盐胁迫是制约果树生长发育、降低果实产量与品质的主要非生物胁迫因子[1]。全球约6%~10%的陆地面积受盐渍化影响,特别是在中国西部盐碱地区占据了大量耕地面积[2]。盐胁迫通过引发离子毒害、渗透失衡和氧化损伤抑制果树生长,导致苹果等重要经济作物显著减产[3]。因此,解析果树耐盐机制并开发新型抗逆策略已成为产业可持续发展的迫切需求。
植物主要通过离子平衡、渗透调节、抗氧化机制以及基因表达调控来响应盐胁迫,包括通过SOS 信号通路维持离子稳态[4]、合成可溶性物质以调节渗透势[5]、激活抗氧化系统清除活性氧[6]、调控耐盐相关基因网络增强耐逆性[7]。近年研究发现,植物-微生物互作可介导植物的耐盐响应。微生物信号分子N-酰基高丝氨酸内酯(AHLs)可被植物识别并诱导抗逆响应。例如,N-酰基高丝氨酸内酯家族的3OC6-HSL 可通过调节生长素合成、离子平衡以及增强抗氧化能力以提高拟南芥的耐盐性[8-9],但在果树等木本作物中的相关研究尚未见报道。
笔者以苹果砧木八棱海棠(Malus robusta)实生苗为材料,旨在探究AHLs 家族成员3OC8-HSL 对其耐盐性的潜在调控作用,重点分析3OC8-HSL 对实生苗生长、离子平衡及耐盐相关基因表达的影响,并结合转录组数据解析其可能的分子调控机制,为果树砧木改良和抗盐栽培提供理论依据。
1 材料和方法
1.1 试验材料
试验所用砧木为八棱海棠实生苗,购于山东寿禾种业有限公司。试剂:N-羰基辛酰基高丝氨酸内酯(3OC8-HSL)(CAS:174495-39-9)购自阿拉丁试剂(上海)有限公司。植物生长所用基质土的土壤配方为丹麦品氏泥炭土(进口土,货号为msh-1013)∶江苏培蕾基质(江苏蓓蕾基质科技发展有限公司的播种育苗基质)∶蛭石=4∶2∶1(体积比,后同)。实生苗在华中农业大学玻璃温室中培养,白天温度20~28 ℃,夜间15~20 ℃,相对湿度60%~80%,以自然光照为主,日均光照10~14 h。
1.2 试验方法
1.2.1 八棱海棠种子消毒与种植方法 种子消毒:使用1 mol·L-1 的NaOH 溶液浸泡八棱海棠种子15 min,去除果胶。随后,使用3%的NaClO 消毒15 min,使用无菌水洗涤5~7 次。催芽:将消毒后的种子平铺在湿润的灭菌沙土上,放入4 ℃冰箱黑暗培养6~7 周。待种子露白后,散播在灭菌的蛭石中,在温室中培养30 d左右后移植至装有灭菌的商道基质∶泥炭土∶蛭石为4∶2∶1 的混合基质的育苗盆内。实验共设置4 个处理组:对照组(CK,蒸馏水)、3OC8-HSL 处理组(AC,10 μmol·L-1 3OC8-HSL)、盐胁迫组(Salt,200 mmol·L-1 NaCl+蒸馏水)以及3OC8-HSL+盐胁迫组(AS,200 mmol·L-1 NaCl+10 μmol·L-1 3OC8-HSL)。在试验过程中,3OC8-HSL处理组(AC)和3OC8-HSL+盐胁迫处理组(AS)的每株实生苗根部均匀滴加10 mL 3OC8-HSL 溶液;对照组(CK)和盐胁迫组(Salt)则滴加等体积蒸馏水,以保证试验处理条件一致。每个试验处理组设置12株实生苗,总计48株。每个处理组随机摆放于培养箱内,以减少位置效应对试验结果的影响。3OC8-HSL 处理分别在移栽后的第1、7、14 天进行,共计3次;而盐处理则采用200 mmol·L-1 NaCl溶液,每3 d 处理1 次,并持续整个试验周期,以模拟长期盐胁迫环境。所有实生苗在华中农业大学玻璃温室中生长45 d,其间采用自然光照,并通过通风和遮阳调节环境条件。
1.2.2 八棱海棠实生苗Na+和K+元素检测方法 样品处理:将新鲜八棱海棠实生苗置于105 ℃的烘箱中杀青30 min,并在85 ℃下进行烘干至恒质量,称量干质量。样品消解:称取5 mg 干样,加入125 μL 30% H2O2和250 μL 65% HNO3混合液,于消解管中进行消解。将消解管置于加热块上,在140 ℃下消解80 min,直至样品完全消解且溶液澄清。元素测定:消解后的样品用去离子水定容,并稀释至终浓度含3.5%或7% HNO3溶液。Na+和K+含量采用电感耦合等离子体发射光谱仪(ICP-OES)进行测定。
1.2.3 八棱海棠实生苗叶绿素含量检测方法 采用植物叶绿素含量检测试剂盒(Chlorophyll Assay Kit,100T/96S,北京索莱宝科技有限公司),按照试剂盒说明书提取叶片叶绿素,在645 nm 和663 nm 波长下测定吸光度,计算叶绿素a、b及总叶绿素含量。
1.2.4 八棱海棠实生苗丙二醛(MDA)含量检测方法 采用丙二醛(MDA)含量检测试剂盒(Malondialdehyde (MDA) Content Assay Kit,100T/96S,北京索莱宝科技有限公司),参照试剂盒说明书,在532 nm波长下比色测定。
1.2.5 转录组测序及分析 转录组测序委托北京擎科生物科技股份有限公司完成。测序获得原始的数据,去除测序读长低质量、接头污染和高含量的未知碱基读长。读长过滤后,采用HISAT2 软件将Clean Reads 比对到苹果参考基因组(Malus domestica GDDH13 v1.1)。基因表达量以FPKM(Fragments Per Kilobase of transcript per Million mapped reads)进行归一化。差异表达分析采用DESeq2,筛选条件为|log2FoldChange|>1 且P. adjust<0.05。对筛选到的差异表达基因(differentially expressed genes,DEGs)通过topGO 软件进行GO 功能富集分析,显著性标准为P. adjust<0.05。
1.2.6 实时荧光定量PCR 分析 利用RNAprep Pure 多糖多酚植物总RNA 提取试剂盒(DP441)(天根生化科技有限公司,北京)提取RNA。超微量紫外分光光度法检测RNA样品纯度,1%琼脂糖凝胶电泳检测RNA 完整性。采用PrimeScriptTM RT reagent Kit with gDNAEraser(TaKaRa,大连)将RNA 反转录成cDNA。用TBGreen® Premix Ex TaqTMⅡ荧光定量试剂盒(TaKaRa,大连)进行RT-qPCR 反应。扩增体系10 μL,每个样品3次重复,所用引物序见表1。
表1 RT-qPCR 所用引物
Table 1 Primers used in RT-qPCR

基因名称Gene name EF-1α基因号Gene ID DQ341381 MdPAL MD14G1224400 MdGST MD15G1132200 MdHKT1 MD05G1258000 MdLRR-RLK MD17G1099500引物序列(5′-3′)Primer sequence F:ATTCAAGTATGCCTGGGTGC R:CAGTCAGCCTGTGATGTTCC F:CTTAGTGAGAAAGTCGGTGAGCT R:CTGACAAGAACATGACCTCCTCT F:AAATCCACCCAACGTCTACTACC R:CACAAATGCGATCGAACTTCTCTC F:ATGTGGGATGCAGTTTCAAAGC R:CCTCCTTTAACGTCTCCTCCTTC F:GGATGAGTATGAAAAGCTCTGCC R:ATGGTTCCTGCTGTCTATGTGAG
使用LightCycler® 480 实时荧光定量PCR 仪(Roche,瑞士)进行扩增,反应程序为:95 ℃ 3 min,95 ℃ 10 s,60 ℃ 5 s,72 ℃ 25 s,40 个循环。以苹果EF-1α(DQ341381)为内参基因,用2-ΔΔCT法计算基因相对表达量。
1.2.7 数据分析方法 使用 Excel 2020 软件整理数据并作图,采用GraphPad Prism 9.3.0 软件进行统计分析。数据采用单因素方差分析(one-way ANOVA)并进行Tukey多重比较(P<0.05)。
2 结果与分析
2.1 施用3OC8-HSL对八棱海棠实生苗生长的影响
不同处理对八棱海棠实生苗株高和鲜质量的影响如图1-A所示。正常条件下,3OC8-HSL处理的幼苗鲜质量和株高分别增加了17.23%和23.74%;在盐胁迫条件下,3OC8-HSL 处理的幼苗鲜质量和株高分别增加了26.38%和11.61%(图1-B~C)。

图1 正常和盐胁迫下施用3OC8-HSL 对八棱海棠实生苗生长的影响
Fig. 1 Effects of 3OC8-HSL application on the growth of Malus robusta seedlings under normal and salt stress conditions
2.2 施用3OC8-HSL 对八棱海棠实生苗叶绿素和丙二醛(MDA)含量的影响
正常条件下,3OC8-HSL 处理显著提高了八棱海棠幼苗的叶绿素含量,叶绿素总含量、叶绿素a和叶绿素b 含量分别增加了4.06%、4.10%和3.94%(图2-A~C);同时根系的MDA 含量降低了2.22%(图2-D)。盐胁迫条件下,3OC8-HSL 处理显著提高了叶绿素含量,叶绿素总含量、叶绿素a 和叶绿素b 含量分别增加了36.98%、40.43%和28.15%(图2-A~C);同时根系的MDA 含量降低了36.20%(图2-D)。
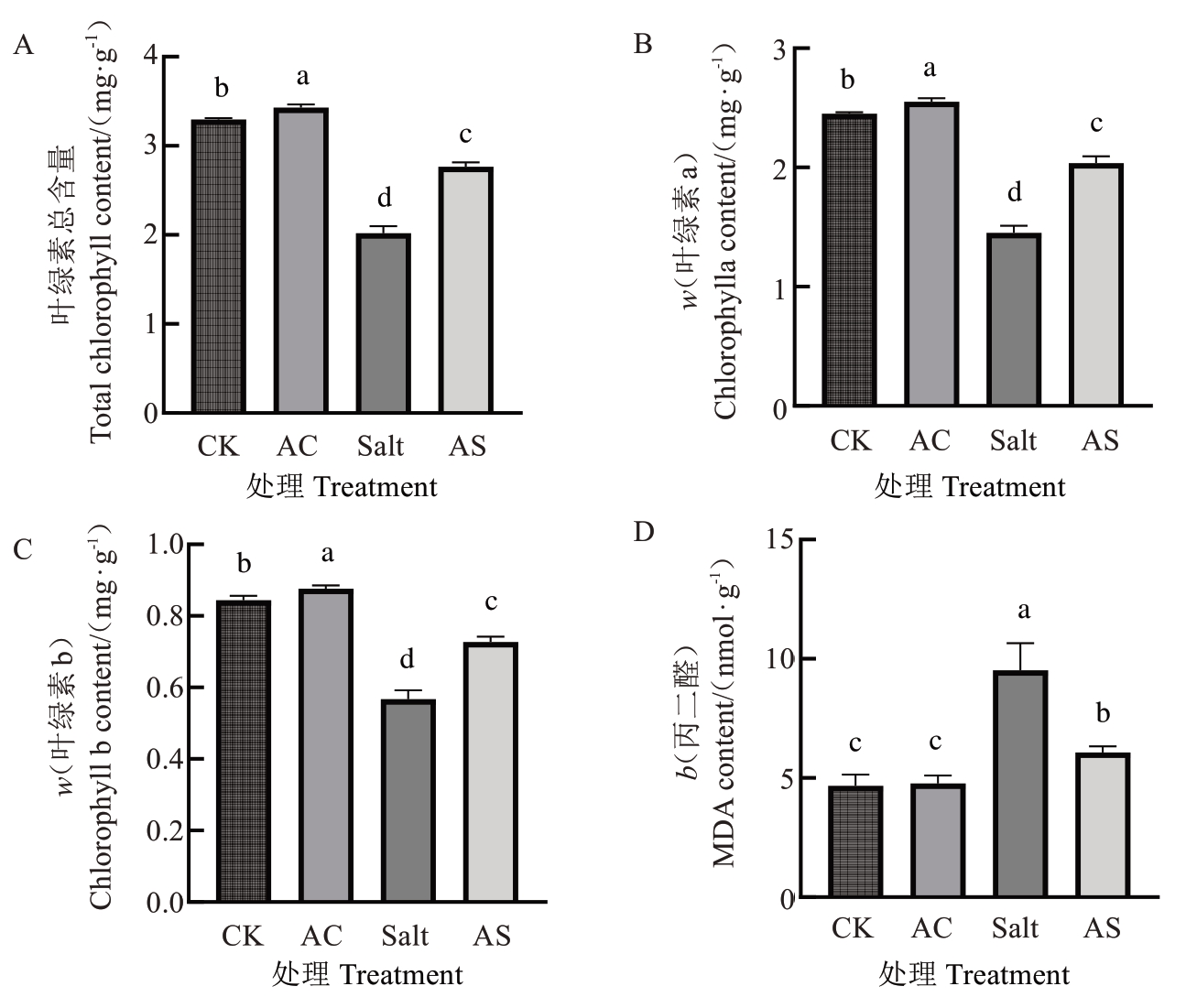
图2 正常和盐胁迫下施用3OC8-HSL 对八棱海棠实生苗叶绿素和MDA 含量的影响
Fig. 2 Effects of 3OC8-HSL application under normal and salt stress conditions on chlorophyll and MDA contents in Malus robusta seedlings
2.3 施用3OC8-HSL 对八棱海棠实生苗Na+和K+含量的影响
正常条件下,3OC8-HSL处理的八棱海棠幼苗叶片Na+含量减少了12.54%(图3-A),K+含量增加了15.00%(图3-B),Na+/K+比值降低了23.98%(图3-C)。盐胁迫条件下,3OC8-HSL处理的八棱海棠幼苗叶片Na+含量减少了11.01%(图3-A),K+含量增加了19.46%(图3-B),Na+/K+比值下降了22.17%(图3-C)。
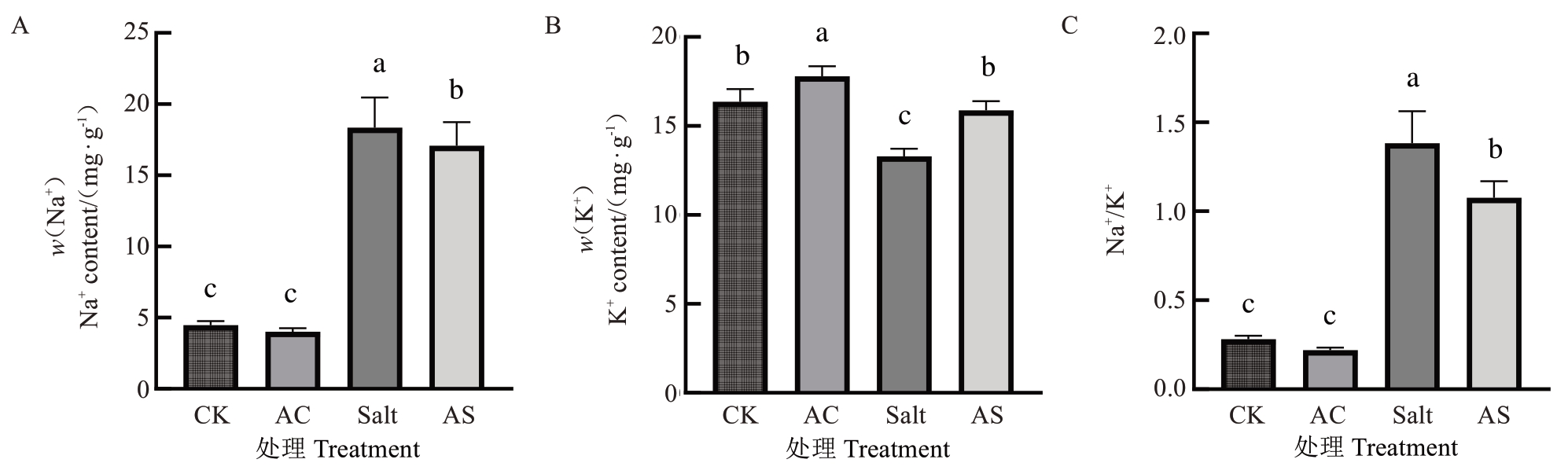
图3 正常和盐胁迫下施用3OC8-HSL 对八棱海棠实生苗Na+含量和K+含量的影响
Fig. 3 Effects of 3OC8-HSL application under normal and salt stress conditions on Na+ and K+ content in Malus robusta seedlings
2.4 施用3OC8-HSL 对八棱海棠实生苗根系转录组的影响
为探究3OC8-HSL 增强八棱海棠耐盐性的潜在机制,利用转录组测序技术分析了盐胁迫条件下八棱海棠根系中受3OC8-HSL 诱导的差异表达基因。与对照相比,3OC8-HSL处理产生560个DEGs,其中177个上调表达,383个下调表达(图4-A)。这些DEGs的GO富集分析发现,生物学过程(biological process,BP)富集的前5 个类别分别是伤害反应(response to wounding)、次级代谢物生物合成过程(secondary metabolite biosynthetic process)、苯丙烷类化合物生物合成过程(phenylpropanoid biosynthetic process)、苯丙烷类代谢过程(phenylpropanoid metabolic process)和角质层发育(cuticle development)(图4-B)。
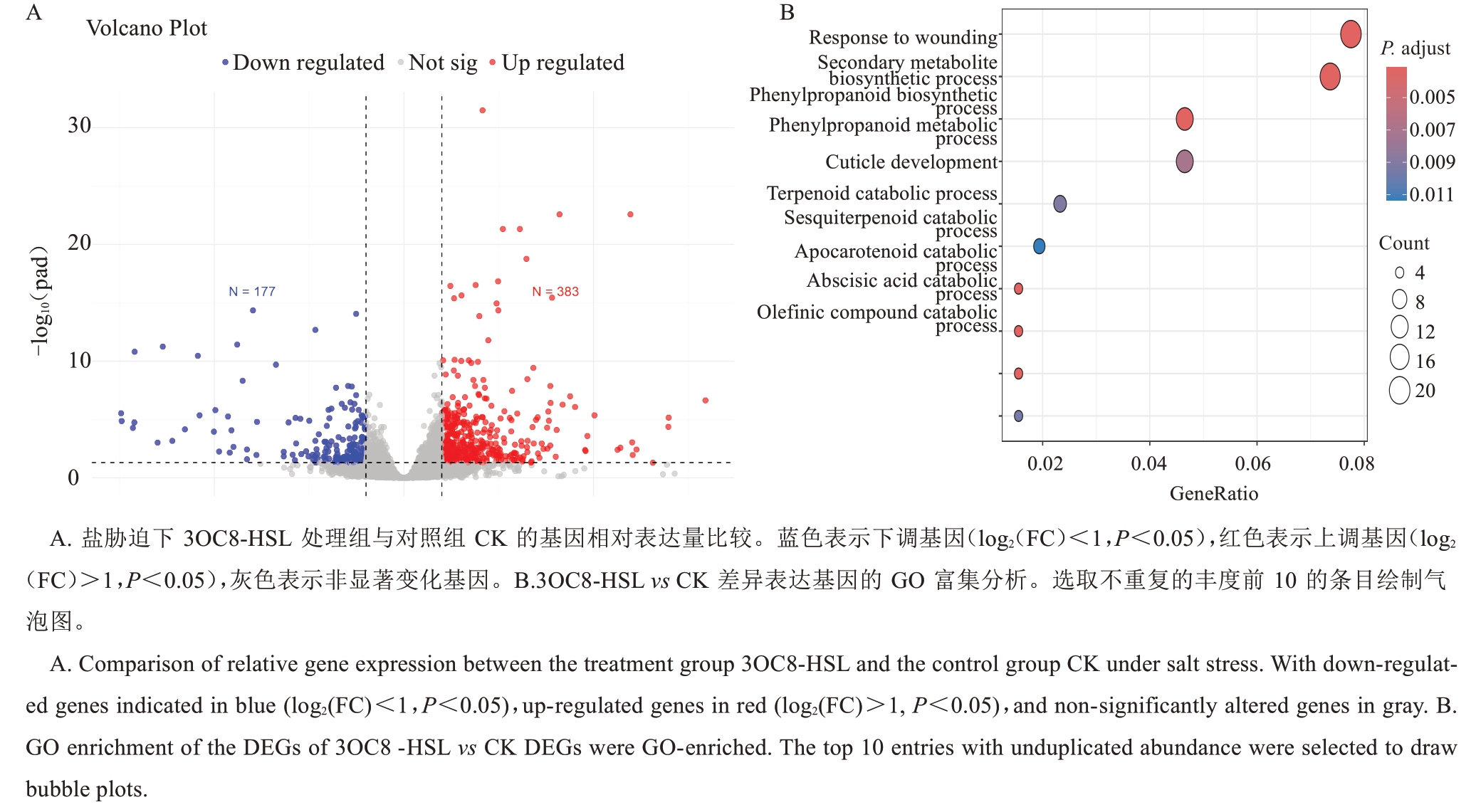
图4 盐胁迫下施加3OC8-HSL 诱导基因差异表达图
Fig. 4 The application of 3OC8-HSL induces differential gene expression under salt stress
2.5 盐胁迫下施用3OC8-HSL 诱导八棱海棠实生苗根系抗逆相关基因上调表达
在盐胁迫条件下,基于转录组差异分析结果及GO/KEGG 富集通路,同时参考耐盐相关文献,笔者选取了4 个代表性抗逆基因(MdPAL、MdGST、MdHKT1 和MdLRR-RLK)进行qPCR 验证。结果显示,3OC8-HSL 处理显著上调了八棱海棠根系中4 个关键基因的表达,包括编码苯丙氨酸解氨酶的MdPAL(MD14G1224400)、编码谷胱甘肽-S-转移酶的MdGST(MD15G1132200)、编码高亲和性钾转运蛋白的MdHKT1(MD05G1258000)以及编码类受体蛋白激酶的MdLRR-RLK(MD17G1099500)。其中,MdPAL、MdHKT1 和MdLRR-RLK 在处理7 d 后表达量分别上调4.6、9.6 和8.4 倍(P<0.05 或P<0.01),MdGST在24 h上调6.4倍(P<0.01)(图5)。
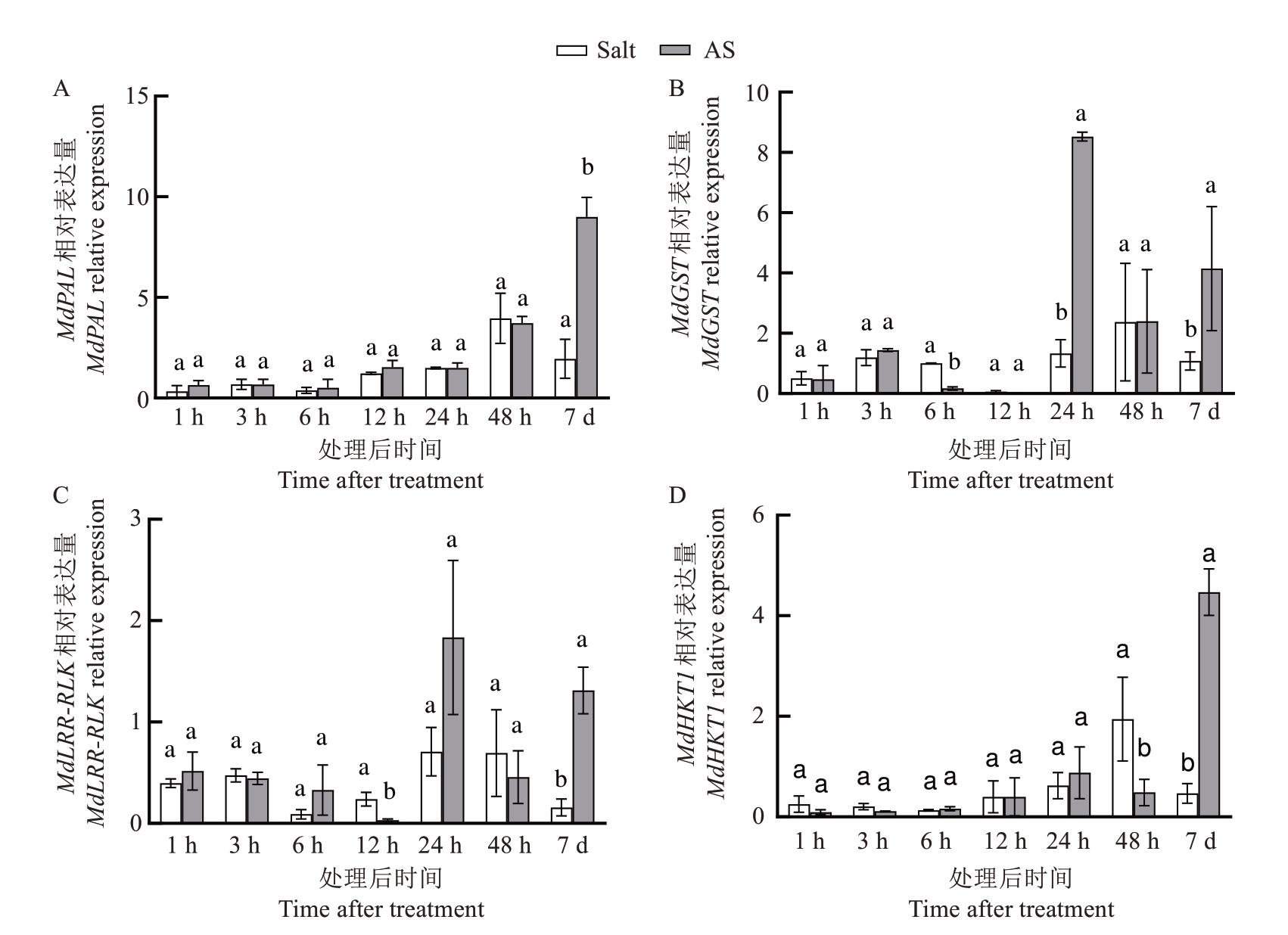
图5 盐胁迫下3OC8-HSL 处理调控抗盐基因表达
Fig. 5 Analysis of the regulation of salt-responsive gene expression by 3OC8-HSL treatment under salt stress
3 讨 论
N-酰基高丝氨酸内酯(AHLs)作为细菌群体感应信号分子,近年来发现能够被多种植物识别,从而调控植物的生长发育及抗逆性[10-12]。Zhao 等[8]研究发现,施用3OC6-HSL 可显著提高盐胁迫下拟南芥和小麦的生物量和叶绿素含量,并显著降低MDA积累量和Na+/K+比值,从而提升植物的耐盐性。蔡硕珍[13]也指出,AHLs可通过激活植物体内的信号转导通路,促进渗透调节物质积累和离子平衡,有效缓解盐胁迫造成的生理损伤。然而,AHLs 在木本果树中的作用鲜有系统报道。笔者首次证明,3OC8-HSL 在八棱海棠实生苗中可显著提升株高、鲜质量及叶绿素含量,优化盐胁迫下的生长表现,拓展了AHLs在果树耐盐性调控中的应用前景。
盐胁迫下,离子毒害和氧化损伤是影响植物生长的主要因素,维持细胞内Na+/K+平衡是提高植物耐盐性的关键机制[14-16]。本研究结果显示,3OC8-HSL 处理显著降低了八棱海棠叶片和根系中的Na+含量,提高了K+含量,并有效降低了Na+/K+比值,缓解了盐胁迫带来的离子失衡,与Zhao等[8]在拟南芥、小麦等草本植物中的研究一致。此外,3OC8-HSL还显著降低了丙二醛含量,表明其可减轻盐胁迫诱导的膜脂过氧化损伤,增强抗氧化能力。综上,3OC8-HSL可能通过调控Na+和K+的吸收与分配,维持离子稳态,并减轻氧化胁迫,从而提高了八棱海棠对盐胁迫的适应性。
本试验结果表明,3OC8-HSL 处理诱导了大量差异表达基因。GO 富集分析表明,差异基因主要涉及苯丙烷代谢、谷胱甘肽代谢、角质层发育和胁迫响应等生物学过程和代谢通路。这些通路均与植物的耐盐、抗氧化和细胞稳态密切相关。基于转录组数据、富集结果及相关文献,选取了4个代表性耐盐通路的关键基因进行qPCR 验证,包括调控木质素合成和抗逆性的MdPAL[17]、参与抗氧化和解毒的MdGST[18]、调节Na+/K+平衡的MdHKT1 以及感知胁迫信号的MdLRR-RLK[19-21]。结果显示,在3OC8-HSL 处理下,这些基因的表达显著上调,与转录组数据一致,进一步证明了3OC8-HSL 可通过激活多条耐盐相关分子通路增强八棱海棠的耐盐性。核心耐盐相关基因的协同上调表明,3OC8-HSL 能多层次调控离子稳态、抗氧化和信号转导等抗逆机制,提升了八棱海棠对盐胁迫的适应能力。研究结果丰富了AHLs 在木本果树耐盐性调控方面的理论基础,为后续功能基因挖掘与耐盐分子设计育种提供了新思路。
4 结 论
本研究结果揭示了3OC8-HSL 在提升八棱海棠耐盐性中的作用。结果表明,施加3OC8-HSL 能显著促进八棱海棠实生苗的生长,增强其在盐胁迫下的适应性,具体表现为株高、鲜质量及叶绿素含量增加,Na+/K+比值下降,MDA 含量降低(P<0.05)。3OC8-HSL 可能通过激活苯丙烷生物合成、谷胱甘肽代谢等多条耐盐相关通路,上调MdPAL、MdGST、MdHKT1、MdLRR-RLK 等关键基因表达,维持离子稳态,增强抗氧化能力,减轻细胞膜损伤。上述结果不仅丰富了AHLs 信号分子在木本果树耐盐性调控领域的作用机制,也为AHLs 类物质在果树耐盐性改良及盐碱地高效栽培中的应用提供了理论依据和实践支撑。
[1] EL- RAMADY H,FAIZY S,AMER M,ELSAKHAWY T,OMARA A E,EID Y,BREVIK E. Management of salt-affected soils:A photographic mini-review[J]. Environment,Biodiversity and Soil Security,2022,6(2022):61-79.
[2] MUNNS R,TESTER M. Mechanisms of salinity tolerance[J].Annual Review of Plant Biology,2008,59:651-681.
[3] 王雷,郭岩,杨淑华. 非生物胁迫与环境适应性育种的现状及对策[J]. 中国科学:生命科学,2021,51(10):1424-1434.WANG Lei,GUO Yan,YANG Shuhua. Designed breeding for adaptation of crops to environmental abiotic stresses[J]. Scientia Sinica (Vitae),2021,51(10):1424-1434.
[4] MUNNS R. Approaches to identifying genes for salinity tolerance and the importance of timescale[J]. Methods in Molecular Biology,2010,639:25-38.
[5] 孙聪聪,赵海燕,郑彩霞. NaCl 胁迫对银杏幼树渗透调节物质及脯氨酸代谢的影响[J]. 植物生理学报,2017,53(3):470-476.SUN Congcong,ZHAO Haiyan,ZHENG Caixia. Effects of Na-Cl stress on osmolyte and proline metabolism in Ginkgo biloba seedling[J]. Plant Physiology Journal,2017,53(3):470-476.
[6] SAHA J,BRAUER E K,SENGUPTA A,POPESCU S C,GUPTA K,GUPTA B. Polyamines as redox homeostasis regulators during salt stress in plants[J]. Frontiers in Environmental Science,2015,3:21.
[7] ZHU J K. Plant salt tolerance[J]. Trends in Plant Science,2001,6(2):66-71.
[8] ZHAO Q,YANG X Y,LI Y,LIU F,CAO X Y,JIA Z H,SONG S S. N-3-oxo-hexanoyl-homoserine lactone,a bacterial quorum sensing signal,enhances salt tolerance in Arabidopsis and wheat[J]. Botanical Studies,2020,61(1):8.
[9] 艾秋实. N-酰基高丝氨酸内酯调控拟南芥抗盐能力及其与GCR2 结合特征的研究[D]. 保定:河北农业大学,2015.AI Qiushi. Research of N-acyl-homoserine lactones regulation of Arabidopsis salt resistance and its binding characteristics with GCR2[D]. Baoding:Hebei Agricultural University,2015.
[10] BABENKO L M,KOSAKIVSKA I V,ROMANENKO K O.Molecular mechanisms of N-acyl homoserine lactone signals perception by plants[J]. Cell Biology International,2022,46(4):523-534.
[11] JIN G P,LIU F,MA H,HAO S Y,ZHAO Q,BIAN Z R,JIA Z H,SONG S S. Two G-protein-coupled-receptor candidates,Cand2 and Cand7,are involved in Arabidopsis root growth mediated by the bacterial quorum-sensing signals N-acyl-homoserine lactones[J]. Biochemical and Biophysical Research Communications,2012,417(3):991-995.
[12] HARTMANN A,KLINK S,ROTHBALLER M. Importance of N-acyl-homoserine lactone-based quorum sensing and quorum quenching in pathogen control and plant growth promotion[J].Pathogens,2021,10(12):1561.
[13] 蔡硕珍. N-羰基辛酰基高丝氨酸内酯调控植物生长和抗病反应过程及其应用初探[D]. 保定:河北农业大学,2021.CAI Shuozhen. N-(3-oxooctanoyl)-L-homoserine lactone regulates plant growth and disease resistance and its application[D].Baoding:Hebei Agricultural University,2021.
[14] ACOSTA-MOTOS J R,ORTUÑO M F,BERNAL-VICENTE A,DIAZ-VIVANCOS P,SANCHEZ-BLANCO M J,HERNANDEZ J A. Plant responses to salt stress:Adaptive mechanisms[J]. Agronomy,2017,7(1):18.
[15] BITTENCOURT P P,ALVES A F,FERREIRA M B,DA SILVA IRINEU L E S,PINTO V B,OLIVARES F L. Mechanisms and applications of bacterial inoculants in plant drought stress tolerance[J]. Microorganisms,2023,11(2):502.
[16] LIU F,BIAN Z,JIA Z H,ZHAO Q,SONG S S. The GCR1 and GPA1 participate in promotion of Arabidopsis primary root elongation induced by N-acyl-homoserine lactones,the bacterial quorum-sensing signals[J]. Molecular Plant-Microbe Interactions,2012,25(5):677-683.
[17] CHEYNIER V,COMTE G,DAVIES K M,LATTANZIO V,MARTENS S. Plant phenolics:Recent advances on their biosynthesis,genetics,and ecophysiology[J]. Plant Physiology and Biochemistry,2013,72:1-20.
[18] CHRONOPOULOU E G,LABROU N E. Glutathione transferases:Emerging multidisciplinary tools in red and green biotechnology[J]. Recent Patents on Biotechnology,2009,3(3):211-223.
[19] LUO M H,CHU J,WANG Y,CHANG J Y,ZHOU Y,JIANG X Y. A high-affinity potassium transporter (MeHKT1) from cassava (Manihot esculenta) negatively regulates the response of transgenic Arabidopsis to salt stress[J]. BMC Plant Biology,2024,24(1):372.
[20] TANG J,HAN Z F,SUN Y D,ZHANG H Q,GONG X Q,CHAI J J. Structural basis for recognition of an endogenous peptide by the plant receptor kinase PEPR1[J]. Cell Research,2015,25(1):110-120.
[21] SOLTABAYEVA A,DAULETOVA N,SERIK S,SANDYBEK M,OMONDI J O,KURMANBAYEVA A,SRIVASTAVA S. Receptor-like kinases (LRR-RLKs) in response of plants to biotic and abiotic stresses[J]. Plants,2022,11(19):2660.