核桃是胡桃科核桃属(Juglans)多年生落叶果树,又称胡桃、羌桃、万岁子等[1]。核桃具有丰富的营养价值,为世界四大坚果之一,在全世界范围内分布广泛[2]。核桃种仁主要含有脂肪、蛋白质、淀粉、糖、维生素、氨基酸、黄酮、多酚、单宁和矿质元素等。核桃种仁表面的薄膜称为种皮,一般呈淡黄色,其中含有大量的单宁等化学物质[3],产生苦涩味。种皮氧化后呈黑褐色或褐色,影响感官品质,因此在食品加工过程中常被去除[4]。但是,由于核桃种皮还富含多种有助于人类身体健康的氨基酸等物质,直接去除核桃种皮,不但会丢失许多营养成分,还会造成资源浪费。
现有核桃种皮相关研究主要聚焦于对核桃酚类物质的测定,涉及单宁、酚酸类、黄酮类等关键成分的定性定量研究。俞文君等[5]发现核桃苦涩味指标中单宁、绿原酸、没食子酸、儿茶素的多样性指数较大,反映了这些性状的遗传多样性及丰富程度。周高兴等[6]研究发现,核桃果实种皮内单宁等酚类物质含量最高。刘雨霞等[7]研究了薄壳香等5 个核桃品种的种皮中总单宁、酚酸类组分、黄酮类组分和生物碱组分等物质含量,发现总单宁、绿原酸、表儿茶素等是影响核桃种皮苦涩味的主要物质。随着研究的不断深入,关于核桃种皮的研究逐渐呈现出从成分解析向分子机制探索转变的趋势[8]。盛芳[9]、Zhao等[10]通过转录组等组学技术,已初步揭示种皮发育过程中苯丙氨酸代谢通路的动态调控规律,鉴定出部分调控多酚合成的关键候选基因,为解析种皮中多酚积累的分子机制提供了新依据。因此,十分有必要对核桃种皮中单宁积累部位及其与多酚类物质相关性进行研究。探究核桃种皮中单宁等酚类物质的生物合成与积累规律,对核桃生产和再加工具有重要意义。笔者主要对不同核桃品种内种皮的细胞形态、单宁等酚类物质进行研究,以期进一步明晰核桃种皮中单宁等酚类物质的生物合成与积累规律,为核桃品种改良提供重要的理论依据。
1 材料和方法
1.1 试验材料
新疆实生优系低单宁含量品种兵塔1 号(Juglans regia ‘Bingta No. 1’,简称BT-1)和中、高单宁含量新疆主栽品种温185(J. regia ‘Wen 185’,简称W-185)和扎343(J. regia ‘Zha 343’,简称Z-343)采于新疆生产建设兵团第一师三团核桃种质资源圃(N 40°23′,E 80°03′)。选取树龄均为5 a(年)及以上,生长健壮、水肥管理水平一致的树体。于核桃果实膨大期开始采样,随果实生长逐渐延长采样周期。分别在6月12日、6月18日、6月28日、7月5日、7月15日、7 月25 日、8 月9 日和8 月24 日采集核桃果实,命名为T1、T2、T3、T4、T5、T6、T7 和T8。采样时从每个植株的东、西、南、北4个方向各采集30个果实,混合均匀。将新鲜核桃种皮用镊子从种仁上剥离,一部分种皮置于FAA 溶液中进行固定,用于核桃种皮石蜡切片的制作;另一部分经冷冻干燥后研磨成粉末,于-80 ℃干燥保存,用于后续各项指标的测定。
1.2 核桃种皮形态观察
参照冶倩[11]的方法,对样品进行脱水、透蜡、包埋并制备切片,采用番红-固绿双重染色法进行染色,使用显微镜观察并摄影。
1.3 总单宁含量的测定
总单宁含量的测定参照宋岩等[12]的方法,标准曲线回归方程为y=0.079 9x+0.000 3,R2=0.999 3;单宁含量的染色参照关长飞[13]的方法,将新鲜核桃果实进行横切,将核桃果实横切面均匀地压在制备好的滤纸上染色,拍照记录,对图像进行对比分析。
1.4 总酚含量的测定
总酚含量的测定参照周高兴[14]的方法,标准曲线回归方程为:y=0.088 6x-0.085,R2=0.999 6。
1.5 总黄酮含量的测定
总黄酮含量的测定参照周晔[15]的方法,标准曲线回归方程为:y=0.018 4x-0.018 3,R2=0.999 7。
1.6 酚类物质成分及含量测定
参照刘雨霞[16]的方法,采用液相色谱法进行测定。色谱条件:Agilent 5 TC-C18(2)色谱柱(250 mm×4.6 mm);流速0.8 mL·min-1;柱温35 ℃;进样量10 μL;压力值25.0 MPa,流动相A 为甲醇溶液(50%,φ),B为乙酸溶液(0.2%,φ)。洗脱梯度为:0~4 min:92%A~92%A;4~10 min:92%A~79%A;10~18 min:79%A~79%A;18~23 min:79%A~79%A;23~28 min:79%A~71%A;28~32 min:71%A~60%A;32~35 min:60%A~8%A;35~40 min:8%A~92%A。
2 结果与分析
2.1 果实发育期不同品种核桃种皮单宁含量的动态变化
核桃果实采摘后沿横截面进行切割,并对核桃果实横切面进行印迹染色。单宁经印迹法染色处理后在试纸上呈紫色,据此可观察核桃果实在不同发育期单宁物质的沉积变化。染色结果如图1,试纸上出现核桃种仁轮廓形状的紫色印迹,表明核桃果实中的单宁物质主要集中在核桃种皮。随着核桃果实的生长发育,3个品种的果实单宁染色深度整体呈现先变浅后加深再变浅的动态变化趋势。兵塔1 号单宁染色深度在T1 到T3时期逐渐变浅,T4至T6时期逐步加深,T6时期之后再度变浅;温185单宁染色深度在T1到T3时期变浅,T3~T4时期迅速加深,T4时期之后整体变浅;扎343单宁染色深度则在T1到T2时期逐渐变浅,T3、T4时期迅速加深,T4时期之后整体变浅。
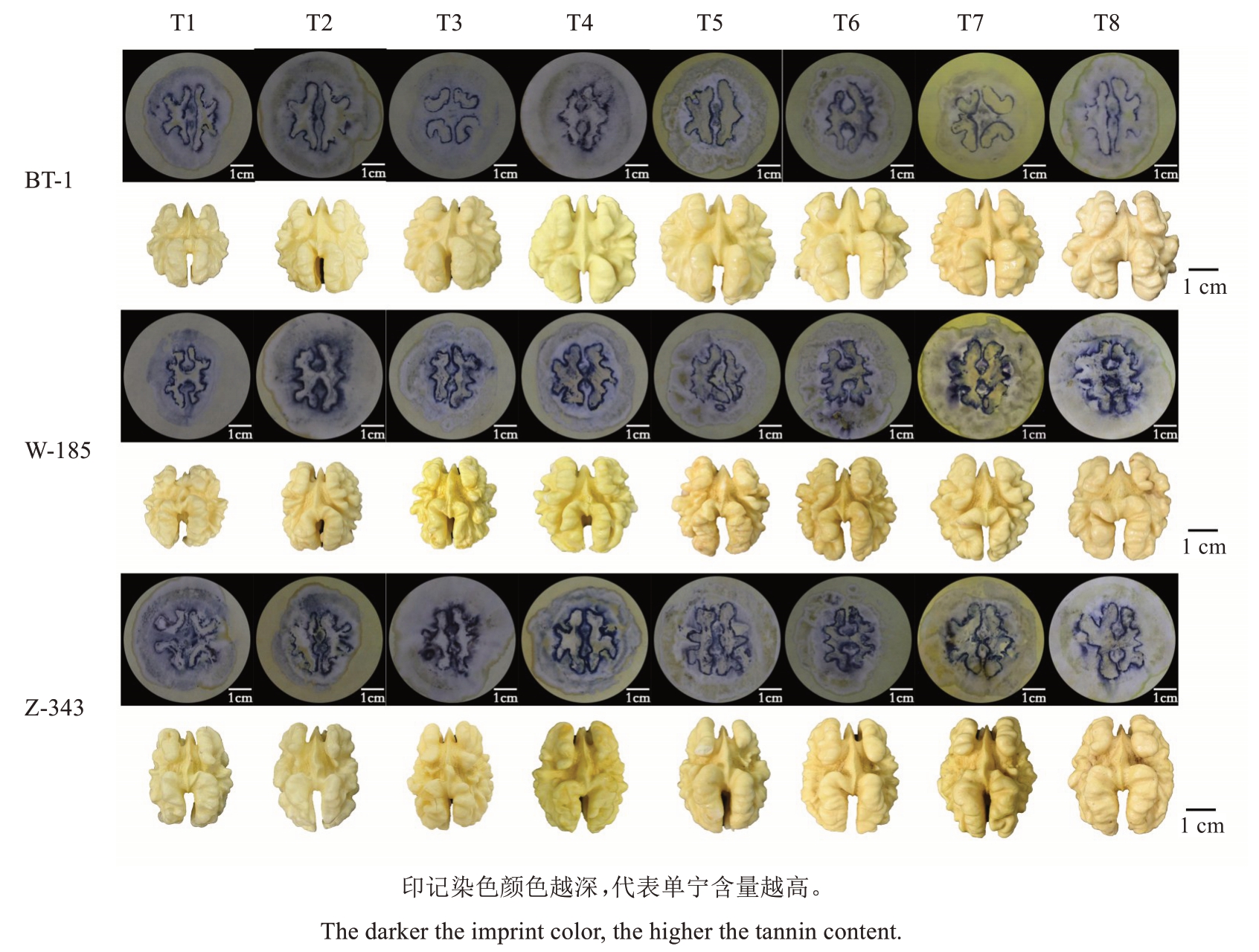
图1 3 个核桃品种8 个时期果实印迹染色
Fig. 1 The fruit cross section imprints of three walnut varieties at eight stages
动态测定核桃果实发育期内种皮的总单宁含量(图2),发现3 个核桃品种种皮的总单宁含量整体呈现出先下降后上升再下降的趋势。兵塔1 号和温185 在T2~T4 时期、扎343 在T1~T3 时期的单宁含量变化比较大,并且不同核桃品种的单宁积累也存在较大差异,兵塔1 号单宁含量最高时仅为352.75 mg·g-1,温185和扎343单宁含量最高时分别为386.84 mg·g-1、448.99 mg·g-1。其中,兵塔1 号单宁含量在整个发育期低于温185 和扎343,与图1 的印迹染色结果一致。
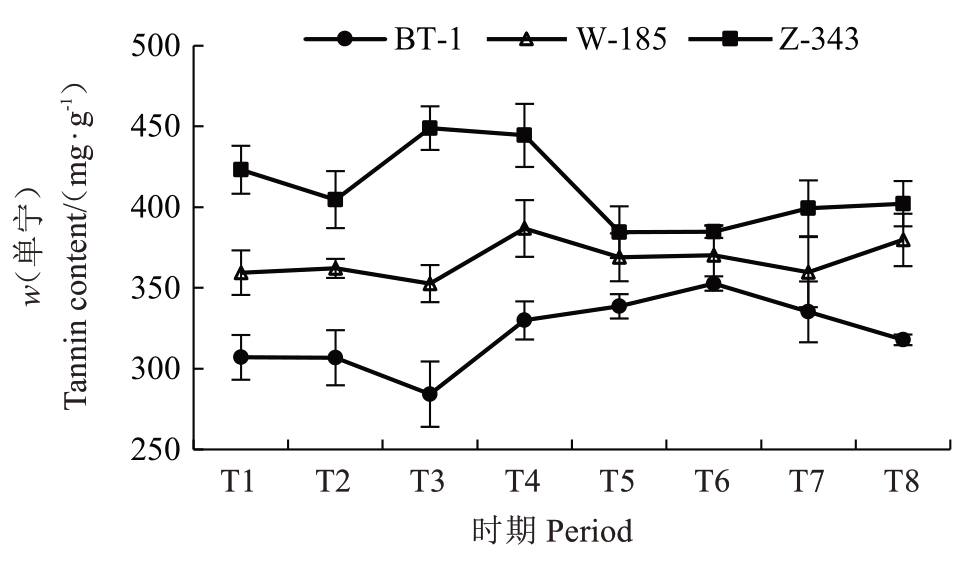
图2 不同生长发育时期核桃种皮总单宁含量的变化
Fig. 2 Changes in total tannin content in walnut kernel pellicle at different development stages
2.2 核桃种皮形态观察
结合各品种核桃种皮单宁含量变化的关键时期,采用石蜡切片和番红-固绿对染技术对核桃种皮细胞进行解剖结构观察,单宁物质经番红-固绿对染后可呈紫红色[17]。种皮石蜡切片(图3、图4)观察表明,种皮最外层由1~5 层大型的厚壁表皮细胞组成,表皮细胞无叶绿体,无气孔,通常为矩形、椭圆形或不规则形,细胞的垂周壁呈直线状,排列紧密;种皮薄壁细胞在种皮表皮细胞下方,细胞体型较小,呈不规则状,排列紧密;在种皮中间有不规则分布的维管束结构,在维管束周围零散分布着一些不规则的单宁细胞,即合成单宁或贮存、转运单宁的细胞;种皮的下表皮细胞即分隔种仁和种皮的细胞,通常为一层排列紧密的厚壁小型细胞,无叶绿体,无气孔,通常为矩形,细胞的垂周壁呈直线状。将单宁积累峰值时期的切片图放大(图4),发现维管束周围的单宁细胞越发达、越活跃、数量越多,种皮表皮细胞中单宁物质积累越快,整体单宁含量越高,并且在单宁含量达到峰值后,维管束周围的单宁细胞数量呈现减少趋势。综合番红-固绿对染情况发现,兵塔1 号种皮细胞中单宁含量低的原因是维管束周围用来合成、贮存和转运单宁物质的单宁细胞数量少,进而导致核桃种皮单宁合成速率低。
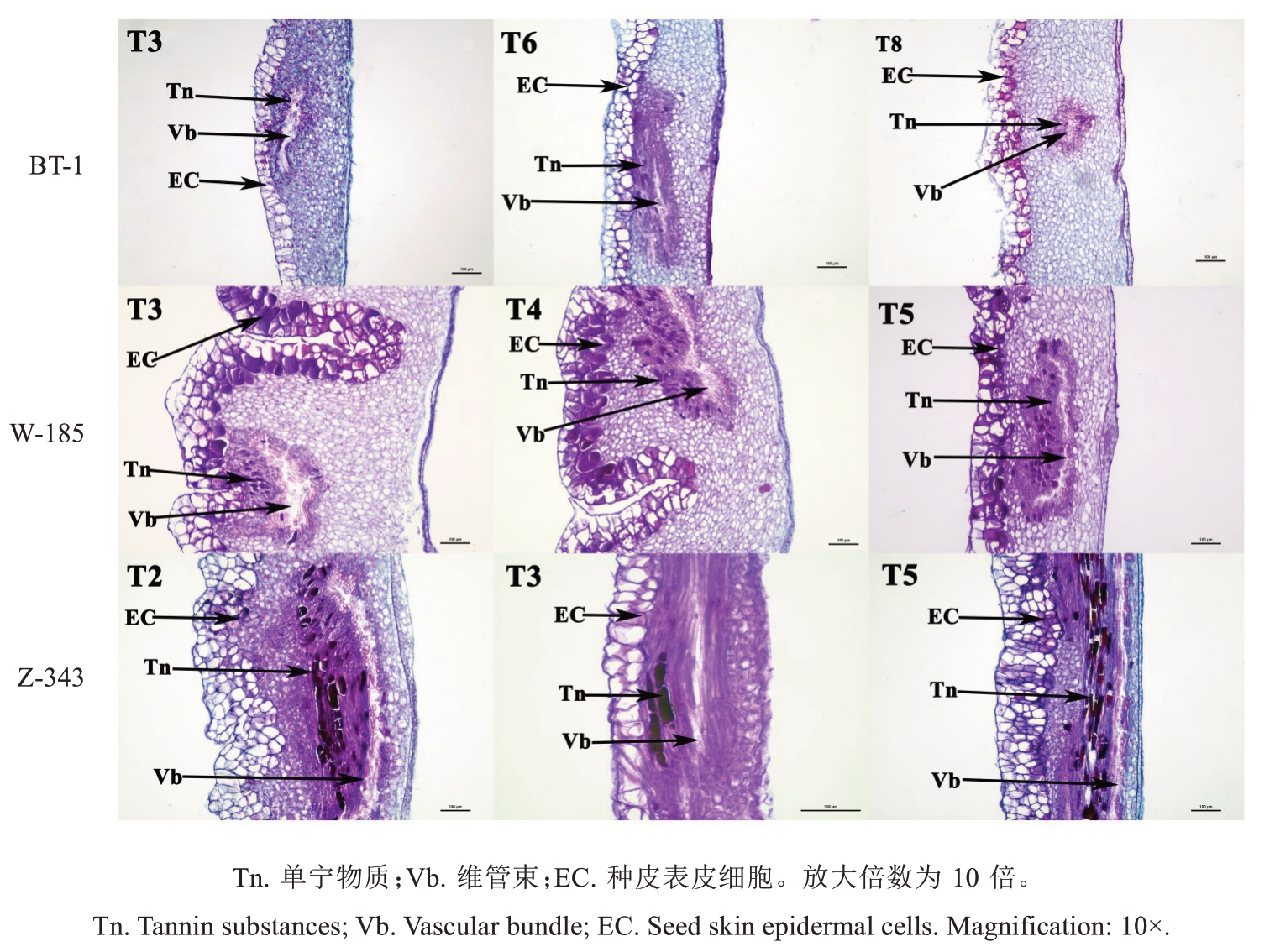
图3 果实不同发育阶段种皮细胞的变化
Fig. 3 Seed skin cell changes at different developmental stages of the fruit
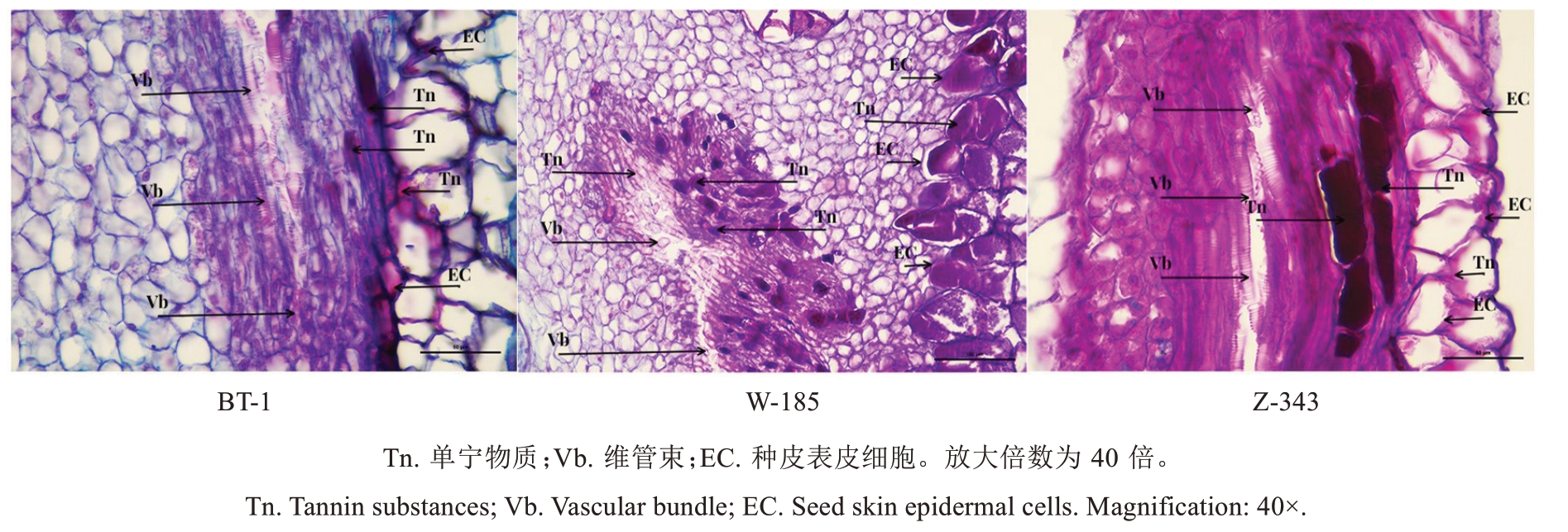
图4 不同品种间核桃种皮单宁含量最高时细胞对比
Fig. 4 Comparison of tannin abundant cells in walnut kernel pellicle among different varieties
2.3 果实发育期不同品种核桃种皮总酚、总黄酮含量的动态变化
对果实发育期内核桃种皮的总酚、总黄酮含量进行测定分析(图5~图6)。兵塔1 号在整个发育期内,其总酚、总黄酮含量低于温185和扎343。兵塔1号总酚含量在成熟期呈下降趋势,在T8 时期含量(w,后同)最低,为339.66 mg·g-1;温185 总酚含量在发育期内呈先上升后下降趋势,在T8 时期含量最低,为362.82 mg·g-1;扎343总酚含量在T4时期达到峰值,在T5时期快速下降至最低值(389.39 mg·g-1)。兵塔1 号黄酮含量在T3 时期最高,温185 在T7 时期最高,扎343 在T4 时期最高,分别为48.81 mg·g-1、64.40 mg·g-1和61.73 mg·g-1。结合单宁与总酚、总黄酮含量的差异结果,发现低单宁含量的兵塔1 号中,黄酮与总酚含量较低。因此,各酚酸与黄酮类物质可能是品种间单宁含量差异的原因,应对其组分进一步分析。
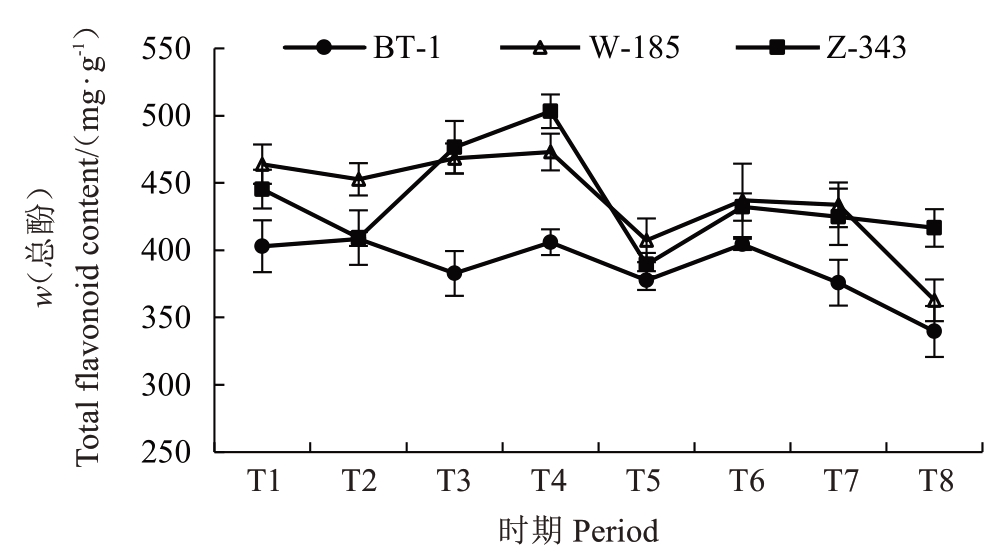
图5 不同生长发育时期核桃种皮总酚含量的变化
Fig. 5 Changes in total phenolic content of walnut kernel pellicle during fruit development
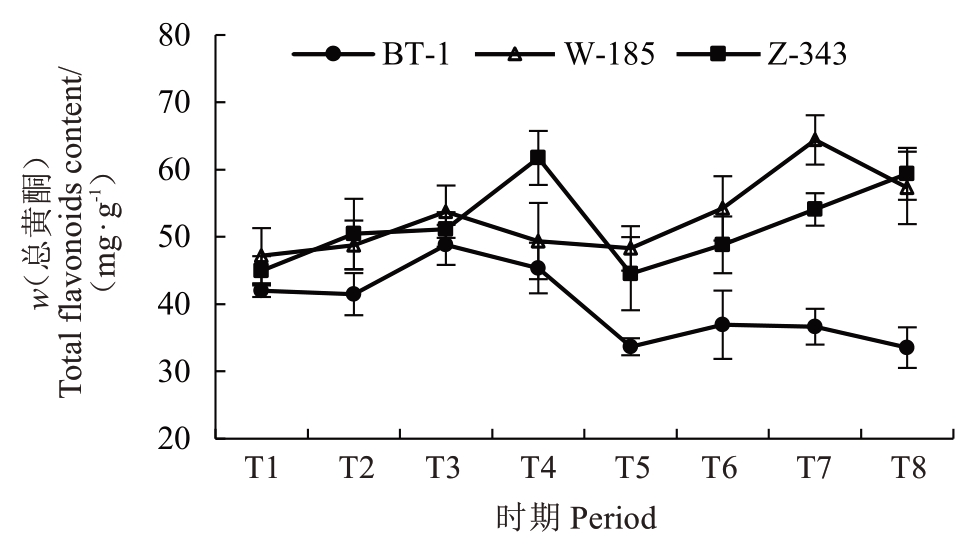
图6 不同生长发育时期核桃种皮总黄酮含量的变化
Fig. 6 Changes in total flavonoids content of walnut kernel pellicle during fruit development
2.4 不同品种核桃种皮中部分酚酸、黄酮组分含量的动态变化
根据核桃种皮单宁、总酚和黄酮的分析结果,结合当前已知的单宁合成路径(水解单宁、缩合单宁以及木质素分化3 个模块,图7),进一步对3 个核桃品种种皮中的6种酚酸、4种黄酮组分含量进行动态变化分析,结果如图8~图9所示。
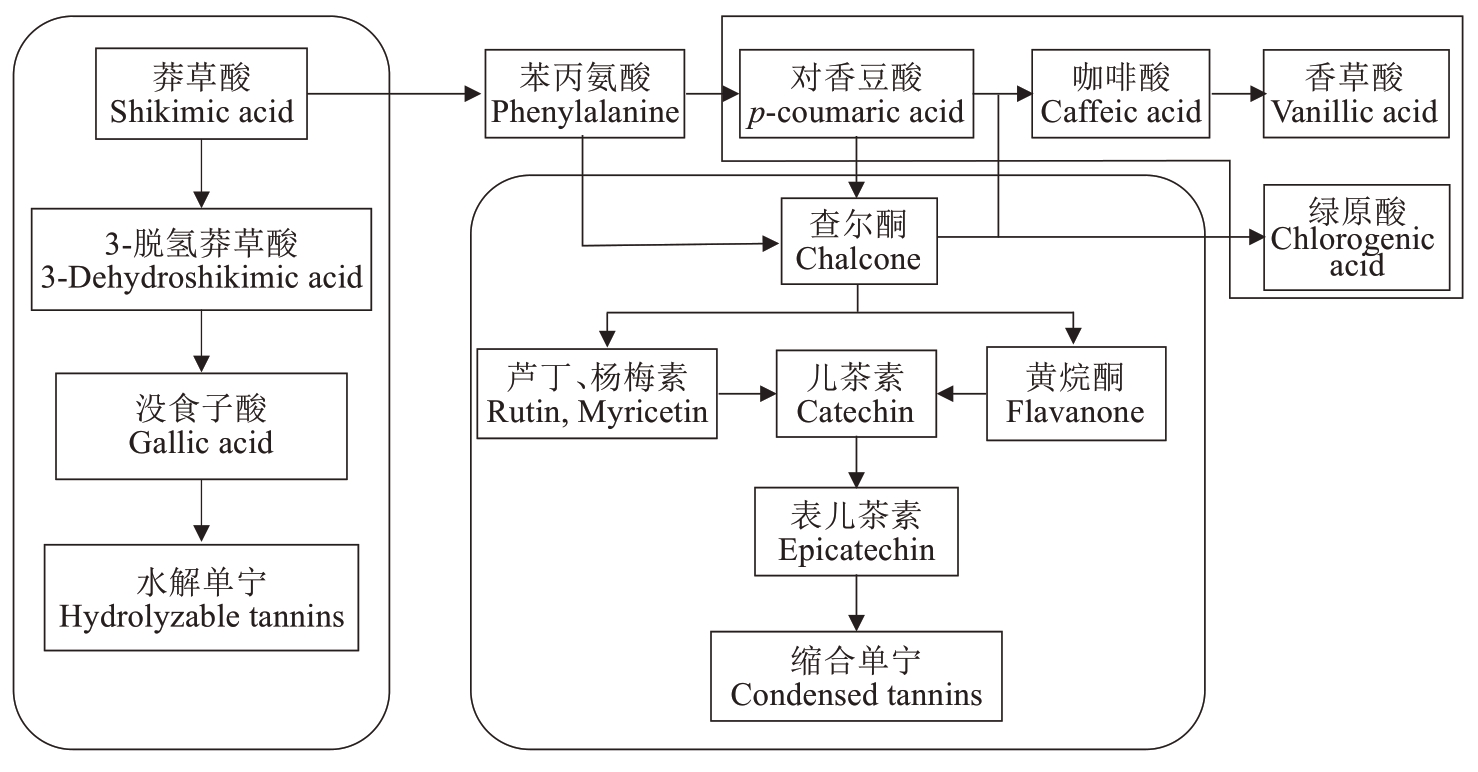
图7 单宁合成途径
Fig. 7 Tannin synthesis pathway

图8 不同生长发育时期核桃内种皮酚酸类物质含量的变化
Fig.8 Changes in phenolic acids mntent in walnut kernel pellicle during fruit development
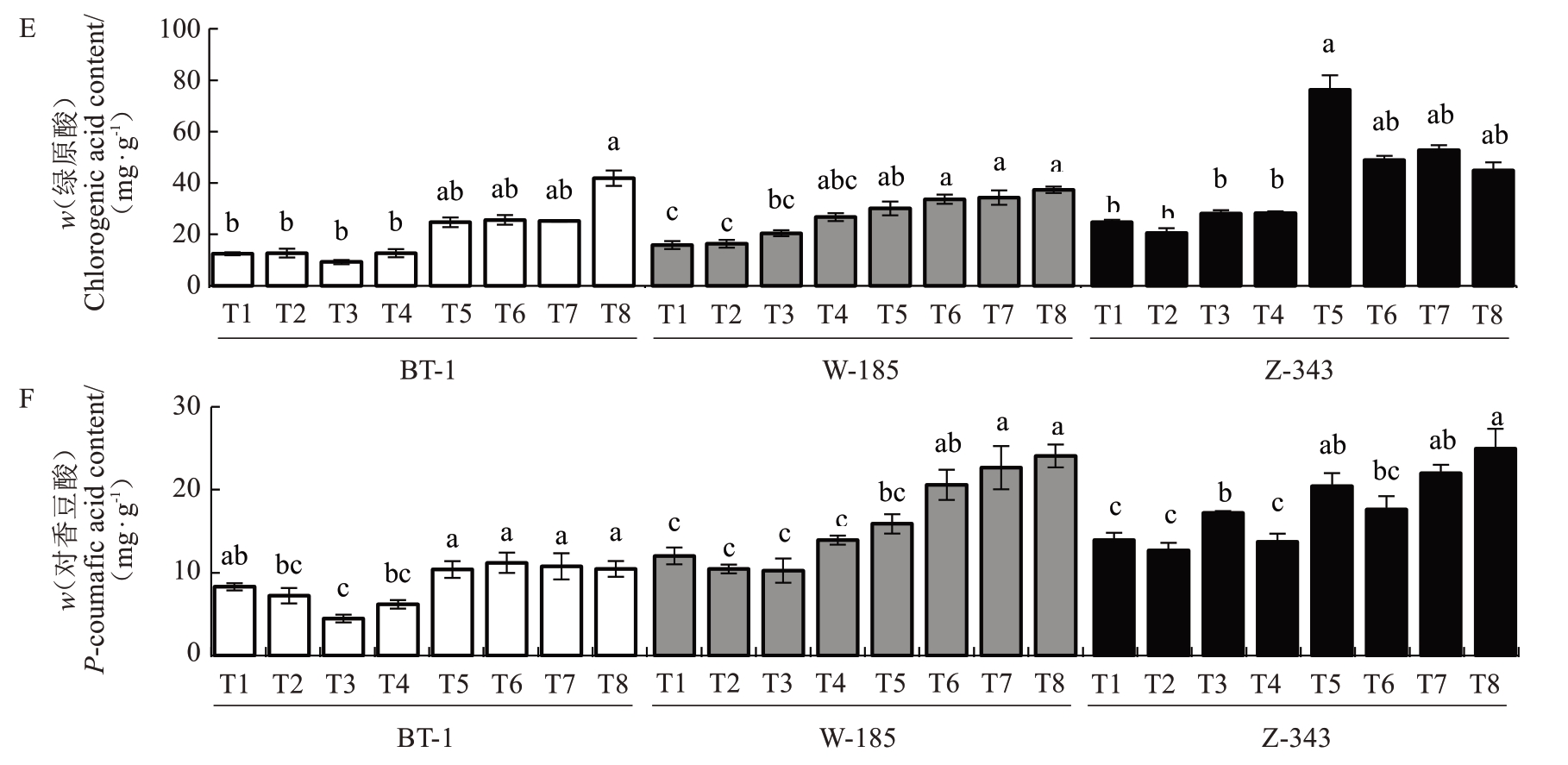
图8 (续)Fig. 8 (Continued)
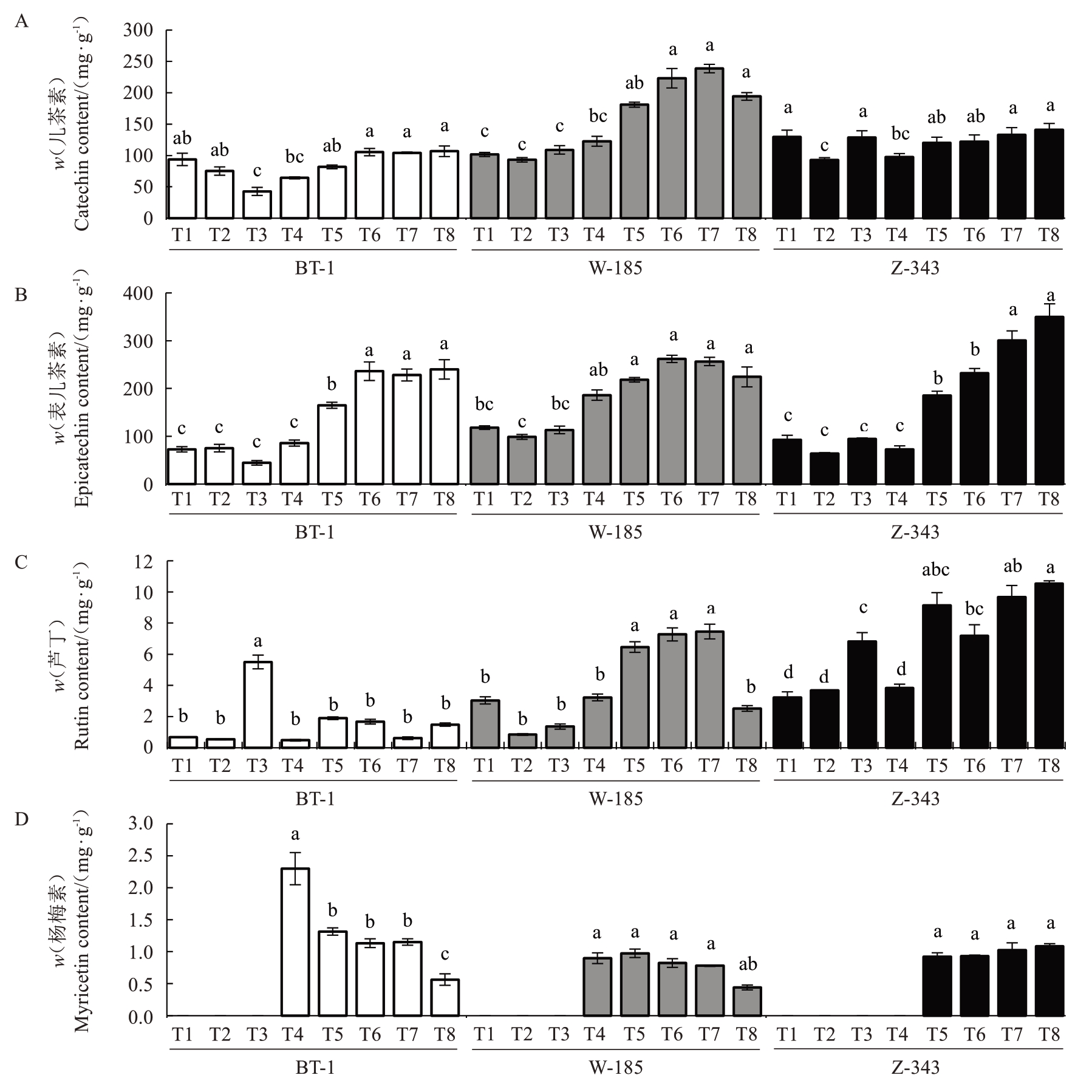
图9 不同生长发育时期核桃内种皮黄酮类物质含量的变化
Fig. 9 Changes in flavonoids content in walnut kernel pellicle during fruit development
酚酸组分分析结果(图8)表明,莽草酸是含量最高的酚酸类物质,没食子酸含量最低,各组分含量在品种间具有显著差异。兵塔1号莽草酸、香草酸和绿原酸含量表现出持续积累特征,而咖啡酸则呈持续下降趋势;温185 与扎343 的总酚酸含量高于兵塔1号,扎343的绿原酸含量在T5时期出现异常激增,含量由28.32 mg·g-1增至76.29 mg·g-1;而温185的香草酸含量在T5 时期发生骤降,含量由16.94 mg·g-1降至2.72 mg·g-1。值得注意的是,没食子酸与对香豆酸含量在3个品种中呈现相似的变化规律,但兵塔1号的含量整体偏低,平均含量分别为0.56 mg·g-1、8.61 mg·g-1,而温185 与扎343 分别为0.73 mg·g-1、16.23 mg·g-1和1.05 mg·g-1、17.82 mg·g-1。结果表明,6 种酚酸的动态变化与单宁含量显著相关。绿原酸的短时期迅速积累和没食子酸、对香豆酸等酚类前体物质的高水平积累可能会促使单宁含量增高。
核桃种皮黄酮组分含量(图9)表明,黄酮组分中含量最高的是表儿茶素,含量最低的是杨梅素。兵塔1 号核桃种皮的表儿茶素、儿茶素和芦丁含量显著低于其他品种,其中儿茶素、表儿茶素的积累过程均呈现先下降后上升趋势,含量最高值分别为106.70 mg·g-1、240.01 mg·g-1;兵塔1号的芦丁含量在T3时期急剧上升,然后又急剧下降,含量由0.54 mg·g-1上升到5.51 mg·g-1再下降到0.48 mg·g-1;温185的芦丁含量先下降再上升不断积累到T7 时期后又出现下降情况,含量最高值为7.46 mg·g-1;扎343 的芦丁含量整体呈不断上升趋势,在T3~T6 时期出现先升高再降低再升高的趋势,含量最高值为10.54 mg·g-1;杨梅素在核桃种皮发育前期积累较少,主要在后期积累,3 个核桃品种杨梅素含量最高值分别为2.30 mg·g-1、0.98 mg·g-1和1.09 mg·g-1。儿茶素与表儿茶素在单宁积累达峰值前的同一性变化和后期的差异性积累、芦丁的品种间差异分化和杨梅素的积累滞后现象,表明了在核桃种皮中充足的前体物质是决定单宁含量的重要因素。
2.5 不同品种核桃种皮各组分间相关性分析
为了探究单宁与各种多酚物质之间的相关性,利用Pearson 相关系数对其进行相关性分析(图10)。不同品种间单宁与各酚类物质含量的相关性趋势存在显著差异:在兵塔1号中,单宁积累与没食子酸等多数酚类物质含量呈正相关,其中与对香豆酸、表儿茶素含量的相关性最为显著,而与芦丁、莽草酸、咖啡酸含量呈负相关;温185的单宁含量与没食子酸含量呈极显著正相关,但与咖啡酸、总酚及总黄酮含量呈负相关;扎343的单宁含量仅与总酚、黄酮和咖啡酸含量呈正相关,其中与总酚含量的相关性达到极显著水平,而与杨梅素等多数酚酸组分含量呈负相关。各酚类物质之间的相关性差异特征表明,不同品种间核桃种皮多酚物质的协同积累模式存在明显的分化现象,其单宁的合成调控机制也存在品种差异性。
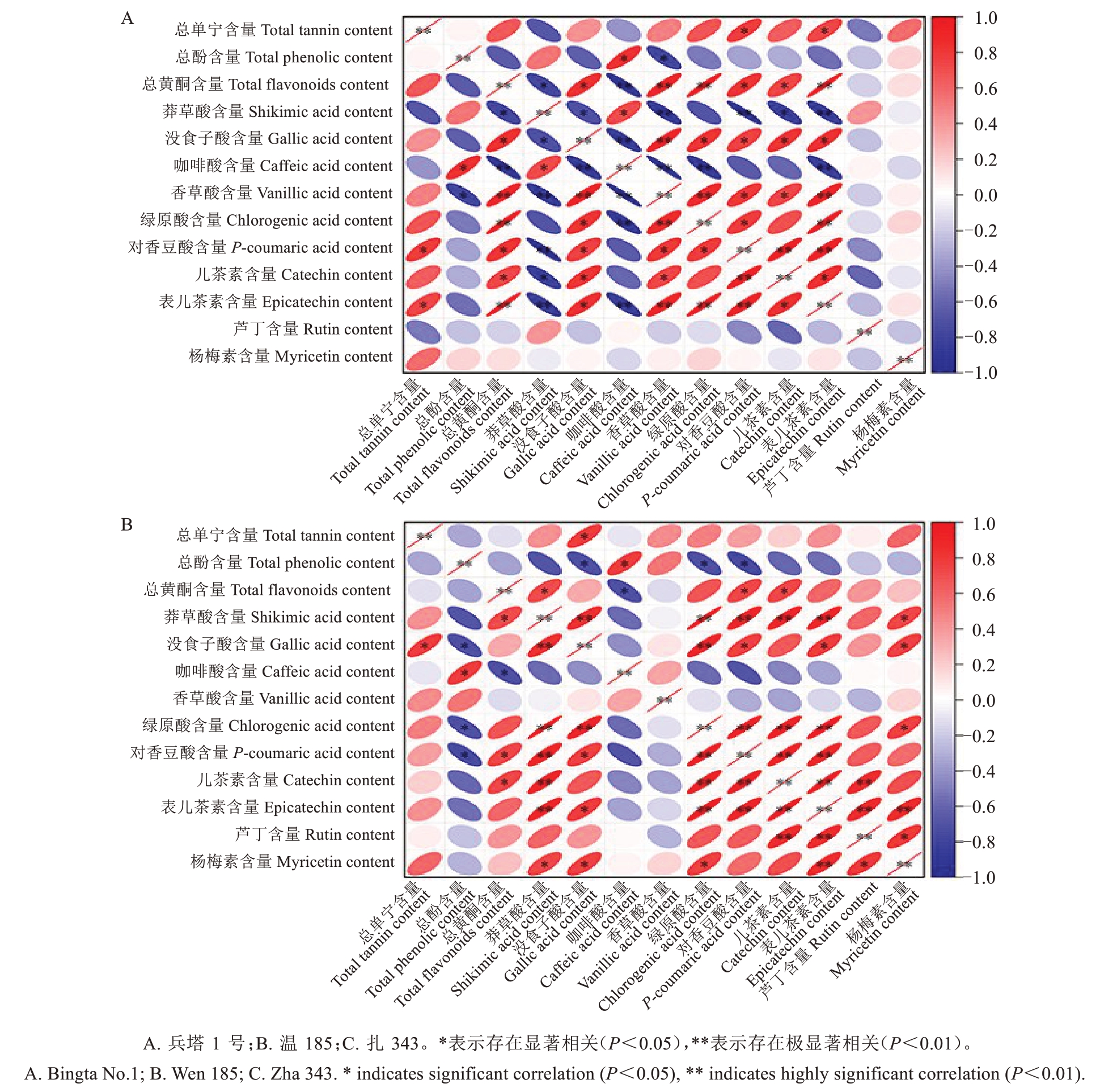
图10 核桃种皮单宁与各组分含量相关性分析
Fig. 10 Correlations between tannin and various components content in walnut kernel pellicle
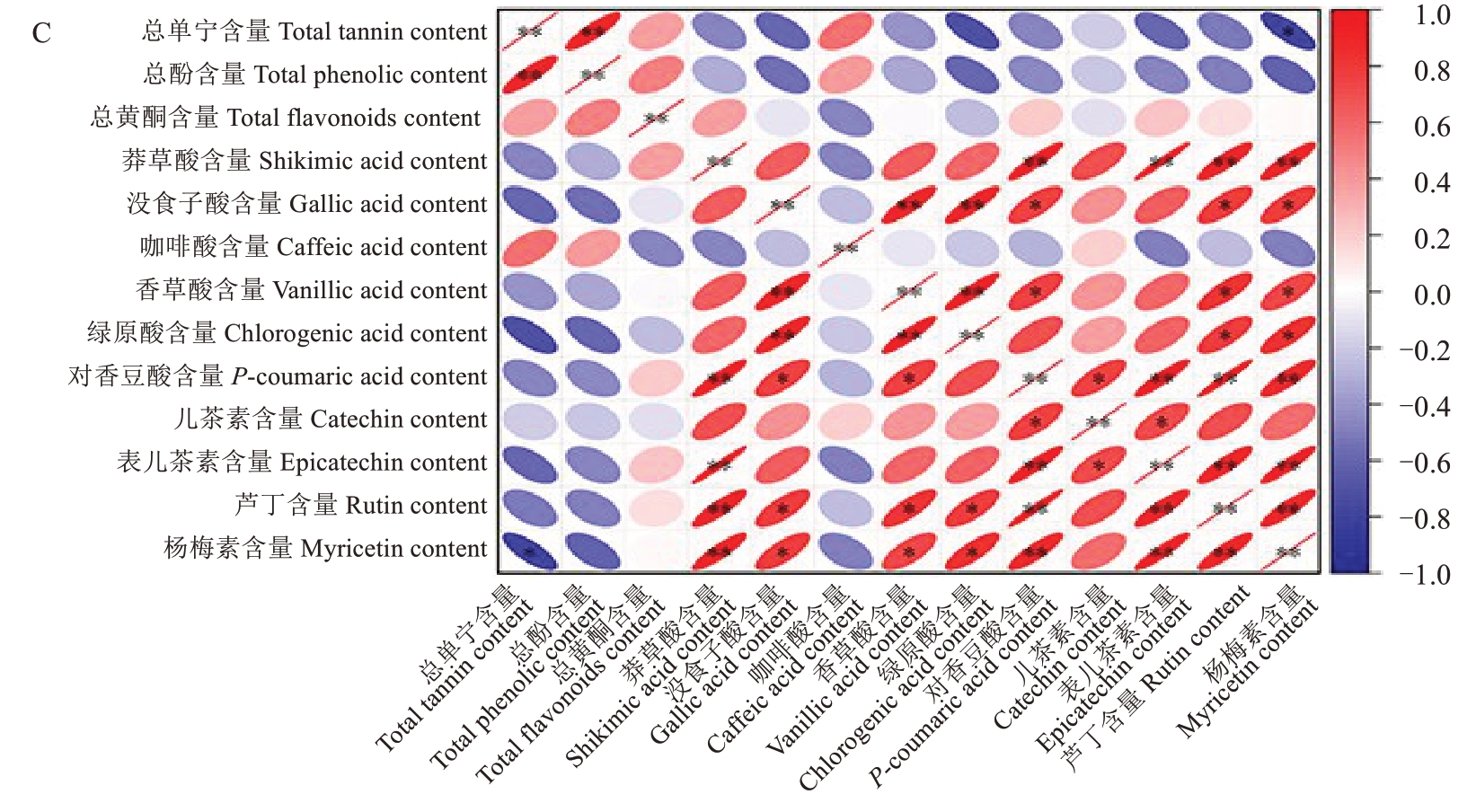
图10 (续) Fig. 10 (Continued)
3 讨 论
研究表明,酚类物质是核桃种皮的主要代谢物质之一[18],单宁的代谢动态与其涩味品质及抗氧化特性密切相关,而这一关联在核桃种仁中具体体现为:单宁类物质的含量直接影响其涩味表现与抗氧化能力[19]。Jin 等[20]研究表明,核桃种皮中的单宁含量随生育进程而逐渐上升,在成熟期达到峰值后,单宁积累停止并出现一定程度的下降。不同核桃品种种皮的单宁含量在发育进程中呈现显著差异:兵塔1 号的单宁积累量低于温185 和扎343,三者均表现出随生育期延长而积累至峰值后下降的规律。石蜡切片染色分析表明,3 个核桃品种的单宁转运规律具有一致性,其转运过程均呈现出从维管束系统经不规则单宁细胞,再向种皮表皮细胞渐进式堆积的特征。但是,单宁细胞的形态差异会影响单宁物质的合成与转运,初步从形态学角度揭示了单宁在种皮中的时空分布特征。
笔者发现不同核桃品种种皮中多酚类物质的积累模式存在显著差异,核桃果实在发育过程中,种皮内部多酚物质积累的合成途径相同,均为苯丙烷代谢途径[21-23]。苯丙烷代谢途径作为植物次生代谢的核心途径[24-26],其分支代谢流的分配直接影响酚酸、黄酮及单宁的合成。在莽草酸途径中,莽草酸作为苯丙氨酸的前体物质,其持续积累趋势表明该通路在核桃种皮发育过程中保持活跃。本研究结果表明,兵塔1 号的莽草酸含量低于温185 和扎343,其可能通过降低苯丙氨酸供应影响下游代谢流,导致其总酚和总黄酮含量显著低于其他品种。单宁的合成依赖于类黄酮通路的下游延伸,涉及花青素还原酶和无色花青素还原酶催化的黄烷-3-醇单体聚合[22]。兵塔1 号的表儿茶素含量显著低于其他品种,表儿茶素作为单宁的主要单体单元[7],其含量不足可能直接限制了单宁的聚合效率。相关性分析结果进一步表明,在兵塔1 号中表儿茶素与单宁含量呈显著正相关,这与白花色苷还原酶基因通过其表达量调控单宁聚合的研究结果一致[27]。咖啡酸和对香豆酸作为苯丙烷代谢途径的重要分支产物,在不同品种中表现出显著的调控差异。3 个核桃品种的咖啡酸含量在发育后期均持续下降,兵塔1 号对香豆酸与单宁含量呈显著正相关,在温185和扎343中分别呈不显著正相关和负相关,这可能与4-香豆酸辅酶A 连接酶对羟基肉桂酸的底物偏好性有关[28]。仅扎343 中咖啡酸与单宁含量呈正相关,暗示其可能通过调控查尔酮合成酶活性分流代谢[28],导致黄酮醇积累增加。芦丁作为黄酮醇的代表物质,在兵塔1 号中与单宁含量呈负相关,这可能是由于黄酮醇合成酶与二氢黄酮醇-4-还原酶对共同底物的竞争[29]。比较特别的是,扎343中杨梅素等9个物质与单宁呈负相关,可能反映了该品种特有的代谢调控模式,可进一步通过基因表达分析验证。结合细胞结构与酚类物质含量差异的比较,发现单宁细胞的稀疏分布可能会导致莽草酸等前体物质从维管束向单宁细胞的运输路径延长,兵塔1 号莽草酸含量较扎343 降低的结果进一步表明,细胞数量不足可能直接削弱了前体物质的有效供给。表儿茶素作为单宁聚合的关键单体,在兵塔1 号中的含量(240.01 mg·g-1)较扎343(312.56 mg·g-1)下降23.2%,进一步印证单宁细胞数量不足可能降低了黄烷-3-醇单体的聚合效率。
核桃种皮中涩味物质主要为单宁、香豆酸、咖啡酸、绿原酸、儿茶素、表儿茶素等[17,30-31]。核桃种皮中单宁含量与核桃的贮藏[13]、营养[14]和口感[15]相关。核桃中单宁积累过程是一个复杂的生物合成和代谢调控过程,涉及多个途径和调控机制。结合多酚等物质的代谢途径可以发现,合成单宁的前体物质积累规律存在差异[32-34],导致了总单宁含量存在较大差异。笔者观察到品种间代谢差异可能与关键酶基因的遗传变异[8]有关。鉴于兵塔1 号在单宁代谢方面的独特优势,该品种可作为核桃多酚类物质研究及遗传改良的理想材料,通过多组学技术解析关键基因功能,培育低涩味、高品质的优异核桃品种。
4 结 论
不同品种核桃种皮中细胞形态、单宁等多酚类物质积累存在显著差异,且随着核桃果实生长发育而发生变化。核桃种皮单宁物质呈现单宁细胞→表皮细胞的积累模式,而兵塔1 号种皮单宁细胞数量少是其单宁合成速率与转运效率低下的形态学原因。结合已知的单宁合成路径,初步探究了核桃种皮多酚代谢的品种特异性,发现莽草酸含量低、没食子酸和对香豆酸等前体物质的积累不足、儿茶素积累提前结束及芦丁的竞争性抑制可能是兵塔1号单宁含量显著低于其他品种的关键因素。
[1] 相重扬. 《中国果树志·核桃卷》评介[J]. 中国果树,1997(2):57.XIANG Chongyang. Review of Chinese fruit tree and walnut roll[J]. China Fruits,1997(2):57.
[2] 王亚萍. 采收及采后处理对核桃内种皮颜色和酚类物质的影响[D]. 北京:北京林业大学,2022.WANG Yaping. Effects of harvest and post harvest treatment on the color and phenolics of walnut kernel pellicle[D]. Beijing:Beijing Forestry University,2022.
[3] 张燕,马家辉,王伟,任丽秋,李晓芹,朱建津,成向荣. 不同色泽核桃内种皮多酚和核桃仁分离蛋白理化性质的差异[J]. 浙江农业学报,2023,35(12):2923-2934.ZHANG Yan,MA Jiahui,WANG Wei,REN Liqiu,LI Xiaoqin,ZHU Jianjin,CHENG Xiangrong. Differences in physicochemical properties of polyphenols from walnut pellicle with different colors and proteins from walnut kernel[J]. Acta Agriculturae Zhejiangensis,2023,35(12):2923-2934.
[4] 崔茂凯,黄海涛,李庆杨,沈丹玉,肖鹏飞,刘毅华. 核桃仁内种皮中缩合单宁多聚体的分离与抗氧化功能分析[J/OL]. 中国油脂,2024:1-13. (2024-01-19). https://doi.org/10.19902/j.cnki.zgyz.1003-7969.230612.CUI Maokai,HUANG Haitao,LI Qingyang,SHEN Danyu,XIAO Pengfei,LIU Yihua. Isolation and analysis of antioxidant function of condensed tannin polysomes in the inner seed skin of walnut kernel[J/OL]. China Oils and Fats,2024:1-13. (2024-01-19). https://doi.org/10.19902/j.cnki.zgyz.1003-7969.230612.
[5] 俞文君,金强,李根,张锐,吴翠云,王新建,于军. 基于果实苦涩味新疆核桃资源遗传多样性分析[J]. 食品工业科技,2020,41(13):234-240.YU Wenjun,JIN Qiang,LI Gen,ZHANG Rui,WU Cuiyun,WANG Xinjian,YU Jun. Genetic diversity analysis of walnut germplasm resources based on bitter and astringent of fruits in Xinjiang[J]. Science and Technology of Food Industry,2020,41(13):234-240.
[6] 周高兴,伊再提古丽·加帕尔,李欣,王新建. 新疆核桃与域外核桃品种果实总多酚差异分析[J]. 北方园艺,2023(22):31-37.ZHOU Gaoxing,Yizaitiguli · Japarli,LI Xin,WANG Xinjian.Analysis of fruit total polyphenol differences between Xinjiang walnut and extra-territorial walnut varieties[J]. Northern Horticulture,2023(22):31-37.
[7] 刘雨霞,田鑫,杨笑,张小军,段国锋,刘群龙. 不同核桃品种内种皮苦涩味物质差异分析[J]. 果树学报,2021,38(2):222-230.LIU Yuxia,TIAN Xin,YANG Xiao,ZHANG Xiaojun,DUAN Guofeng,LIU Qunlong. Analysis of the differences in bitter and astringent substances in the pellicle of different walnut varieties[J]. Journal of Fruit Science,2021,38(2):222-230.
[8] HUANG R M,ZHOU Y,ZHANG J P,JI F Y,JIN F,FAN W,PEI D. Transcriptome analysis of walnut (Juglans regia L.) embryos reveals key developmental stages and genes involved in lipid biosynthesis and polyunsaturated fatty acid metabolism[J].Journal of Agricultural and Food Chemistry,2021,69(1):377-396.
[9] 盛芳. 核桃多酚代谢关键基因筛选及JrMYB8 功能解析[D].武汉:华中农业大学,2022.SHENG Fang. Screening of key genes in polyphenolic metabolism and functional characterization of JrMYB8 in walnut[D].Wuhan:Huazhong Agricultural University,2022.
[10] ZHAO W,LIU Y H,LI L,MENG H J,YANG Y,DONG Z B,WANG L,WU G L. Genome-wide identification and characterization of bHLH transcription factors related to anthocyanin biosynthesis in red walnut (Juglans regia L.)[J]. Frontiers in Genetics,2021,12:632509.
[11] 冶倩. 基于解剖学原理的核桃内果皮发育研究[D]. 阿拉尔:塔里木大学,2023.YE Qian. Development of walnut endocarp based on anatomical principle[D]. Alar:Tarim University,2023.
[12] 宋岩,王小红,张锐,刘春花,鱼尚奇,高山,张锐利. 新疆核桃品种间品质差异比较[J]. 中国粮油学报,2019,34(8):91-97.SONG Yan,WANG Xiaohong,ZHANG Rui,LIU Chunhua,YU Shangqi,GAO Shan,ZHANG Ruili. Comparison of quality differences among varieties of walnut from Xinjiang[J]. Journal of the Chinese Cereals and Oils Association,2019,34(8):91-97.
[13] 关长飞. 中国甜柿cDNA-SSAP 文库构建及自然脱涩相关基因功能验证[D]. 武汉:华中农业大学,2016.GUAN Changfei. Construction of cDNA-SSAP library and functional confirmation of genes involved in natural deastringency in C-PCNA persimmon[D]. Wuhan:Huazhong Agricultural University,2016.
[14] 周高兴. 新疆薄皮核桃种仁种皮褐变差异分析及温度和氧气对其影响的研究[D]. 阿拉尔:塔里木大学,2024.ZHOU Gaoxing. Differential analysis of seed coat browning of thin-skinned walnut seed kernels in Xinjiang and the effects of temperature and oxygen on it[D]. Alar:Tarim University,2024.
[15] 周晔. 核桃内种皮多酚分析与抗氧化活性[D]. 北京:中国林业科学研究院,2013.ZHOU Ye. Analysis of walnut pellicle polyphenols and antioxidant activities[D]. Beijing:Chinese Academy of Forestry,2013.
[16] 刘雨霞. 不同核桃品种内种皮苦涩味物质差异研究[D]. 太谷:山西农业大学,2021.LIU Yuxia. Study on the differences of bitter and astringent substances in walnut kernel pellicle of different varieties[D]. Taigu:Shanxi Agricultural University,2021.
[17] 陈旋勐,张岗,高静,颜永刚,魏瑶,陈莹. 山茱萸果实发育过程中单宁物质的分布与积累特征[J]. 西北植物学报,2021,41(11):1834-1842.CHEN Xuanmeng,ZHANG Gang,GAO Jing,YAN Yonggang,WEI Yao,CHEN Ying. Dynamic changes of tannins during fruit development of Cornus officinalis[J]. Acta Botanica Boreali-Occidentalia Sinica,2021,41(11):1834-1842.
[18] 时羽杰,邬晓勇,糜加轩,赵倩,刘欢欢,万雪琴. 核桃内种皮苦涩味品质代谢组学分析[J]. 西北农林科技大学学报(自然科学版),2021,49(6):54-64.SHI Yujie,WU Xiaoyong,MI Jiaxuan,ZHAO Qian,LIU Huanhuan,WAN Xueqin. Metabonomics of bitter taste quality of walnut kernel pellicle[J]. Journal of Northwest A & F University(Natural Science Edition),2021,49(6):54-64.
[19] 胡文朝,刘雪薇,郑雄,姚佩,史单华,刘群龙,张小军. 单宁酶对核桃内种皮酚类物质含量的影响[J]. 果树资源学报,2024,5(3):36-41.HU Wenzhao,LIU Xuewei,ZHENG Xiong,YAO Pei,SHI Danhua,LIU Qunlong,ZHANG Xiaojun. The effect of tannase on the content of phenolic substances in walnut kernel pellicle[J].Journal of Fruit Resources,2024,5(3):36-41.
[20] JIN Q,GAO S,MO R L,SHENG F,ZHANG Q L,WU C Y,ZHANG R,LUO Z R. A preliminary study for identifying genes associated with pellicle development in Xinjiang walnut (Juglans regia L.)[J]. Horticulturae,2022,8(9):784.
[21] JING S,WANG C L,YANG Y J,ZHU B H,HOU D H,GUO X C,ZHU Q G,KOU L P. Integrated transcriptomics and physiological parameters reveal the husk browning difference between two cultivars of pomegranate fruit[J]. Scientia Horticulturae,2024,336:113413.
[22] WANG J H,LI F J,ZHANG X Y,SUN W E,ALI M,LI X A,ZHANG X H. Combined transcriptomic and targeted metabolomic analysis reveals the mechanism of flesh browning in cold stored ‘Fuji’ apple fruit[J]. Scientia Horticulturae,2023,320:112195.
[23] LI Y,WANG J H,WANG K T,LYU S H,REN L Y,HUANG C Y,PEI D,XING Y L,WANG Y G,XU Y F,LI P P,XI J W,SI X L,YE H Y,HUANG J Q. Comparison analysis of widely-targeted metabolomics revealed the variation of potential astringent ingredients and their dynamic accumulation in the seed coats of both Carya cathayensis and Carya illinoinensis[J].Food Chemistry,2022,374:131688.
[24] ZHANG J,FENG R Y,XING X Y,HOU W H,MU X P,ZHANG J C,GAO Y G,DU J J,WANG P F. Transcriptome and weighted gene co-expression network analysis reveal key genes involved in the proanthocyanidin biosynthesis in Cerasus humilis[J]. Scientia Horticulturae,2024,325:112717.
[25] LIANG C M,YANG B,WEI Y,ZHANG P F,WEN P F. SA incubation induced accumulation of flavan-3-ols through activated VvANR expression in grape leaves[J]. Scientia Horticulturae,2021,287:110269.
[26] YU K J,DIXON R A,DUAN C Q. A role for ascorbate conjugates of (+)-catechin in proanthocyanidin polymerization[J]. Nature Communications,2022,13(1):3425.
[27] ROBINSON S P,BOGS J,MCDAVID D A J,HOOPER L C,SPEIRS J,WALKER A R. Transgenic grapevines with decreased expression of tannin synthesis genes have altered grape and wine flavonoid composition[J]. Australian Journal of Grape and Wine Research,2021,27(1):106-117.
[28] LI C X,HUANG D,WEI R,HONG Y Y,ZHANG W E,PAN X J. Genome-wide characterization,identification,and function analysis of candidate JsMYB genes involved in regulating flavonol biosynthesis in Juglans sigillata Dode[J]. Scientia Horticulturae,2023,317:112044.
[29] LEI T,HUANG J,RUAN H X,QIAN W,FANG Z,GU C Y,ZHANG N N,LIANG Y X,WANG Z Y,GAO L P,WANG Y S. Competition between FLS and DFR regulates the distribution of flavonols and proanthocyanidins in Rubus chingii Hu[J].Frontiers in Plant Science,2023,14:1134993.
[30] 王世明. 几个核桃品种内种皮苦涩味物质比较[J]. 中国果业信息,2021,38(2):57.WANG Shiming. Comparison of bitter and astringent substances in the endocarp of several walnut varieties[J]. China Fruit News,2021,38(2):57.
[31] 王芳,郑先强,糜加轩,吕健,朱艳,时羽杰,万雪琴,杨汉波. 10个核桃无性系鲜果内种皮苦涩味相关物质及易剥性比较[J].经济林研究,2022,40(1):66-73.WANG Fang,ZHENG Xianqiang,MI Jiaxuan,LÜ Jian,ZHU Yan,SHI Yujie,WAN Xueqin,YANG Hanbo. Comparison of bitter taste-related substances and peeling difficulty of fresh nut endotesta among ten walnut clones[J]. Non-wood Forest Research,2022,40(1):66-73.
[32] 徐徐. 山核桃果实发育过程类黄酮合成及代谢分析[D]. 杭州:浙江农林大学,2021.XU Xu. Analysis of flavonoid synthesis and metabolism during the development of pecan fruit[D]. Hangzhou:Zhejiang A & F University,2021.
[33] 陆胜波. 铁核桃果实青皮和内种皮芦丁合成的转录组与代谢组分析[D]. 贵阳:贵州大学,2020.LU Shengbo. Transcriptomes and metabonomics profiling reveals rutin biosynthesis of green husk and kernel pellicle in walnut fruit (Juglans sigillata Dode)[D]. Guiyang:Guizhou University,2020.
[34] 李琳. 红瓤核桃颜色形成及相关基因研究[D]. 郑州:河南农业大学,2022.LI Lin. Study on color formation and related genes of red walnut[D]. Zhengzhou:Henan Agricultural University,2022.