观赏海棠(Malus crabapples)属蔷薇科(Rosaceae)苹果属(Malus),主要分布在亚欧大陆与北美洲,是极具观赏性的苹果资源[1]。观赏海棠在中国具有非常悠久的种植历史,因其花繁茂,花色丰富,素有“华贵妃”美誉[2]。近年来,随着农业产业结构的转型、农村经济的发展,人们对园林景观多样性和园艺植物美的需求不断提升。观赏性海棠因具有花色鲜艳、花期集中、成花量大等特点广泛应用于景区、园林、城市绿化以及盆栽庭院美化等领域[3-4]。21世纪初,中国观赏海棠主要从北美、欧洲等地引进,杂交选育工作也以鲜食苹果为主。观赏海棠育种研究起步较晚,目前主要以描述型性状如花期、物候期、花色等性状为育种目标[5-6]。
观赏海棠遗传背景复杂,属于高度杂合体,杂交后代花的性状会表现出较大的差异。目前,对花器官数量性状的系统性研究较少,制约了优异观赏资源的挖掘与选育[3,6-7]。在观赏性状中,花直径、花瓣数量、雄蕊与雌蕊数量,影响花序的立体层次感与观赏质感。重瓣性品种通常具有更高的美学价值;花梗长度则突显花朵的空间层次感,具有较强的视觉冲击。这些性状在牡丹[8]、百合[9]和月季[10]等关于花性状研究中均表现为连续变异的数量性状特征,通常是由主基因与多基因,或主基因与微效多基因协同调控。
红勋1 号是从中国新疆红肉苹果[Malus sieversii f. neidzwetzkyana(Dieck)Langenf]中选育的优异资源,具有早花早果、花色鲜红、花型单瓣等特点,其枝、叶、花和果实均呈现出艳丽的浓红色,具有较高的观赏价值,同时也具有丰富的必需氨基酸和花青苷[11]。重瓣海棠(Malus spp.)则是一种花色淡粉色、花型重瓣的苹果野生种,其花朵重瓣层叠、花期集中、花量大、花香四溢,具有极高的观赏价值[12]。因此,笔者以花器官表型差异较大的红勋1 号和重瓣海棠为亲本构建的F1代杂交群体为研究对象,对其花直径、花瓣数量、雄蕊数量、雌蕊数量以及花梗长度5 个数值型性状以及花色和单重瓣2 个描述型性状进行遗传变异和遗传模式分析,为观赏海棠优良品种选育提供理论支撑。
1 材料和方法
1.1 材料
以2015 年通过红勋1 号×重瓣海棠杂交获得的204 个F1代株系为试验材料,其中红勋1 号为母本。该杂交群体定植于辽宁省兴城市中国农业科学院果树研究所,于每个株系的盛花期采集10 枚花朵进行花表型性状调查。
1.2 表型性状调查与测定方法
参照《苹果种质资源描述规范和数据标准》[13]对F1代杂交群体204 个株系的花直径、花瓣数量、雄蕊数量、雌蕊数量和花梗长度5 个数值型性状以及花色和单重瓣2 个描述型性状进行测定与调查。
1.3 数据处理与统计分析
1.3.1 表型变异及遗传分析 使用IBM SPSS Statistics 27 软件对试验数据进行遗传分析,使用Origin 2018 软件绘图。参照郭淑青等[14]、潘静等[15]、叶雪莲等[16]的方法,计算亲本值P1 和P2、方差Vp1 和Vp2、中亲值MP,计算杂交后代群体的平均值F、标准差s、方差VH、变异系数CV、遗传传递力Ta、广义遗传力Hb2、杂种优势率H、超高亲率HH,计算公式如下:
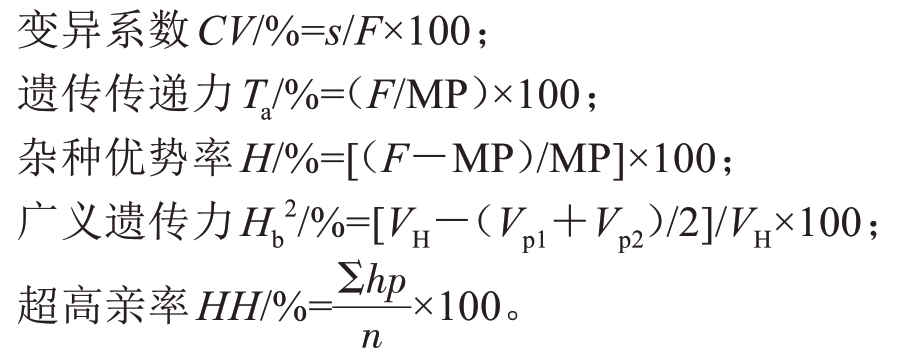
式中,hp 为表型值超过高亲的杂交F1代,n 为杂交F1群体数量。
1.3.2 遗传模型分析 根据数量性状主基因+多基因混合遗传模型分析方法,调用SEA 软件包分析杂交F1代(假F2)5 个数值型性状单个分离世代的遗传模型[15],采用最小AIC(Akaike's information criterion)值准则筛选最适合的遗传模型,最小二乘法估计相应的主基因效应[17]。
2 结果与分析
2.1 花数值型性状遗传分析
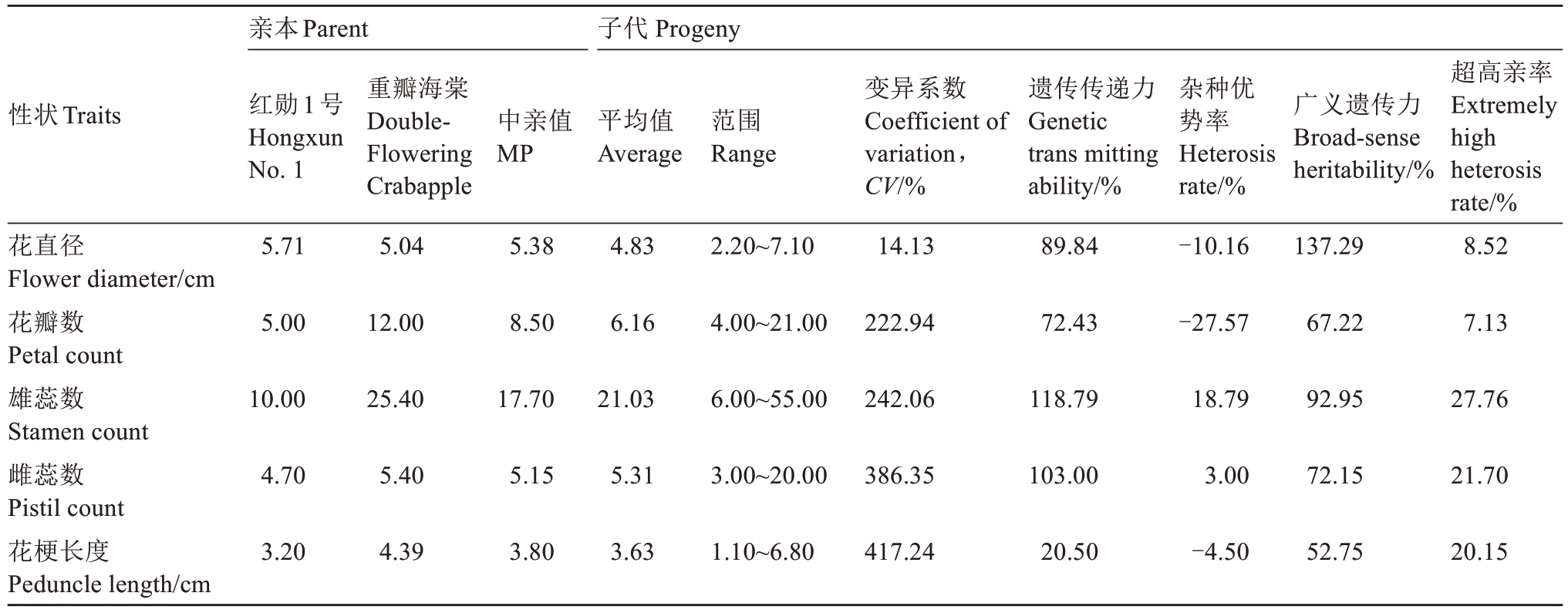
2.1.1 花直径的遗传变异 亲本红勋1 号和重瓣海棠的花直径分别为5.71 cm 和5.04 cm。子代花直径平均值为4.83 cm,变幅在2.20~7.10 cm 之间,符合正态分布(图1),变异系数为14.13%,低于20%,表明花直径在F1代中存在一定程度的变异,但相较于其他性状变异程度较小,未表现出广泛的分离现象。遗传传递力为89.84%,花直径可能受亲本影响程度较高。杂种优势率为-10.16%,呈现负向杂种优势,F1代花直径平均值低于中亲值。广义遗传力为137.29%,表明遗传因素对花直径表型变异的贡献较大(表1)。
表1 红勋1 号和重瓣海棠F1群体花数值型性状的遗传变异
Table 1 The heritable variation of quantitative traits in the F1 progenies of Hongxun No. 1×Double-Flowering Crabapple
亲本Parent 子代 Progeny性状Traits 红勋1号Hongxun No. 1中亲值MP平均值Average范围Range广义遗传力Broad-sense heritability/%花直径Flower diameter/cm花瓣数Petal count雄蕊数Stamen count雌蕊数Pistil count花梗长度Peduncle length/cm 5.71重瓣海棠Double-Flowering Crabapple 5.04 5.38 4.83 2.20~7.10变异系数Coefficient of variation,CV/%14.13遗传传递力Genetic trans mitting ability/%89.84杂种优势率Heterosis rate/%-10.16 137.29超高亲率Extremely high heterosis rate/%8.52 5.00 12.00 8.50 6.16 4.00~21.00 222.94 72.43-27.57 67.22 7.13 10.00 25.40 17.70 21.03 6.00~55.00 242.06 118.79 18.79 92.95 27.76 4.70 5.40 5.15 5.31 3.00~20.00 386.35 103.00 3.00 72.15 21.70 3.20 4.39 3.80 3.63 1.10~6.80 417.24 20.50-4.50 52.75 20.15
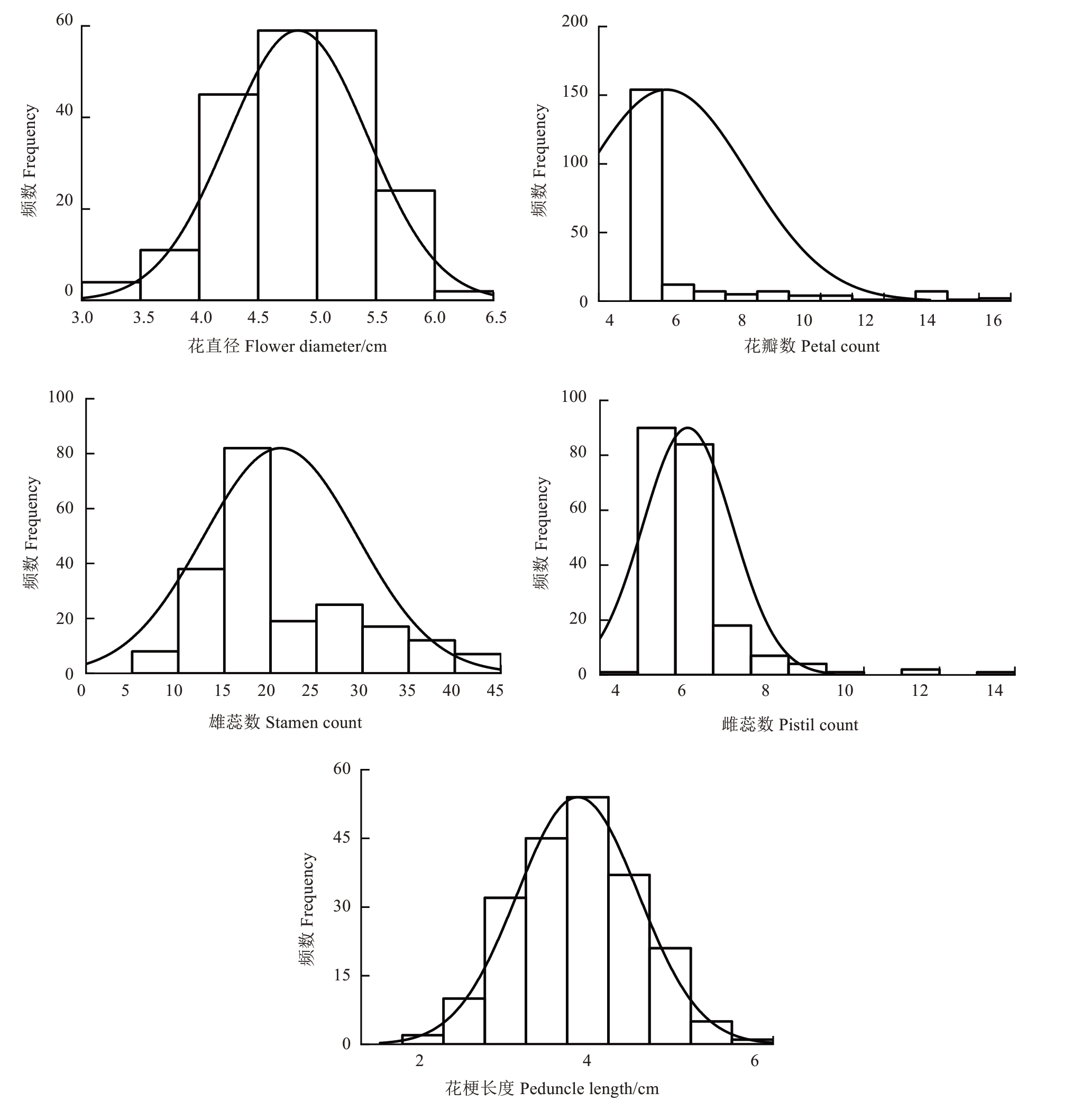
图1 红勋1 号和重瓣海棠F1代群体花数值型性状频率分布图
Fig. 1 Frequency distribution of quantitative traits in the F1 progenies of Hongxun No. 1×Double-Flowering Crabapple
2.1.2 花瓣数的遗传变异 红勋1 号和重瓣海棠的花瓣平均数分别为5.00 和12.00。子代花瓣数平均值为6.16,范围为4.00~21.00,变异系数为222.94%,表明花瓣数在子代中变异极为丰富,遗传传递力为72.43%,子代花瓣数受亲本影响程度相对适中。杂种优势率为-27.57%,负向杂种优势明显,子代花瓣数平均值远低于中亲值。广义遗传力为67.22%,表明遗传因素对花瓣数变异具有一定的影响。超高亲率为7.13%,表明F1杂交后代中仅有7.13%的子代个体花瓣数超过高亲值(表1)。
2.1.3 雄蕊数的遗传变异 亲本雄蕊平均数分别为10.00 和25.40。子代雄蕊平均数为21.03,范围在6.00~55.00,符合正态分布(图1),变异系数为242.06%,变异程度较大。 遗传传递力达到118.79%,杂种优势率为18.79%,F1群体雄蕊数在一定程度上高于中亲值,呈现出正向遗传传递趋势及正向杂种优势。广义遗传力为92.95%,表明遗传因素对雄蕊数变异的控制作用显著。超高亲率为27.76%,超半数子代个体雄蕊数超过高亲值(表1)。
2.1.4 雌蕊数的遗传变异 红勋1 号和重瓣海棠雌蕊平均数分别是4.70 和5.40,中亲值为5.15。子代雌蕊平均数为5.31,略高于中亲值。变幅为3.00~20.00,变异系数高达386.35%,是变异程度极高的性状。遗传传递力为103.00%,杂种优势率为3.00%,广义遗传力为72.15%,表明遗传因素对该性状变异有较大影响(表1)。
2.1.5 花梗长度的遗传变异 亲本花梗平均长度分别为3.20 cm 和4.39 cm,中亲值为3.80 cm。子代花梗平均长度为3.63 cm,低于中亲值。幅度为1.10~6.80 cm,符合正态分布(图1),变异系数为417.24%,变异程度极大。遗传传递力为20.50%,广义遗传力为52.75,遗传因素对花梗长度具有一定影响,但相较于其他性状的遗传贡献率略低。杂种优势率为-4.5%,呈现负向杂种优势(表1)。
2.2 杂交群体描述型性状遗传变异
如表2所示,亲本红勋1号和重瓣海棠花色性状分别为红和粉红,单重瓣性状分别为单瓣和重瓣。F1杂交后代花色性状分离极为明显,分离表现为白(4.46%)、粉红(50.50%)、红(33.66%)和紫红(11.38%);F1 杂交后代单瓣(82.57%)和重瓣(17.43%)。F1杂交后代中出现亲本中不存在的白色单瓣和紫红重瓣变异类型。
表2 红勋1 号和重瓣海棠F1杂交群体花描述型性状遗传变异
Table 2 The heritable variation of descriptive traits in the F1 progenies of Hongxun No. 1×Double-Flowering Crabapple

亲本Parent性状Traits杂交F1代性状分离比例Segregation ratio of traits in F1 hybrids/%白White红花色 Flower color单重瓣Single or doble flower红勋1号Hongxun No. 1红 Red单瓣Single flower重瓣海棠Double-Flowering Crabapple粉红 Pink重瓣Double flower 4.46粉红Pink 50.50 Red 33.66紫红Purple 11.38单瓣Single flower 82.57重瓣Double flower 17.43
2.3 花数值型性状遗传模式分析
为了探讨红勋1号与重瓣海棠F1杂交后代花部数值性状的遗传模式,分别对花直径、花瓣数、雄蕊数、雌蕊数和花梗长度5 个性状进行主基因+多基因混合模型分析。通过比较模型的最大似然函数值(MLV)和AIC 值,筛选出每个性状最优的3 个候选模型(表3)。结果表明,大多数性状的最优模型均为双主基因控制(2MG),并表现出加性效应(A)或加性-显性效应(AD),其中花瓣数、雄蕊数和雌蕊数的最佳模型均为2MG-AD,推测这些花器官数目性状可能受到相似的遗传机制调控。花直径和花梗长度的最优模型分别为2MG-EA 和2MG-AD,说明其主基因作用明显,且存在一定的等加性效应。
表3 花数值型性状候选遗传模型及其极大似然函数值和AIC 值
Table 3 Candidate genetic models of floral quantitative traits and their maximum log likelihood values and AIC values
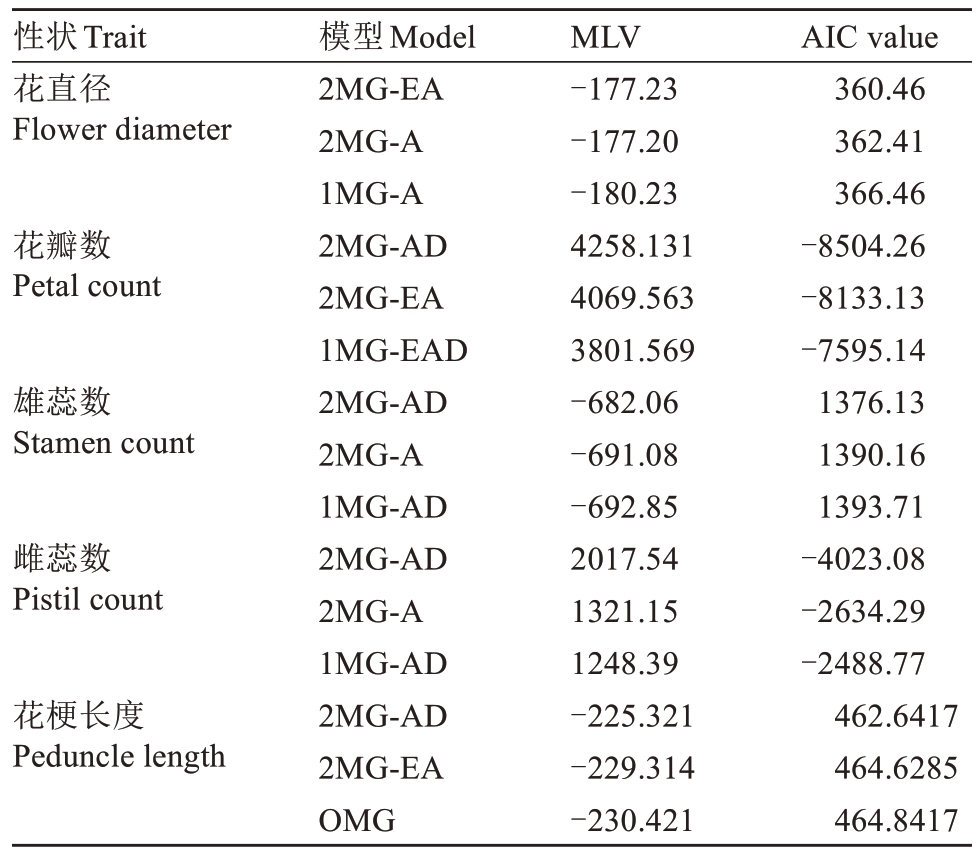
注:MLV. 极大似然函数值;MG. 主基因;EA. 等加性;A. 加性;AD. 加性-显性;EAD. 等加显性;OMG. 一对主基因。
Note:MLV. Log max likelihood Value; MG. Major gene; EA. Equal additive; A. Additive; AD. Additive-dominant; EAD. Equal additive dominant; OMG. One major gene.
AIC value 360.46 362.41 366.46-8504.26-8133.13-7595.14 1376.13 1390.16 1393.71-4023.08-2634.29-2488.77 462.6417 464.6285 464.8417性状Trait花直径Flower diameter花瓣数Petal count雄蕊数Stamen count雌蕊数Pistil count花梗长度Peduncle length模型Model 2MG-EA 2MG-A 1MG-A 2MG-AD 2MG-EA 1MG-EAD 2MG-AD 2MG-A 1MG-AD 2MG-AD 2MG-A 1MG-AD 2MG-AD 2MG-EA OMG MLV-177.23-177.20-180.23 4258.131 4069.563 3801.569-682.06-691.08-692.85 2017.54 1321.15 1248.39-225.321-229.314-230.421
2.4 候选模型的适合性检验
每个数值型性状的备选模型均通过均匀性检验(U12、U22、U32)、Smirnov 检验(nW2)和Kolmogorov检验(Dn),确定各性状的最佳遗传模型(表4)。结果表明,花直径、雄蕊数以及花梗长度3个模型通过最适性检验,结合候选模型AIC值,最终确定花直径性状最佳遗传模型为2MG-EA(2对等加性主基因),雄蕊数和花梗长度的最佳遗传模型为2MG-AD(2对加性-显性主基因)。花瓣数的3 个候选模型2MG-AD、2MG-EA 和1MG-EAD(1 对完全显性+加性-显性多基因)达到显著水平的个数分别为5、4和5,结合最小AIC 值,最终确定花瓣数最佳模型为2MG-AD;雌蕊数候选模型经过适合性检验后发现其最适模型为2MG-A(2对加性主基因模型)。
表4 花数值型性状候选模型最适性检验
Table 4 Adaptability test of candidate genetic models for floral quantitative traits

注:括号内为概率值。
Note:Data in brackets are probability.
性状Trait花直径Flower diameter Dn 0.026 2(0.999 0)0.021 6(0.993 0)0.030 0(0.992 8)0.532 7(0.000 0)0.650 7(0.000 0)0.496 9(0.000 0)0.025 7(0.999 2)0.056 1(0.530 2)0.035 1(0.959 8)0.209 7(0.000 0)0.287 9(0.000 0)0.169 6(0.000 0)0.023 9(0.999 8)0.032 4(0.981 8)0.029 9(0.992 5)U1 2 U2 2 U3 2花瓣数Petal count雄蕊数Stamen count雌蕊数Pistil count花梗长度Peduncle length模型Model 2MG-EA 2MG-A 1MG-A 2MG-AD 2MG-EA 1MG-EAD 2MG-AD 2MG-A 1MG-AD 2MG-AD 2MG-A 1MG-AD 2MG-AD 2MG-EA OMG 0.000 2(0.988 2)0.008 3(0.927 4)0.000 2(0.989 2)44.785 7(0.000 0)143.146 5(0.000 0)34.045 9(0.000 0)0.000 4(0.984 5)0.092 3(0.761 3)0.002 4(0.960 6)0.718 8(0.396 6)24.760 9(0.000 0)0.594 2(0.440 8)0.013 7(0.907 0)0.004 4(0.946 8)0.007 2(0.932 3)0.001 0(0.974 9)0.007 6(0.930 5)0.001 0(0.975 1)16.080 7(0.000 0)130.035 0(0.000 0)17.453 7(0.000 0)0.001 3(0.971 2)0.068 1(0.794 2)0.001 1(0.974 0)1.290 9(0.255 9)26.350 5(0.000 0)0.276 7(0.598 9)0.014 5(0.904 1)0.005 8(0.939 3)0.000 1(0.990 2)0.004 7(0.945 3)0.000 0(0.996 6)0.005 3(0.942 2)97.585 4(0.000 0)0.525 2(0.468 6)34.661 3(0.000 0)0.004 8(0.944 6)0.017 6(0.894 4)0.003 8(0.951 1)1.590 8(0.207 2)1.590 1(0.207 3)0.776 9(0.378 1)0.000 9(0.976 5)0.002 1(0.963 2)0.142 1(0.142 1)nW2 0.011 0(0.999 9)0.011 0(0.999 9)0.031 5(0.971 0)10.481 0(0.020 9)19.507 7(0.000 0)8.712 9(0.013 3)0.009 6(0.999 9)0.079 4(0.706 2)0.041 4(0.926 1)0.761 0(0.009)2.744 1(0.000 0)0.511 2(0.037 9)0.008 6(1.000 0)0.017 5(0.998 8)0.023 0(0.993 2)
2.5 最适模型遗传参数的评估
通过计算F1杂交组合各表型最优遗传模型以及对应的遗传参数可知(表5),花直径第1对主基因加性效应(da)为0.31,主基因遗传率(hmg2)为98.79%;花瓣数第1 对主基因加性效应为3.40,第2对主基因加性效应(db)为0.05,第1 对主基因显性效应(ha)为-2.13,第2 对主基因显性效应(hb)为0.08,表明第1 对主基因对花瓣数影响较大,且为负向遗传效应;雄蕊数主基因遗传率为92.00%,主基因方差(σmg2)为64.28,第1对和第2对主基因加性效应分别为9.72和4.98,第1对和第2对主基因的显性效应分别为-3.72 和-2.45,主基因的显性效应均为负值,表现出负向遗传效应;雌蕊数2 对主基因加性效应分别为0.03 和0.14;花梗长度主基因遗传率为24.34%,主基因方差为0.133,第1对和第2对主基因加性效应分别为0.54和0.45,2对主基因显性效应分别为0.72和0.11。
表5 花数值型性状的遗传参数评估
Table 5 Estimation of genetic parameters for floral quantitative traits
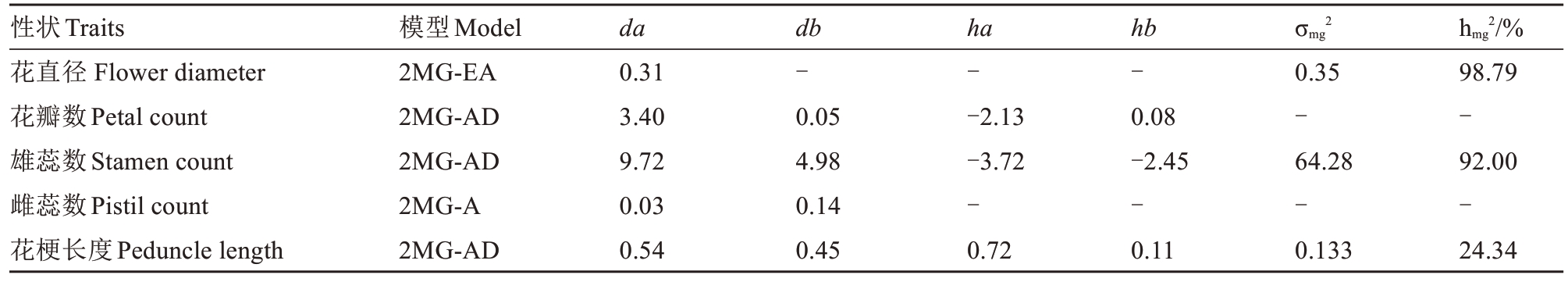
注:da、db. 主基因A 和B 的加性效应;ha、hb. 主基因A 和B 的显性效应;σmg2. 主基因方差;hmg2. 主基因遗传率。“-”表示该遗传效应在特定模型下未被检测到、未计算或无显著贡献。
Note:da, db. Additive effect of major gene A and B; ha, hb. Dominant effect of major gene A and B; σmg2. Major gene variance; hmg2. Heritability of major gene.“ -” indicates that the genetic effect was not detected, calculated or made no significant contribution under a specific model.
性状Traits花直径 Flower diameter花瓣数Petal count雄蕊数Stamen count雌蕊数Pistil count花梗长度Peduncle length hmg2/%98.79-92.00-24.34模型Model 2MG-EA 2MG-AD 2MG-AD 2MG-A 2MG-AD da 0.31 3.40 9.72 0.03 0.54 db-0.05 4.98 0.14 0.45 ha--2.13-3.72-0.72 hb-0.08-2.45-0.11 σmg2 0.35-64.28-0.133
3 讨 论
目前,中国观赏海棠选育以杂交育种为主,杂交育种是种质创新及新品种选育的主要途径[7,15]。笔者围绕红勋1号和重瓣海棠杂交F1代的花部性状进行了系统的遗传变异分析,对F1代204 个株系的5个数值型性状和2 个描述型性状进行表型鉴定,后代表现出丰富的遗传多样性。杂交后代在描述型性状花色及单重瓣上性状分离明显,出现了白色单瓣、红色重瓣、紫红重瓣等变异单株,具有选育优异观赏种质的潜力。
观赏海棠属于高度杂合的树种,遗传背景复杂,通常在杂交F1代表现出性状分离,与其他纯系亲本F1代(不分离世代)不同[18-19]。因此,利用数值型性状主基因+多基因混合遗传体系中单一分离世代类型进行遗传模型预测[15]。5 个数值型性状在F1代中均表现出不同程度的变异,其中花直径的变异系数最小,为14.13%,且表现出较高的遗传传递力和广义遗传力,表明其受主基因控制显著,遗传基础较稳定。花直径的最适遗传模型为2MG-EA(2对等加性主基因),主基因加性效应为正,主基因遗传率高达98.79%。因此,适合通过常规选择育种快速固定优良性状。
尽管花瓣数遗传传递力和广义遗传力较高,但杂种优势率为-27.57%,F1代中多数个体的花瓣数低于中亲值,重瓣表型不稳定。同时,最适性检验分析表明花瓣数的最适模型为2MG-AD(2 对加性-显性主基因),其中第1 对主基因具有显著的负向显性效应(-2.13),显示该主基因在杂交后代中抑制重瓣形成,这是造成大量单瓣个体出现的主要遗传因素,崔佳璇[20]和聂超仁[21]的研究结果也表明单瓣与重瓣海棠的杂交后代更倾向于单瓣。
雄蕊和雌蕊数在F1代中均表现出较大变异,具有一定的遗传改良潜力。雄蕊数表现出明显的正向杂种优势,具有较高的超高亲率、遗传传递力和广义遗传力,最优遗传模型为2MG-AD。加性效应为正,显性效应为负(分别为-3.72 和-2.45),主基因遗传率高达92.00%,表明其遗传基础稳定,加性遗传控制强。尽管显性基因对雄蕊数有一定抑制作用,但加性基因仍是主要的遗传驱动力。雄蕊数受显性基因控制,并起到抑制作用,具有较大的遗传潜力,是育种中可重点选择的目标性状。
雌蕊数变异系数、遗传传递力和广义遗传力较高,最优模型为2MG-A(2对加性主基因模型),加性效应主导遗传表达,具备一定选择潜力。雌蕊数作为花部结构功能性状之一[22],其显著变异对后续花果发育和繁殖能力可能具有一定影响,在育种中需结合综合表现进行筛选。
相比之下,花梗长度尽管符合正态分布,但其变异系数高达417.24%,是所有性状中变异程度最大的,表明其在环境响应上具有较强的可塑性。遗传传递力仅为20.50%,广义遗传力为52.75%,表明环境对其影响显著。F1代平均值略低于中亲值,表现出负向杂种优势(-4.50%),主基因遗传率也较低,为24.34%。其最优模型为2MG-AD,但主基因的加性与显性效应相对较弱。综合来看,花梗长度虽有一定的遗传基础,但育种进展依赖于多代选择及环境条件优化,不宜作为短期快速改良的主要性状。
4 结 论
通过对红勋1 号×重瓣海棠F1群体的花器官进行研究,探明了观赏海棠花器官表型的遗传变异与分离规律。花直径和雄蕊数表现出较高的遗传力和稳定的主基因控制能力,是优良性状选择与固定的优先目标;花瓣数具有主基因与显性抑制效应共同作用的复杂遗传机制,在重瓣品种育种中需选择均为重瓣型的品种作为亲本;雌蕊数虽变异显著,但受主基因加性效应调控,具备一定的遗传改良潜力;而花梗长度遗传稳定性和可选择性较差,在育种应用中应结合表型与环境响应综合评估。
[1] DA SILVA J A T,GULYÁS A,MAGYAR-TÁBORI K,WANG M R,WANG Q C,DOBRÁNSZKI J. In vitro tissue culture of apple and other Malus species:Recent advances and applications[J].Planta,2019,249(4):975-1006.
[2] 杨晓倩,张往祥,张龙,刘爱林,周婷,熊青青. ‘小甜甜’等7个观果海棠半同胞家系表型多样性及子代优选[J/OL]. 南京林业大学学报(自然科学版),2025:1-13. (2025-05-18). https://link.cnki.net/urlid/32.1161.S.20250517.1445.002.YANG Xiaoqian,ZHANG Wangxiang,ZHANG Long,LIU Ailin,ZHOU Ting,XIONG Qingqing. Phenotypic diversity and selection of superior strains from 7 Half-sib familys of Malus including ‘Sweet Sugartyme’[J/OL]. Journal of Nanjing Forestry University (Natural Sciences Edition),2025:1-13. (2025-05-18).https://link.cnki.net/urlid/32.1161.S.20250517.1445.002.
[3] 耿一然,高寿利,孙纪霞,张英杰,张京伟,潘香君. 花果兼用海棠的育种研究进展[J]. 烟台果树,2022(2):1-4.GENG Yiran,GAO Shouli,SUN Jixia,ZHANG Yingjie,ZHANG Jingwei,PAN Xiangjun. Research progress on breeding of ornamental and fruit-bearing crabapples[J]. Yantai Fruits,2022(2):1-4.
[4] 彭兰兰,彭飞,彭俊. 红叶海棠在园林绿化中的应用:以精河县为例[J]. 新疆农垦科技,2021,44(6):33-34.PENG Lanlan,PENG Fei,PENG Jun. Application of red-leaf begonia in landscape design:A case study of Jinghe county[J]. Xinjiang Farm Research of Science and Technology,2021,44(6):33-34.
[5] 张往祥,魏宏亮,江志华,曹福亮,汤庚国. 观赏海棠品种群的花期物候特征研究[J]. 园艺学报,2014,41(4):713-725.ZHANG Wangxiang,WEI Hongliang,JIANG Zhihua,CAO Fuliang,TANG Gengguo. Studies on flowering phenological characteristics of ornamental crabapple cultivar group[J]. Acta Horticulturae Sinica,2014,41(4):713-725.
[6] 葛新新,吴静妍,姚强,窦树军,李海英. 观赏海棠花色及叶色研究进展[J/OL]. 分子植物育种,2025:1-9. (2025-04-16). https://link.cnki.net/urlid/46.1068.S.20250416.1319.002.GE Xinxin,WU Jingyan,YAO Qiang,DOU Shujun,LI Haiying.Research progress on flower color and leaf color of ornamental crabapple[J/OL]. Molecular Plant Breeding,2025:1-9. (2025-04-16). https://link.cnki.net/urlid/46.1068.S.20250416.1319.002.
[7] 李红莲,张冰冰,梁英海,赵晨辉,王珊珊,宋宏伟. 中国近30年苹果育种亲本选择与选配分析[J]. 分子植物育种,2020,18(21):7155-7161.LI Honglian,ZHANG Bingbing,LIANG Yinghai,ZHAO Chenhui,WANG Shanshan,SONG Hongwei. Analysis of apple breeding parents selection and crosses in recent 30 years of China[J]. Molecular Plant Breeding,2020,18(21):7155-7161.
[8] 张琳,郭丽丽,郭大龙,侯小改. 牡丹杂交F1 代性状分离规律及混合遗传分析[J]. 南京林业大学学报(自然科学版),2018,42(6):51-60.ZHANG Lin,GUO Lili,GUO Dalong,HOU Xiaogai. Separation analysis and mixed genetic analysis of phenotypic traits in F1 progenies of tree peony[J]. Journal of Nanjing Forestry University (Natural Sciences Edition),2018,42(6):51-60.
[9] 王欢,孔滢,郎利新,包放,窦晓莹,白锦荣,尚宏忠. 亚洲百合与大花卷丹杂种F1 重要性状的遗传分析[J]. 华北农学报,2017,32(4):114-121.WANG Huan,KONG Ying,LANG Lixin,BAO Fang,DOU Xiaoying,BAI Jinrong,SHANG Hongzhong. Genetic analysis of important characters in F1 hybrids of Lilium Asiatic hybrids and L. leichtlinii var. maximowiczii[J]. Acta Agriculturae Boreali-Sinica,2017,32(4):114-121.
[10] 周利君,于超,常笑,万会花,罗乐,潘会堂,张启翔. 月季F1 代群体表型性状变异分析[J]. 植物研究,2019,39(1):131-138.ZHOU Lijun,YU Chao,CHANG Xiao,WAN Huihua,LUO Le,PAN Huitang,ZHANG Qixiang. Variation analysis of phenotypic traits in F1 population of Rosa spp.[J]. Bulletin of Botanical Research,2019,39(1):131-138.
[11] 韩婷婷,杨天资,赵培磊,祝军,张玉刚,孙晓红. ‘金冠’和‘红勋1 号’杂交后代遗传多样性分析[J]. 青岛农业大学学报(自然科学版),2021,38(1):1-6.HAN Tingting,YANG Tianzi,ZHAO Peilei,ZHU Jun,ZHANG Yugang,SUN Xiaohong. Analysis of genetic diversity of the hybrid populations of ‘Golden Delicious’ × ‘Hongxun No. 1’[J].Journal of Qingdao Agricultural University (Natural Science),2021,38(1):1-6.
[12] 聂继云,吕德国,李静,刘凤之,李海飞,王昆. 22 种苹果种质资源果实类黄酮分析[J]. 中国农业科学,2010,43(21):4455-4462.NIE Jiyun,LÜ Deguo,LI Jing,LIU Fengzhi,LI Haifei,WANG Kun. A preliminary study on the flavonoids in fruits of 22 apple germplasm resources[J]. Scientia Agricultura Sinica,2010,43(21):4455-4462.
[13] 王昆,刘凤之,曹玉芬. 苹果种质资源描述规范和数据标准[M].北京:中国农业出版社,2005.WANG Kun,LIU Fengzhi,CAO Yufen. Descriptors and data standard for apple[M]. Beijing:China Agriculture Press,2005.
[14] 郭淑青,宋慧,杨清华,高金锋,高小丽,冯佰利,杨璞. 谷子株高及穗部性状主基因+多基因混合遗传模型分析[J]. 中国农业科学,2021,54(24):5177-5193.GUO Shuqing,SONG Hui,YANG Qinghua,GAO Jinfeng,GAO Xiaoli,FENG Baili,YANG Pu. Analyzing genetic effects for plant height and panicle traits by means of the mixed inheritance model of major gene plus polygene in foxtail millet[J].Scientia Agricultura Sinica,2021,54(24):5177-5193.
[15] 潘静,孟志浩,王森,王海波,何平,常源升,郑文燕,李林光,王琛,王平,何晓文. 苹果‘金冠’和‘长富2 号’正反交F1 代果实品质性状多样性及综合评价[J]. 中国农业科学,2024,57(24):4945-4963.PAN Jing,MENG Zhihao,WANG Sen,WANG Haibo,HE Ping,CHANG Yuansheng,ZHENG Wenyan,LI Linguang,WANG Chen,WANG Ping,HE Xiaowen. Diversity analysis and comprehensive evaluation of fruit quality traits in reciprocal cross progenies of apple Golden Delicious and Fuji Nagafu No.2[J]. Scientia Agricultura Sinica,2024,57(24):4945-4963.
[16] 叶雪莲,陈靖雯,姚祥坦,权新华,黄鹂. 不结球白菜叶片皱缩性状的遗传分析[J]. 中国农业科学,2024,57(18):3684-3694.YE Xuelian,CHEN Jingwen,YAO Xiangtan,QUAN Xinhua,HUANG Li. Genetic analysis of leaf wrinkling traits in nonheading Chinese cabbage[J]. Scientia Agricultura Sinica,2024,57(18):3684-3694.
[17] 王靖天,张亚雯,杜应雯,任文龙,李宏福,孙文献,葛超,章元明. 数量性状主基因+多基因混合遗传分析R 软件包SEA v2.0[J]. 作物学报,2022,48(6):1416-1424.WANG Jingtian,ZHANG Yawen,DU Yingwen,REN Wenlong,LI Hongfu,SUN Wenxian,GE Chao,ZHANG Yuanming. SEA v2.0:An R software package for mixed major genes plus polygenes inheritance analysis of quantitative traits[J]. Acta Agronomica Sinica,2022,48(6):1416-1424.
[18] 张巍瀚. 苹果群体遗传多样性和品质驯化与改良的基因组学研究[D]. 武汉:华中农业大学,2023.ZHANG Weihan. Genomics analyses on the population diversity,quality domestication and improvement in apple[D]. Wuhan:Huazhong Agricultural University,2023.
[19] 段乃彬,马玉敏,王昆,王效睦,谢坤,白静,杨永义,蒲艳艳,宫永超. 山东省主要栽培苹果基因组重测序及SNP 芯片位点挖掘[J]. 分子植物育种,2021,19(21):7055-7066.DUAN Naibin,MA Yumin,WANG Kun,WANG Xiaomu,XIE Kun,BAI Jing,YANG Yongyi,PU Yanyan,GONG Yongchao.SNP chip loci mining and genome resequencing of main cultivated apple varieties in Shandong Province[J]. Molecular Plant Breeding,2021,19(21):7055-7066.
[20] 崔佳璇. ‘红宝石’海棠杂交子代遗传多样性评价及优良单株选择[D]. 秦皇岛:河北科技师范学院,2023.CUI Jiaxuan. Evaluation of genetic diversity and excellent single plant selection of Malus ‘Red Jewel’ hybrid offspring[D]. Qinhuangdao:Hebei Normal University of Science & Technology,2023.
[21] 聂超仁. 樱花起源进化和重瓣花发育分子机制研究[D]. 北京:北京林业大学,2023.NIE Chaoren. Study on the origin and evolution of flowering cherry and the molecular mechanism of the development of double flowers[D]. Beijing:Beijing Forestry University,2023.
[22] RUIZ H M,NORTH H M. Correlation between number and position of floral organs in Arabidopsis thaliana[J]. Journal of Experimental Botany,2021,62:1259-1270.