄城小叶2号Yicheng Small-leaf No.2峄城小叶3号Yicheng Small-leaf No.3红单红硬Red红Red粉红Pink红Red红Red红Red Single petal单Single petal重Double petal单Single petal单Single petal单Single petal Red青Cyan青Cyan红Red红Red红Red Hard硬Hard硬Hard硬Hard硬Hard硬Hard
表1(续) Table 1 (Continued)

编号Code名称Name花色Flower color瓣型Petal shape果色Pericarp color籽粒性状Seed character 7 8 9白单白硬10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34峄城小籽三白Yicheng Small-seed Sanbai峄城大个红皮Yicheng Big Pomegranate Red-skinned峄城小叶红皮Yicheng Small-leaf Red-skinned峄城大叶Yicheng Large-leaf峄城红籽白石榴Yicheng Red-seed White Pomegranate峄城红皮月季Yicheng Red-skinned Rose峄城发芽红Yicheng Sprouting Red峄城白皮酸Yicheng White-skinned Sour峄城单瓣粉红白皮酸Yicheng Single Petal Pink White-skinned Sour峄城单瓣玛瑙Yicheng Single Petal Agate峄城红牡丹石榴Yicheng Red Peony Pomegranate峄城重瓣白皮酸Yicheng Double Petal White-skinned Sour峄城小红牡丹Yicheng Small Red Peony峄城抗寒砧木1号Yicheng Cold-resistant Rootstock No.1峄城竹叶青Yicheng Bamboo-leaf Cyan峄城重瓣白花酸Yicheng Double Petal White Flower Sour峄城超红Yicheng Ultra Red峄城青皮大籽Yicheng Cyan-skinned Large-seed峄城白楼无刺Yicheng Bailou Thornless峄城和顺庄无刺Yicheng Heshunzhuang Thornless峄城紫粒青皮甜Yicheng Purple-seed Cyan-skinned Sweet峄城超青Yicheng Ultra Cyan峄城超大白皮甜Yicheng Extra-large White-skinned Sweet峄红1号Yihong No.1峄城重瓣红皮酸Yicheng Double Petal Red-skinned Sour峄城大粒青皮岗榴Yicheng Large-seed Cyan-skinned Gangliu峄城峄青Yicheng Yiqing峄城单瓣粉红酸Yicheng Single Petal Pink Sour White红Red红Red红Red白White红Red红Red白White粉红Pink复色Multiple红Red白White红Red红Red红Red白White红Red红Red红Red红Red红Red红Red白White红Red红Red红Red红Red粉红Pink Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal复Multiple petal复Multiple petal重Double petal重Double petal单Single petal复Multiple petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal重Double petal单Single petal单Single petal单Single petal White红Red红Red红Red白White红Red红Red白White白White乳白Milky white红Red白White红Red红Red青Cyan白White红Red青Cyan青Cyan青Cyan青Cyan青Cyan白White红Red红Red青Cyan青Cyan青Cyan Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard
表1(续) Table 1 (Continued)
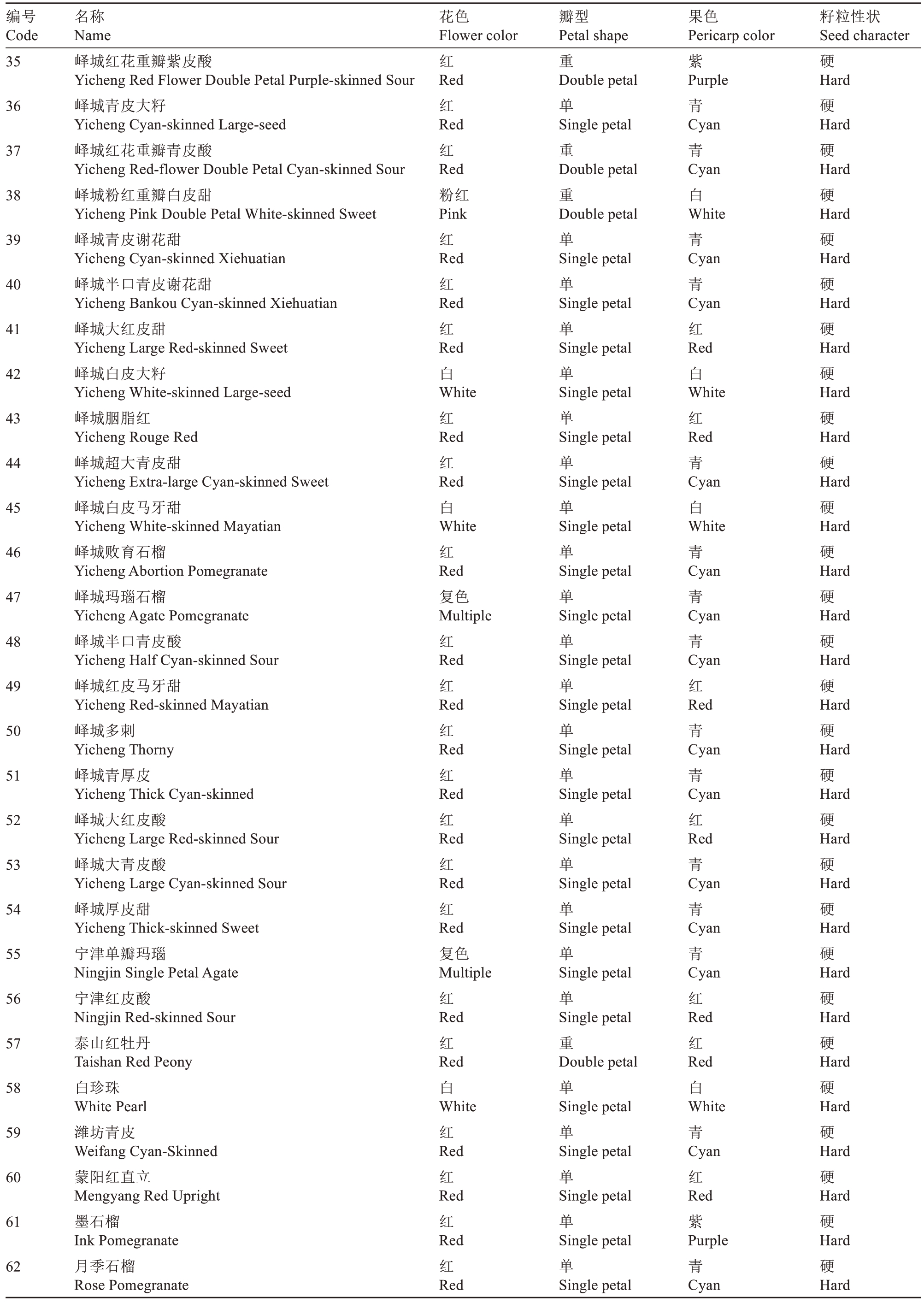
编号Code名称Name花色Flower color瓣型Petal shape果色Pericarp color籽粒性状Seed character 35红重紫硬36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62峄城红花重瓣紫皮酸Yicheng Red Flower Double Petal Purple-skinned Sour峄城青皮大籽Yicheng Cyan-skinned Large-seed峄城红花重瓣青皮酸Yicheng Red-flower Double Petal Cyan-skinned Sour峄城粉红重瓣白皮甜Yicheng Pink Double Petal White-skinned Sweet峄城青皮谢花甜Yicheng Cyan-skinned Xiehuatian峄城半口青皮谢花甜Yicheng Bankou Cyan-skinned Xiehuatian峄城大红皮甜Yicheng Large Red-skinned Sweet峄城白皮大籽Yicheng White-skinned Large-seed峄城胭脂红Yicheng Rouge Red峄城超大青皮甜Yicheng Extra-large Cyan-skinned Sweet峄城白皮马牙甜Yicheng White-skinned Mayatian峄城败育石榴Yicheng Abortion Pomegranate峄城玛瑙石榴Yicheng Agate Pomegranate峄城半口青皮酸Yicheng Half Cyan-skinned Sour峄城红皮马牙甜Yicheng Red-skinned Mayatian峄城多刺Yicheng Thorny峄城青厚皮Yicheng Thick Cyan-skinned峄城大红皮酸Yicheng Large Red-skinned Sour峄城大青皮酸Yicheng Large Cyan-skinned Sour峄城厚皮甜Yicheng Thick-skinned Sweet宁津单瓣玛瑙Ningjin Single Petal Agate宁津红皮酸Ningjin Red-skinned Sour泰山红牡丹Taishan Red Peony白珍珠White Pearl潍坊青皮Weifang Cyan-Skinned蒙阳红直立Mengyang Red Upright墨石榴Ink Pomegranate月季石榴Rose Pomegranate Red红Red红Red粉红Pink红Red红Red红Red白White红Red红Red白White红Red复色Multiple红Red红Red红Red红Red红Red红Red红Red复色Multiple红Red红Red白White红Red红Red红Red红Red Double petal单Single petal重Double petal重Double petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal重Double petal单Single petal单Single petal单Single petal单Single petal单Single petal Purple青Cyan青Cyan白White青Cyan青Cyan红Red白White红Red青Cyan白White青Cyan青Cyan青Cyan红Red青Cyan青Cyan红Red青Cyan青Cyan青Cyan红Red红Red白White青Cyan红Red紫Purple青Cyan Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard
表1(续) Table 1 (Continued)
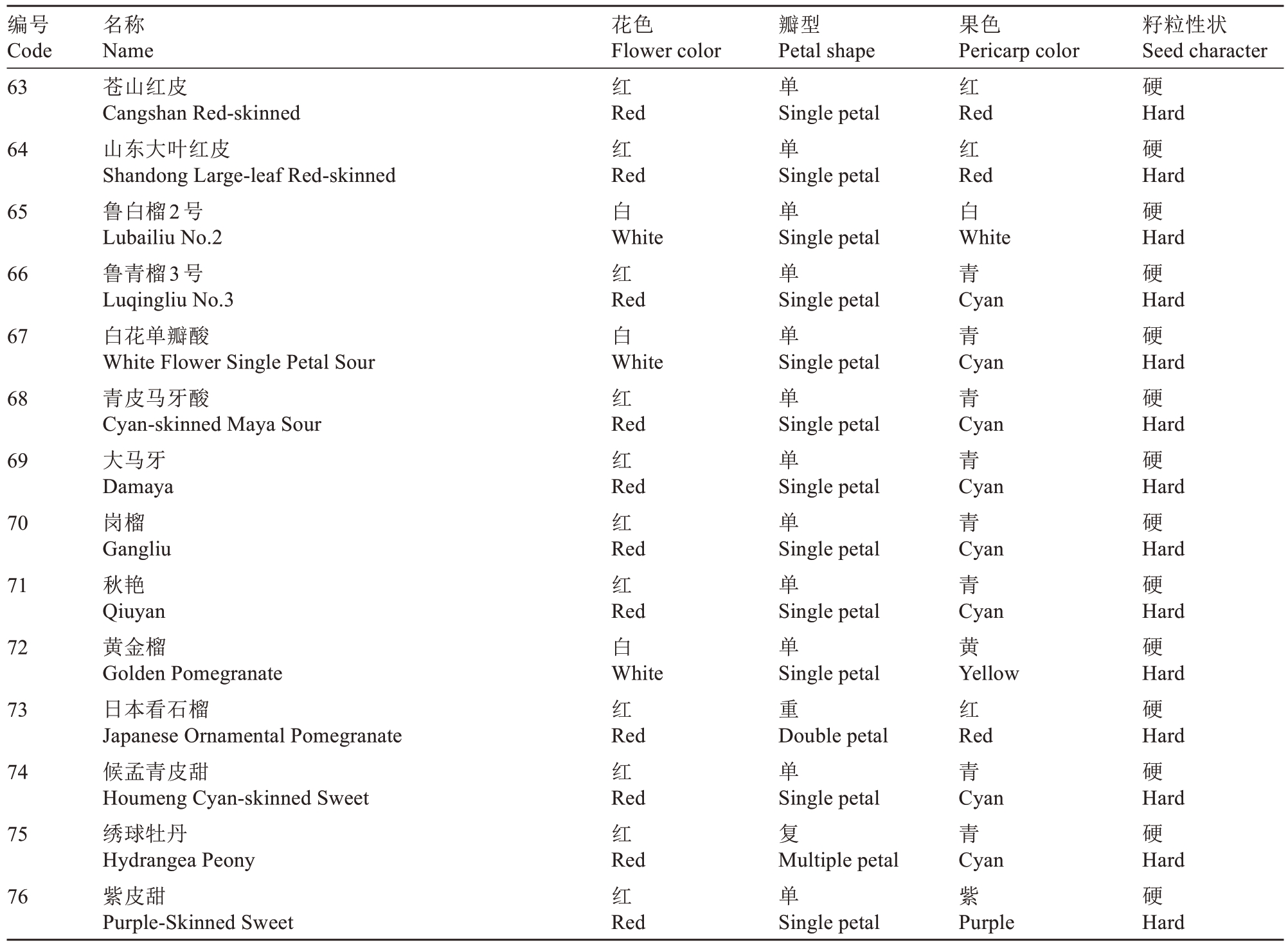
编号Code名称Name花色Flower color瓣型Petal shape果色Pericarp color籽粒性状Seed character 63红单红硬64 65 66 67 68 69 70 71 72 73 74 75 76苍山红皮Cangshan Red-skinned山东大叶红皮Shandong Large-leaf Red-skinned鲁白榴2号Lubailiu No.2鲁青榴3号Luqingliu No.3白花单瓣酸White Flower Single Petal Sour青皮马牙酸Cyan-skinned Maya Sour大马牙Damaya岗榴Gangliu秋艳Qiuyan黄金榴Golden Pomegranate日本看石榴Japanese Ornamental Pomegranate候孟青皮甜Houmeng Cyan-skinned Sweet绣球牡丹Hydrangea Peony紫皮甜Purple-Skinned Sweet Red红Red白White红Red白White红Red红Red红Red红Red白White红Red红Red红Red红Red Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal单Single petal重Double petal单Single petal复Multiple petal单Single petal Red红Red白White青Cyan青Cyan青Cyan青Cyan青Cyan青Cyan黄Yellow红Red青Cyan青Cyan紫Purple Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard硬Hard
1.2 DNA提取及ISSR-PCR扩增检测
石榴嫩叶基因组DNA提取、ISSR-PCR扩增条件及检测参考朱薇等[14]的方法。
1.3 数据分析
以DL2000 DNA Marker(天根生化科技有限公司,北京)的谱带分子质量为参考标准,用Quantity One软件结合肉眼观察获得扩增DNA谱带分子质量,根据谱带在凝胶同一迁移率位置上的有无,按照有扩增谱带记为“1”,无扩增谱带记为“0”的统计标准,形成每条引物标记的(0,1)数据原始矩阵。统计并计算引物多态性百分比率。引物多态性比率=a/(a+b)×100%,a为多态条带数目,b为所有品种共享数目。用PopGene软件统计遗传多样性参数。用NTSYS2.10软件计算遗传相似系数(GS),用UPGMA法绘制遗传关系图。通过聚类分析单引物或引物组合对供试品种的区分效率,品种区分率高的引物用于构建DNA指纹。
2 结果与分析
2.1 ISSR引物扩增结果分析
部分ISSR引物扩增PAGE电泳图如图1所示,供试品种扩增谱带清晰,产生的位点迁移率各异,多态性较好。由表2可知,16个引物所得位点数为14~25个,每条引物平均产生位点19个,多态性位点16.8个,引物多态性比率为70.6%~95.4%。引物UBC834扩增出来的位点最多,多态性比率较好,为92.0%。引物UBC827的多态性位点比率最高,为95.4%。引物平均多态性比率为88.2%,说明这16个引物适合山东石榴种质资源遗传多样性研究。
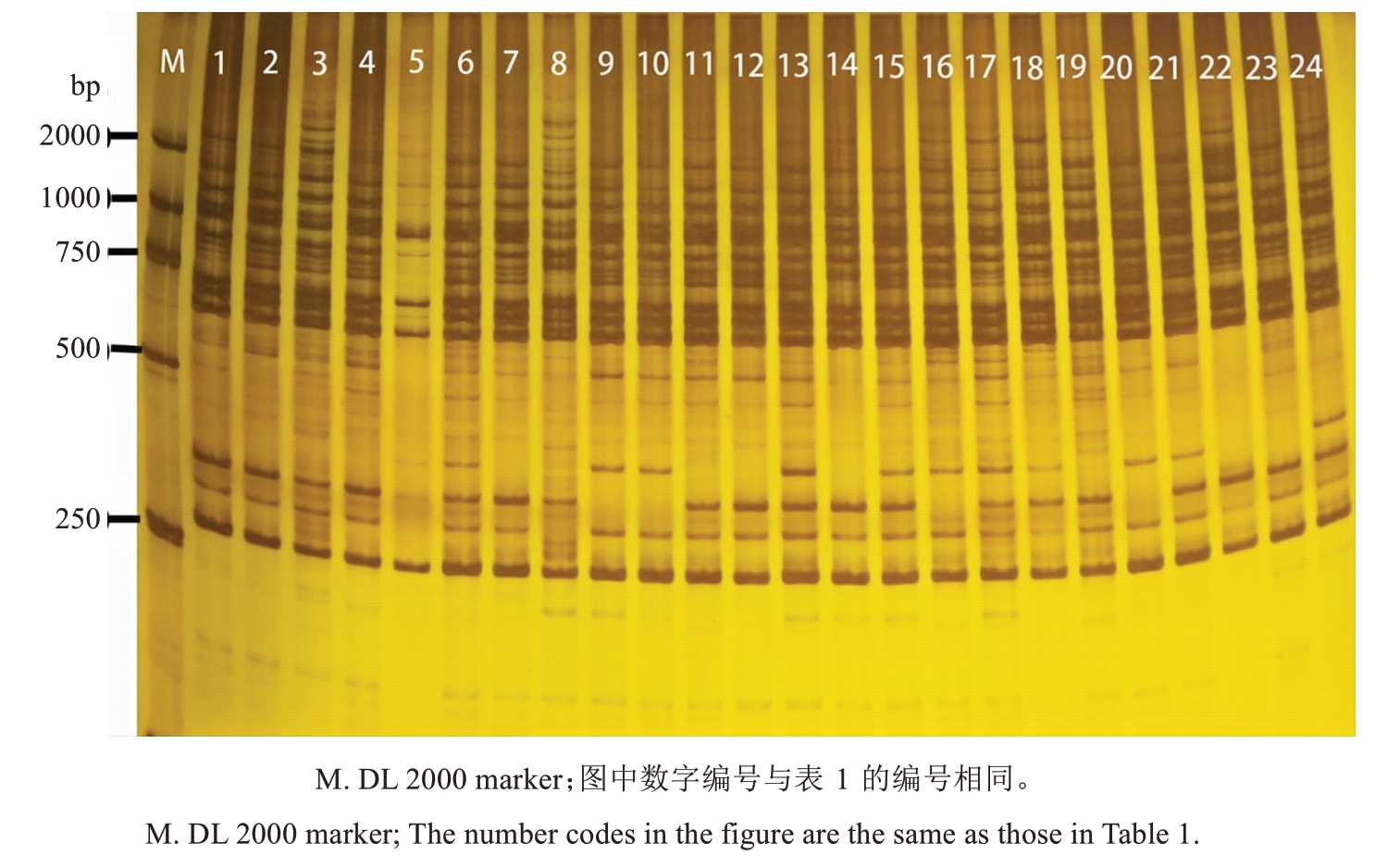
图1 引物UBC835对部分石榴种质的扩增结果
Fig. 1 Amplification results of primer UBC835 for some pomegranate germplasm accessions
表2 ISSR引物信息、扩增结果及品种区分效率
Table 2 ISSR primers information, amplification results and cluster discrimination rate
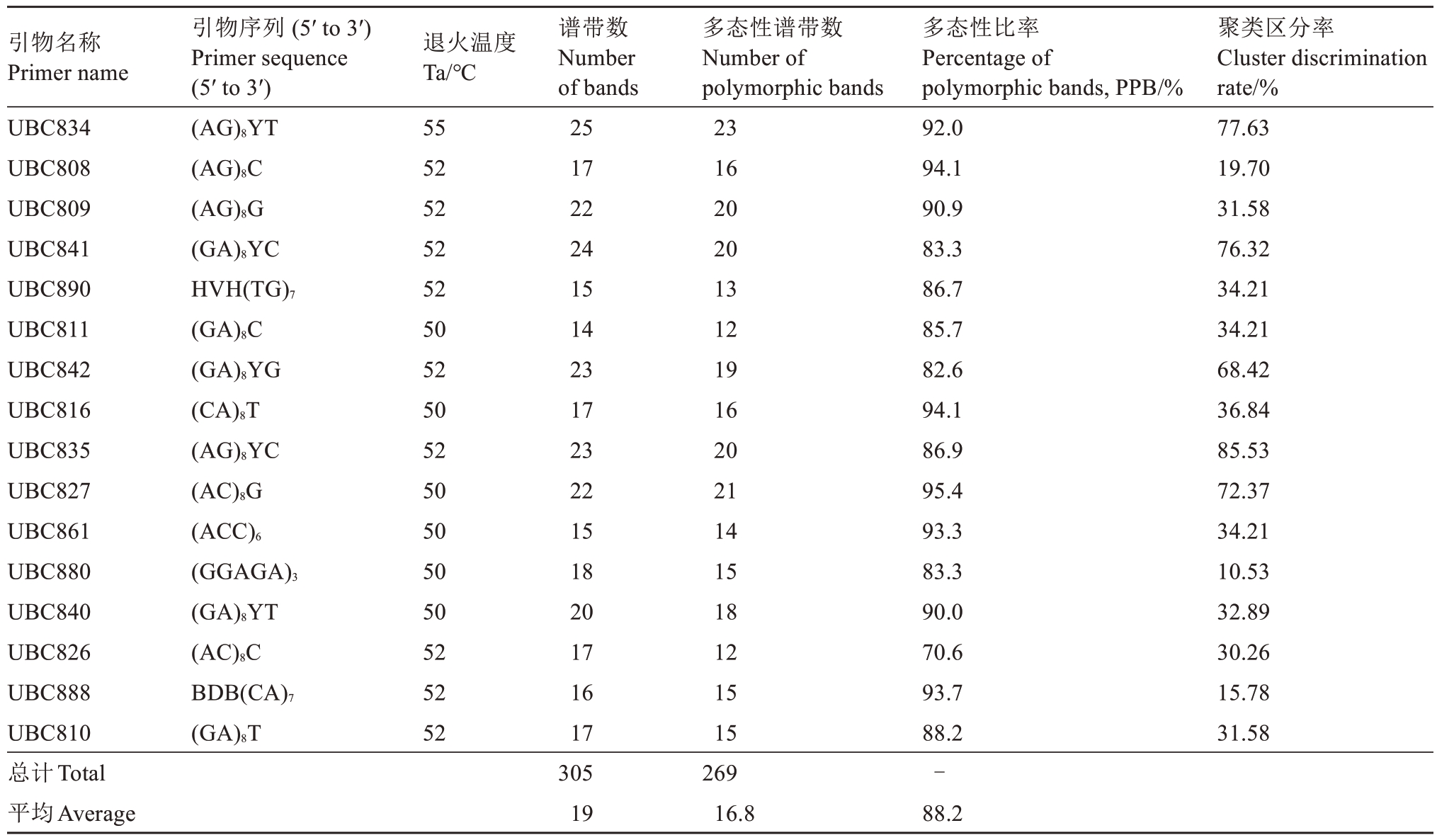
引物名称Primer name UBC834 UBC808 UBC809 UBC841 UBC890 UBC811 UBC842 UBC816 UBC835 UBC827 UBC861 UBC880 UBC840 UBC826 UBC888 UBC810总计Total平均Average引物序列 (5′ to 3′)Primer sequence(5′ to 3′)(AG)8YT(AG)8C(AG)8G(GA)8YC HVH(TG)7(GA)8C(GA)8YG(CA)8T(AG)8YC(AC)8G(ACC)6(GGAGA)3(GA)8YT(AC)8C BDB(CA)7(GA)8T退火温度Ta/℃55 52 52 52 52 50 52 50 52 50 50 50 50 52 52 52谱带数Number of bands 25 17 22 24 15 14 23 17 23 22 15 18 20 17 16 17 305 19多态性谱带数Number of polymorphic bands 23 16 20 20 13 12 19 16 20 21 14 15 18 12 15 15 269 16.8多态性比率Percentage of polymorphic bands, PPB/%92.0 94.1 90.9 83.3 86.7 85.7 82.6 94.1 86.9 95.4 93.3 83.3 90.0 70.6 93.7 88.2-88.2聚类区分率Cluster discrimination rate/%77.63 19.70 31.58 76.32 34.21 34.21 68.42 36.84 85.53 72.37 34.21 10.53 32.89 30.26 15.78 31.58
2.2 单引物的区分效率
统计每个引物对76份种质的区分效率,结果如表2所示:单引物的区分效率为10.53%~85.53%;引物UBC835的区分率最高,为85.53%,但仍有11份种质不能区分开。因此,单引物对76份种质的区分能力较差,需要筛选种质区分率较高的引物组合构建DNA指纹图谱。
2.3 核心引物组合的区分效率
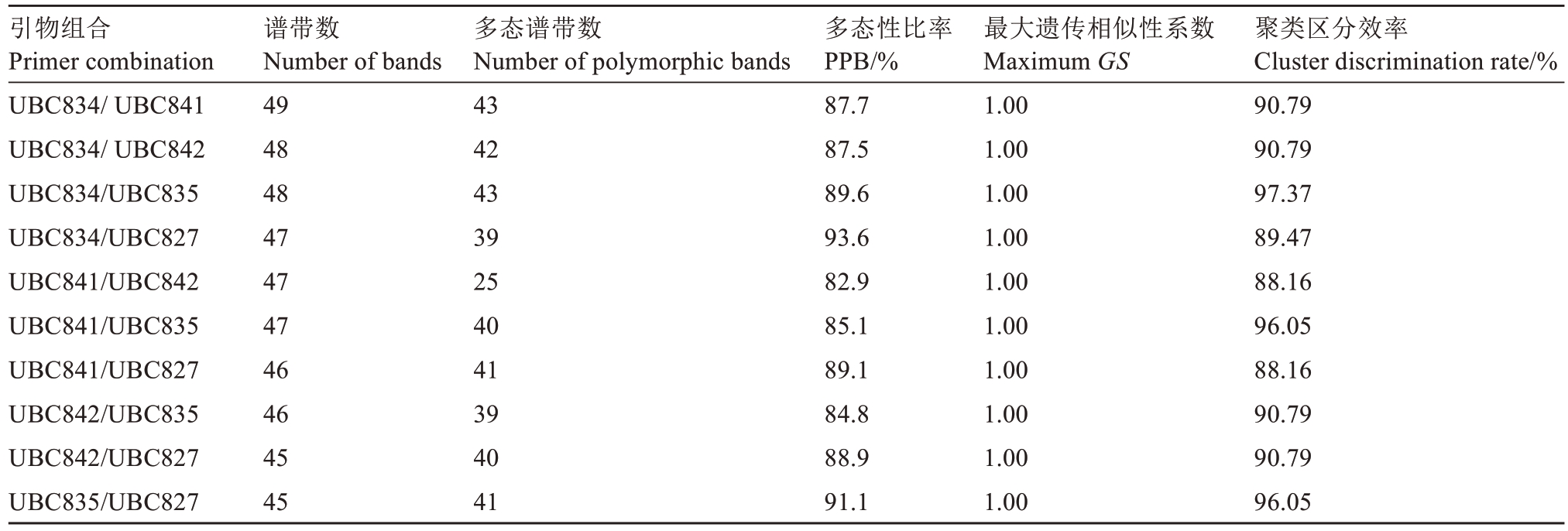
将区分效率较高的引物UBC827、UBC834、UBC835、UBC841和UBC842进行两两组合,分析10组引物对76份种质的区分效率(表3)。
表3 10组引物组合对76份种质的品种区分效率
Table 3 The discrimination rate for 76 accessions of 10 primer combinations
引物组合Primer combination UBC834/ UBC841 UBC834/ UBC842 UBC834/UBC835 UBC834/UBC827 UBC841/UBC842 UBC841/UBC835 UBC841/UBC827 UBC842/UBC835 UBC842/UBC827 UBC835/UBC827谱带数Number of bands 49 48 48 47 47 47 46 46 45 45多态谱带数Number of polymorphic bands 43 42 43 39 25 40 41 39 40 41多态性比率PPB/%87.7 87.5 89.6 93.6 82.9 85.1 89.1 84.8 88.9 91.1最大遗传相似性系数Maximum GS 1.00 1.00 1.00 1.00 1.00 1.00 1.00 1.00 1.00 1.00聚类区分效率Cluster discrimination rate/%90.79 90.79 97.37 89.47 88.16 96.05 88.16 90.79 90.79 96.05
从表3可知,10组引物组合的区分效率均高于单引物,区分效率为88.16%~97.37%,其中引物组合UBC834/UBC835的区分效率为97.37%,仅不能区分75号和76号种质(图2),其原因可能是这两份种质的遗传背景差异较小,本研究中所用引物难以鉴别。根据DNA指纹构建的原则,选取引物组合UBC834/UBC835构建76份种质的DNA指纹图谱(图2)。除第75号和第76号外,其他74份种质的DNA指纹图谱均不同,能够被高效准确地鉴定。

图2 76份石榴种质的DNA指纹图谱
Fig. 2 DNA fingerprints of 76 pomegranate germplasm accessions
2.4 山东石榴种质资源的遗传多样性与遗传关系分析
76份种质资源遗传多样性参数分析表明,Nɑ为1.980 7,Ne为1.651 7,He为0.370 4,I为0.545 1,这表明山东石榴种质资源遗传多样性比较丰富。利用NTSYS2.10软件进行遗传关系聚类,结果表明遗传相似系数GS为0.49~0.98,当GS阈值约为0.688时,将76份石榴种质分为4个类群。
由图3可知,第Ⅰ类群包含40份种质,该类群种质的花色有红色、粉红色,花瓣有单瓣、重瓣等,果实有观赏、食用等多种类型,分类结果与形态分类有较大差异。第Ⅱ类群有27份种质,该类群的花色、果色等形态性状也各异。第Ⅲ类群有2份种质,为峄城小叶2号和峄城单瓣粉红酸石榴。第Ⅳ类群有峄城青皮大籽、白楼无刺等7份种质,这些种质的形态性状也不尽相同。4个类群中,第Ⅲ和第Ⅳ类群的种质与其他种质遗传距离较大,可以选为杂交育种亲本材料,利用杂交优势进行石榴种质资源的遗传改良。
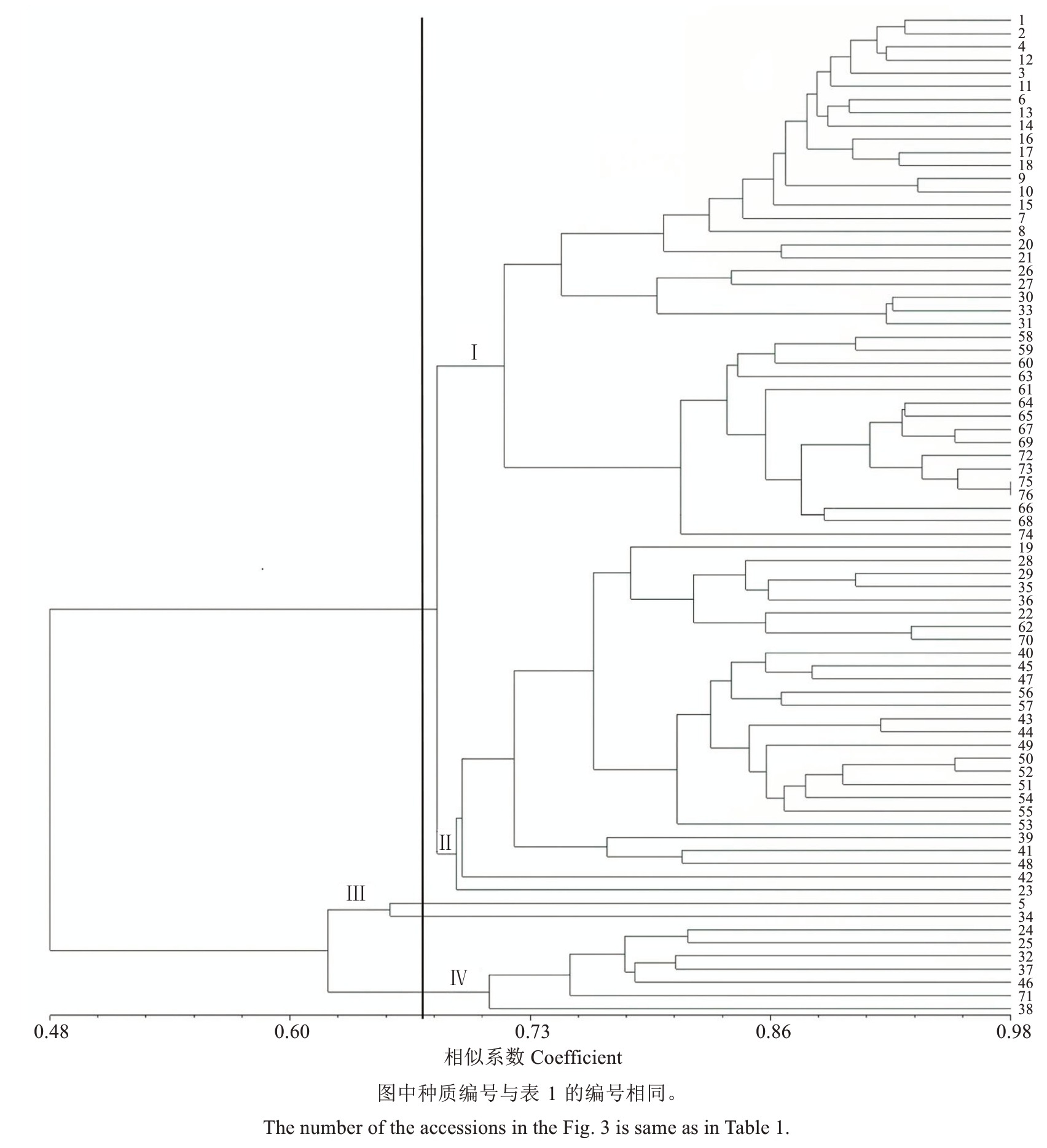
图3 76份种质的UPGMA遗传关系聚类图Fig. 3 UPGMA genetic relationship cluster of 76 accessions
ISSR聚类结果表明同一类群中有些种质的表型性状差异较大,如峄城抗寒砧木1号和峄城竹叶青亲缘关系较近,但性状差异较大;绣球牡丹(75号)和紫皮甜(76号)虽然都属观赏类型,遗传相似系数为0.985,遗传关系最近,但果皮颜色和瓣型都不同,表型性状差异较大。这些结果表明ISSR标记与形态分类结果不一致。在相似系数约为0.678时,第Ⅰ类和第Ⅱ类种质又聚为同一类,包含67份种质,占供试种质总数的88.16%,说明这些种质的遗传关系相对较近。
3 讨 论
关于石榴遗传分析的研究方法主要包括形态学[15]和分子标记[16]。目前,用于石榴遗传多样性、遗传关系研究的DNA标记主要包括RAPD、AFLP、SRAP、ISSR等[17],各种标记的引物多态性不尽相同。Youssef等[18]研究表明,SRAP标记在11个不同石榴品种中的引物多态性平均为74.92%;Faraj等[19]研究表明ISSR在24个石榴品种中呈现79%的引物多态性;Almiahy等[20]研究表明ISSR在不同石榴基因型中的引物多态性为80.6%;Moslemi等[5]研究表明AFLP在67个石榴品种中的多态性为54.13%。本研究中,16个ISSR引物在76份石榴种质中呈现的多态位点比例为88.2%,明显高于前人文献报道的SRAP[18]、AFLP[5]和RAPD[21]的多态性,与Faraj等[19]和Almiahy等[20]的研究结果接近。ISSR标记在其他果树遗传分析中同样展现出优异的多态性特征。对西北喜马拉雅地区50份杏种质资源的遗传变异及群体结构研究显示,ISSR标记检测到83.33%~87.50%的高多态性,精准解析了杏种群的遗传结构[22]。对罗马尼亚本土苹果栽培品种进行研究,ISSR标记多态性达82.3%,可有效用于苹果品种的分子鉴定[23]。本研究中,ISSR标记在石榴中检测到88.2%的高多态性,与杏(83.33%~87.50%)[22]、苹果(82.3%)[23]等的研究结果基本一致。ISSR标记在石榴及其他果树遗传分析中均表现出稳定的高多态性,是种质资源评价的有效工具,未来可结合功能基因组学方法挖掘其在重要性状关联分析中的应用价值。
遗传多样性是评价生物多样性的参考指标之一,物种的遗传多样性越丰富,对环境变化的适应能力就越强。本研究表明山东石榴种质的Ne为1.370 4,He为0.370 4,I为0.545 1,GS为0.49~0.98,与王庆军等[24]、赵丽华等[25]研究的结果相似。76份种质中有67份种质在GS约为0.678时聚为一大类,表明这些山东石榴种质资源的遗传关系较近。为了石榴种质的保存与遗传改良,需要引进外地品种,或者选用遗传关系较远的优良品种(如第Ⅲ和第V类)进行杂交等,以拓宽山东石榴种质的遗传背景、提高多样性。
形态性状分类是通过肉眼对可见性状的异同进行分类,是对基因变异的间接反映,而ISSR是基于DNA层面对基因变异的直接反映。沈进等[26]研究表明石榴品种DNA水平的聚类模式与其发源地和农艺性状无明显相关性。本研究表明ISSR聚类结果与形态学分类不一致,与沈进等[26]结论相一致。这可能是由于长期栽培的人为选育、自然杂交以及品种交换等造成石榴遗传背景比较复杂,形态学特征只是某一性状的外在表现。因此要准确解析种质资源的遗传变异,必须提升到DNA水平,这一观点在多种果树上得到验证。Bădulescu等[23]利用ISSR标记技术评估苹果栽培品种间的遗传变异,ISSR标记成功区分了形态和遗传背景极其相似的Remar和Iris品种,说明该技术能精准鉴定亲缘关系密切的个体基因型。在橄榄种质资源研究中,Raji等[27]证实25株果实性状突出的候选优质橄榄树间存在显著的个体差异,且该差异可通过ISSR标记有效鉴别,其中,CPT14不仅具有独特的形态学特征,其ISSR标记谱型也呈现明显区分度,表明ISSR标记能够精准筛选出形态相似群体中的优异种质,避免了单纯依赖形态可能存在的漏选风险。在杧果遗传多样性研究中,Rocha等[28]证实ISSR标记的遗传分类结果不受地理环境因素的干扰,有效排除了表型因环境不同所造成的形态性状变异,直接从DNA层面揭示了品种间的真实遗传关系。本研究进一步说明ISSR标记同样适用于石榴,能从DNA水平解析其种质的遗传变异和亲缘关系。
DNA指纹图谱构建的方法有多种,如单引物法[29]、引物组合法[30]、特征谱带法[31]等。张安世等[32]采用ISSR技术构建了猕猴桃的DNA指纹,但由于缺乏指纹编码,因此在实际生产应用中受限。王世强等[33]绘制了黄精的SSR指纹,但是复杂的指纹编码限制了实际应用。本研究比较单引物和引物组合对石榴种质的区分效率,结果表明所有单引物均不能将供试种质区分开,最高区分效率为85.53%,而引物组合的区分效率明显高于单引物,最高区分效率为97.37%,仅无法区分2份种质。这一结果与邵雪花等[11]的相似,表明通过优化引物组合可明显提高分子标记的鉴别能力。因此选用引物组合UBC834/UBC835构建了76份种质的DNA指纹图谱,除了75号(绣球牡丹)和76号(紫皮甜)的指纹相同外,其他74份种质的DNA指纹均不相同,具有高度特异性,可用于品种的真实性鉴定。本研究构建的这套ISSR-DNA指纹图谱与形态性状相比能更高效、准确地鉴定石榴种质,为中国石榴指纹数据库的建立提供了理论依据。
4 结 论
本研究证实了ISSR标记能有效解析石榴种质的遗传变异,揭示了山东石榴种质遗传多样性相对丰富,但仍需引进外来优异种质拓宽遗传基础。构建的74份种质DNA指纹图谱具有高度特异性,可用于种质的真实性鉴定。
[1] LYU Y,PORAT R,YERMIYAHU U,HELER Y,HOLLAND D,DAG A. Effects of nitrogen fertilization on pomegranate fruit,aril and juice quality[J] . Journal of the Science of Food and Agriculture,2020,100(4):1678-1686.
[2] BAGHEL R S,KEREN-KEISERMAN A,GINZBERG I. Metabolic changes in pomegranate fruit skin following cold storage promote chilling injury of the peel[J] . Scientific Reports,2021,11:9141.
[3] 罗华,毕润霞,赵登超,舒秀阁,李志强,任艳华,侯乐峰,郝兆祥. 我国石榴产业发展现状与对策[J] . 黑龙江农业科学,2025(6):92-98.LUO Hua,BI Runxia,ZHAO Dengchao,SHU Xiuge,LI Zhiqiang,REN Yanhua,HOU Lefeng,HAO Zhaoxiang. Development status and countermeasures of pomegranate industry in China[J] . Heilongjiang Agricultural Sciences,2025(6):92-98.
[4] 郝兆祥,程作华,侯乐峰,罗华,赵亚伟. 山东枣庄石榴产业发展现状、存在问题及对策[C] . 怀远:第二届中国石榴博览会暨第七届全国石榴生产与科研研讨会论文集,2017:28-34.HAO Zhaoxiang,CHENG Zuohua,HOU Lefeng,LUO Hua,ZHAO Yawei. Current situation,problems and countermeasures of pomegranate industry development in Zaozhuang,Shandong[C] //Huaiyuan:Proceedings of the 2nd China Pomegranate Expo and the 7th National Symposium on Pomegranate Production and Scientific Research,2017:28-34.
[5] MOSLEMI M,ZAHRAVI M,KHANIKI G B. Genetic diversity and population genetic structure of pomegranate (Punicɑ grɑnɑtum L.) in Iran using AFLP markers[J] . Scientia Horticulturae,2010,126(4):441-447.
[6] PATIL P G,SINGH N V,PARASHURAM S,BOHRA A,SOWJANYA R,GAIKWAD N,MUNDEWADIKAR D M,SANGNURE V R,JAMMA S M,INJAL A S,BABU K D,SHARMA J. Genome-wide characterization and development of simple sequence repeat markers for genetic studies in pomegranate (Punicɑ grɑnɑtum L.)[J] . Trees,2020,34(4):987-998.
[7] 洪文娟,郝兆祥,刘康佳,罗华,毕润霞,苑兆和,宗世祥,王君. 基于石榴全基因组序列的SSR标记开发及鉴定[J] . 北京林业大学学报,2019,41(8):38-47.HONG Wenjuan,HAO Zhaoxiang,LIU Kangjia,LUO Hua,BI Runxia,YUAN Zhaohe,ZONG Shixiang,WANG Jun. Development and identification of SSR molecular markers based on whole genomic sequences of Punicɑ grɑnɑtum[J] . Journal of Beijing Forestry University,2019,41(8):38-47.
[8] LU L J,YANG J,ZHANG S M,YU F F,SUN Q B,ZHOU J Y.Advances in molecular marker techniques in pomegranate (Punicɑ grɑnɑtum L.)[J] . Agricultural Science & Technology,2016,17(1):8-11.
[9] 彭磊,包九零,乔光,文晓鹏. 贵州大樱桃种质资源亲缘关系ISSR分析[J] . 种子,2020,39(4):1-4.PENG Lei,BAO Jiuling,QIAO Guang,WEN Xiaopeng. Genetic relationship analysis of sweet cherry germplasm in Guizhou based on ISSR markers[J] . Seed,2020,39(4):1-4.
[10] 刘小英,黄飞龙,刘国强,张游南. 枇杷ISSR分子标记的研究进展[J] . 中国农学通报,2021,37(21):106-110.LIU Xiaoying,HUANG Feilong,LIU Guoqiang,ZHANG Younan. ISSR molecular markers in loquat:research progress[J] .Chinese Agricultural Science Bulletin,2021,37(21):106-110.
[11] 邵雪花,赖多,肖维强,刘传和,贺涵,匡石滋. 番石榴遗传多样性的ISSR分析及指纹图谱构建[J] . 中国农学通报,2023,39(21):39-47.SHAO Xuehua,LAI Duo,XIAO Weiqiang,LIU Chuanhe,HE Han,KUANG Shizi. Psidium guɑjɑvɑ L.:Genetic diversity analysis based on ISSR markers and construction of fingerprints[J] .Chinese Agricultural Science Bulletin,2023,39(21):39-47.
[12] DEMIREL S,PEHLUVAN M,ASLANTAŞ R. Evaluation of genetic diversity and population structure of peach (Prunus persicɑ L.) genotypes using inter-simple sequence repeat (ISSR)markers[J] . Genetic Resources and Crop Evolution,2024,71(3):1301-1312.
[13] VAISHNAV K,TIWARI V,DURGAPAL A,MEENA B,RANA T S. Estimation of genetic diversity and population genetic structure in Gymnemɑ sylvestre (Retz.) R. Br. ex Schult. populations using DAMD and ISSR markers[J] . Journal of Genetic Engineering and Biotechnology,2023,21(1):42.
[14] 朱薇,吴志苹,李雪,王庆军,袁立新,马丽. 观赏石榴品种的ISSR遗传关系与 DNA指纹研究[J] . 北方园艺,2023(15):59-65.ZHU Wei,WU Zhiping,LI Xue,WANG Qingjun,YUAN Lixin,MA Li. Study on genetic relationship and DNA fingerprint of ornamental pomegranate cultivars based on ISSR markers[J] .Northern Horticulture,2023(15):59-65.
[15] MARTÍNEZ J J,MELGAREJO P,HERNÁNDEZ F,SALAZAR D M,MARTÍNEZ R. Seed characterisation of five new pomegranate (Punicɑ grɑnɑtum L.) varieties[J] . Scientia Horticulturae,2006,110(3):241-246.
[16] PATIL P G,SINGH N V,BOHRA A,RAGHAVENDRA K P,MANE R,MUNDEWADIKAR D M,BABU K D,SHARMA J.Comprehensive characterization and validation of chromosomespecific highly polymorphic SSR markers from pomegranate(Punicɑ grɑnɑtum L.) cv. Tunisia genome[J] . Frontiers in Plant Science,2021,12:645055.
[17] CHAUHAN R D,KANWAR K. Biotechnological advances in pomegranate (Punicɑ grɑnɑtum L.) [J] . In Vitro Cellular & Developmental Biology - Plant,2012,48(6):579-594.
[18] YOUSSEF M,ALHAMMADI A S,RAMÍREZ-PRADO J H,SÁNCHEZ-TEYER L F,ESCOBEDO-GRACIAMEDRANO R M. Remarks on genetic diversity and relationship of Punicɑ protopunicɑ and P. grɑnɑtum assessed by molecular analyses[J] . Genetic Resources and Crop Evolution,2018,65(2):577-590.
[19] FARAJ J M. Morpho-phytochemical screening and molecular diversity of pomegranate accessions grown in Halabja Governorate,Kurdistan Region-Iraq[J] . Genetic Resources and Crop Evolution,2023,70(8):2657-2670.
[20] ALMIAHY F H,JUM’A F F.Evaluation of the genetic diversity of pomegranate accessions some Iraqi Pomegranate (Punicɑ grɑnɑtum L.) genotypes using ISSR marker[J] . IOSR Journal of Agriculture and Veterinary Science (IOSR-JAVS),2017(10):44-49.
[21] 杨荣萍,龙雯虹,张宏,许彬,李文祥. 云南25份石榴资源的RAPD分析[J] . 果树学报,2007,24(2):226-229.YANG Rongping,LONG Wenhong,ZHANG Hong,XU Bin,LI Wenxiang. RAPD analysis of 25 Punicɑ grɑnɑtum germplasm resources collected in Yunnan Province[J] . Journal of Fruit Science,2007,24(2):226-229.
[22] SHEIKH Z N,SHARMA V,AHMAD SHAH R,SHARMA N,SUMMUNA B,AL-MISNED F A,EL-SEREHY H A,MIR J I.Genetic diversity analysis and population structure in apricot(Prunus ɑrmeniɑcɑ L.) grown under north-western himalayas using ISSR markers[J] . Saudi Journal of Biological Sciences,2021,28(10):5986-5992.
[23] BĂDULESCU L,UDRIȘTE A A,BUTCARU A C,IORDĂCHESCU M. Genetic diversity analysis among several apple genotypes using ISSR markers[J] . Fruit Growing Research,2022,38:40-44.
[24] 王庆军,马丽,郝兆祥,罗华,毕润霞,赵丽娜,侯乐峰. 观赏石榴种质资源遗传多样性的ISSR分析[J] . 浙江农业学报,2018,30(2):236-241.WANG Qingjun,MA Li,HAO Zhaoxiang,LUO Hua,BI Runxia,ZHAO Lina,HOU Lefeng. Analysis on germplasm genetic diversity of ornamental pomegranate (Punicɑ grɑnɑtum L.) by ISSR markers[J] . Acta Agriculturae Zhejiangensis,2018,30(2):236-241.
[25] 赵丽华,李名扬,王先磊,刘华敏. 石榴种质资源遗传多样性及亲缘关系的ISSR分析[J] . 果树学报,2011,28(1):66-71.ZHAO Lihua,LI Mingyang,WANG Xianlei,LIU Huamin. Genetic diversity and genetic relationship of pomegranate (Punicɑ grɑnɑtum) germplasm evaluated by ISSR markers[J] . Journal of Fruit Science,2011,28(1):66-71.
[26] 沈进. 中国石榴资源初级核心种质构建及其ISSR分子标记研究[D] . 合肥:安徽农业大学,2008.SHEN Jin. Study on construction of primary core collection in pomegranate germplasm resources in China and its ISSR marker[D] . Hefei:Anhui Agricultural University,2008.
[27] RAJI R,SIRIL E A. Genetic diversity analysis of promising Ceylon olive (Elɑeocɑrpus serrɑtus L.) genotypes using morphological traits and ISSR markers[J] . Current Plant Biology,2021,26:100201.
[28] ROCHA A,SALOMÃO L C C,SALOMÃO T M F,CRUZ C D,DE SIQUEIRA D L. Genetic diversity of ‘Ubá’ mango tree using ISSR markers[J] . Molecular Biotechnology,2012,50(2):108-113.
[29] 唐浩,余汉勇,张新明,魏兴华. 水稻新品种测试的标准品种DNA指纹图谱多样性分析[J] . 植物遗传资源学报,2015,16(1):100-106.TANG Hao,YU Hanyong,ZHANG Xinming,WEI Xinghua.Analysis on the diversity of DNA fingerprinting of the example varieties used for the test of rice new varieties[J] . Journal of Plant Genetic Resources,2015,16(1):100-106.
[30] 缪恒彬,陈发棣,赵宏波,房伟民,石丽敏. 应用ISSR对25个小菊品种进行遗传多样性分析及指纹图谱构建[J] . 中国农业科学,2008,41(11):3735-3740.MIAO Hengbin,CHEN Fadi,ZHAO Hongbo,FANG Weimin,SHI Limin. Genetic diversity and construction of fingerprinting of Chrysɑnthemum cultivars by ISSR markers[J] . Scientia Agricultura Sinica,2008,41(11):3735-3740.
[31] 高源,刘凤之,王昆,王大江,龚欣,刘立军. 基于TP-M13-SSR指纹图谱的中国原产苹果属植物分子身份证的建立[J] . 植物遗传资源学报,2015,16(6):1290-1297.GAO Yuan,LIU Fengzhi,WANG Kun,WANG Dajiang,GONG Xin,LIU Lijun. Establishment of molecular identity card for Mɑlus Mill. originated from China based on the fingerprints of TPM13-SSR[J] . Journal of Plant Genetic Resources,2015,16(6):1290-1297.
[32] 张安世,韩臣鹏,齐秀娟,张中海. 基于ISSR标记的猕猴桃品种遗传多样性分析及指纹图谱构建[J] . 植物资源与环境学报,2017,26(3):19-26.ZHANG Anshi,HAN Chenpeng,QI Xiujuan,ZHANG Zhonghai. Genetic diversity analysis and fingerprinting construction of cultivars of Actinidiɑ spp. based on ISSR marker[J] . Journal of Plant Resources and Environment,2017,26(3):19-26.
[33] 王世强,王立儒,刘帅,牛俊峰,冯书超,梁晓艳,强毅,王喆之.基于SSR标记的黄精品种(系)DNA指纹图谱库构建[J] . 分子植物育种,2018,16(6):1878-1887.WANG Shiqiang,WANG Liru,LIU Shuai,NIU Junfeng,FENG Shuchao,LIANG Xiaoyan,QIANG Yi,WANG Zhezhi. Construction of DNA fingerprint database based on SSR marker for Polygonɑtum varieties (lines)[J] . Molecular Plant Breeding,2018,16(6):1878-1887.