柑橘是世界第一大水果,也是中国栽培面积最大、产量最多的经济果树[1]。柚类(Citrus grandis)为柑橘3个基本种之一,是其重要的组成部分[2]。中国柚类的种植面积和产量也同样居世界首位,尤其以沙田柚、三红蜜柚、东试早柚、贡水白柚等我国自主选育、地方改良品种为主[3-4]。近些年来,随着全国各地柑橘柚类野生种、半野生种和栽培种资源的调查及收集工作不断推进,发现柚类品种不仅果皮颜色呈现多种多样,果肉颜色也同样如此[5],如红肉的泰国红宝石柚、越南青柚、三红蜜柚,黄肉的琯溪蜜柚和凤凰柚,绿肉的翡翠柚等。其中,大部分红肉柚品种因果肉中积累了大量的番茄红素或β-胡萝卜素,而呈现红色或浅红色[6]。在鲜果市场上,果肉呈红色的品种更受消费者青睐[7]。红肉柚果实不仅在品质和口味上符合消费者的需求,还富含类黄酮、类胡萝卜素等成分,其具有软化血管、消除体内自由基、抗氧化及抗癌等功能[8]。
为了综合评价植物的内在品质及挖掘功能性成分,往往需要对糖、有机酸、氨基酸、核苷酸、维生素等初生代谢物和黄酮、香豆素、生物碱、酚酸、萜类等次生代谢物的成分及含量进行全面评价[9]。与传统方法相比,采用代谢组学对食用和药用植物的活性物质进行高通量分析,具有耗时短、准确性高、检测更为全面等优点[10-11]。利用液质联用LC-MS[12](Liquid Chromatograph-Mass Spectrometer)、气质联用GC-MS[13](Gas Chromatography-Mass Spectrometry)和核磁共振NMR[14](Nuclear Magnetic Resonance)平台,对植物样品中大量的代谢物进行检测。通过对不同品系、生长时期以及处理方式的植物样本进行多元统计分析,筛选出差异代谢物或者标志物,是代谢组学在植物学领域研究中最常规的方法,广泛应用于种质鉴定、产品开发、遗传改良等研究领域[15]。明家琪[16]通过气相色谱(GC)分析表明,武汉地区HB柚、Chandler红心柚和华农红柚品种汁胞中的果糖和奎宁酸含量显著高于重庆地区红肉文旦柚、Chandler 红心柚和五布红心柚的同质含量。陈婉冰等[17]利用LC-MS和GC-MS进行代谢组学分析,结果表明化州柚正毛、副毛和光青3 个品系的花朵在活性成分上存在显著差异;地奥司明、芸香柚皮苷、芦丁等多种有效成分在副毛和正毛花朵中的含量高于光青。
泰国红宝石柚源自泰国南部洛坤府市巴帕南县,果实成熟时果肉颜色呈似红宝石,故名泰国红宝石柚,是目前中国南方地区主要的柑橘栽培品种之一。该品种树势强,花较大,具有周年开花的特性,初花期为2 月下旬,盛花期为3 月上旬至3 月中旬。果实早熟,以当年2月开花坐果为主,10月上旬进入成熟期,最早可于9 月下旬上市。果实梨形,表面茸毛长而密,果皮易于剥离;果肉红色,无核,化渣性优,有香味,耐储运。在热带、亚热带地区,袋装苗移栽后通常第3 年开始挂果,第5 年进入盛果期,单株产量高达100 kg。目前,泰国红宝石柚以鲜食为主,在果实深加工和功能成分研究方面鲜有报道。因此,笔者以6 个柚类品种作为研究对象,运用代谢组学方法筛选特征代谢物,为泰国红宝石柚果实的功能性成分开发及综合利用提供一定的理论基础。
1 材料和方法
1.1 试验材料
泰国红宝石柚、越南青柚、红玉香柚、三红蜜柚、星露比葡萄柚、暹罗红柚果实均来源于云南省农业科学院热带亚热带经济作物研究所瑞丽柠檬综合试验站柑橘种质资源圃。2024 年11 月20 日采集资源圃中处于成熟期的6 个红肉柚类品种果实样品(图1)。每个品种选择树龄相同、长势一致的挂果树3株,每株分别从东南西北中5 个方位随机采摘成熟度一致、大小均匀、无病虫害的果实1个,作为1次重复。削取果肉,立即置于液氮下速冻后充分混匀,封袋标记,于-80 ℃冰箱保存备用。
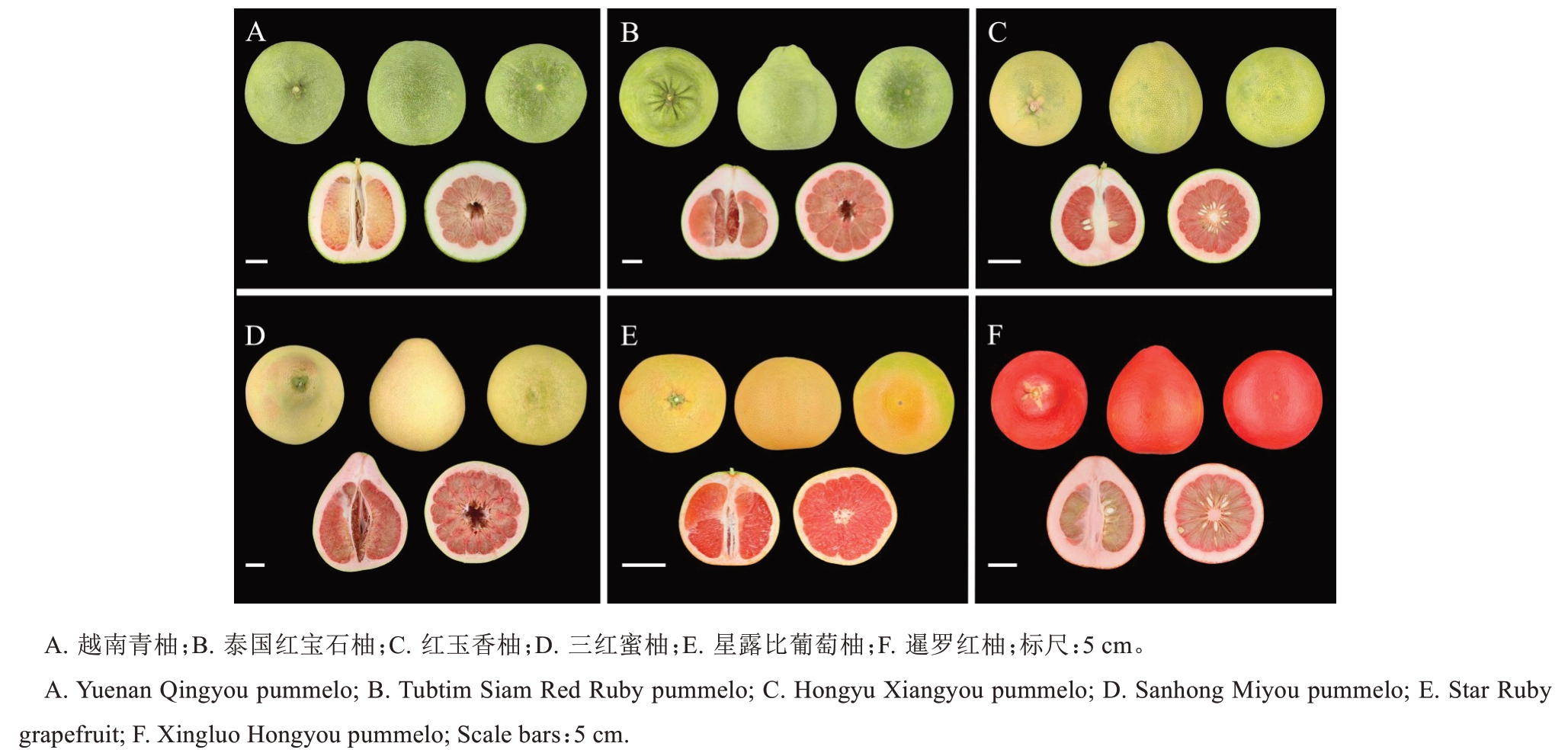
图1 不同红肉柚品种果实表型特征
Fig.1 Fruit phenotypic characteristics of different red-flesh pummelo cultivars
1.2 试剂与仪器
内标2-氯苯丙氨酸购自上海麦克林生化科技股份有限公司;甲酸铵、乙酸铵、甲酸、甲醇、异丙醇、乙腈和氨水购自上海安谱实验科技股份有限公司。
Vanquish 超高效液相色谱仪和Orbitrap Exploris 120 高分辨质谱仪,美国赛默飞世尔科技有限公司生产;JS002 高速离心机,德国Sigma 实验室离心机股份有限公司生产;AS 60-220.R2天平,瑞得威科技有限公司生产;Tiss-L 研磨仪,上海净信科技有限公司生产;DY-100ST 超声仪,深圳市聚力环保科技有限公司生产。
1.3 试验方法
1.3.1 UPLC-MS/MS 样品前处理 将样品转移到2 mL EP 管中,加入两粒钢珠,液氮中冷冻处理至少15 min。研磨机96 孔板预先放入液氮中冷冻处理,60 Hz研磨2 min。取100 mg植物样品,加入1000 μL提取液(70%甲醇,含2-氯苯丙氨酸内标0.5 μg·mL-1),涡旋混匀30 s,超声30 min,-20 ℃静置30 min,4 ℃,12 000 r·min-1离心15 min,0.22 μm 滤膜过滤,取上清液于进样瓶中,冻存于-80 ℃冰箱用于检测。另外,每个样品吸取等量上清液,混合均匀为QC样品,上机检测。
1.3.2 UPLC-MS/MS 条件 液相色谱采用Thermo hypersil gold C18(2.1 mm×100 mm,1.9 μm)色谱柱,流动相A 相为5 mmol·L-1乙酸铵水溶液,B 相为乙腈,梯度洗脱:0 min,95% A,5% B;10 min,5% A,95% B;12.1 min,95% A,5% B,保持2.9 min。流速为0.3 mL·min-1,进样体积为2 μL,样品盘温度为10 ℃,柱温为40 ℃。使用MSDIAL 软件的DDA 模式进行质谱分析。质谱采用ESI离子源,参数如下:离子化电压+3400/-3200 V,鞘气流速320 Arb,辅助气体流速40 Arb,温度(TEM)350 ℃。样品随机进样,周期性地插入QC样品,进行质量控制和矫正峰的漂移。
1.3.3 数据处理及分析 利用UPLC-MS/MS 采集的高通量代谢组学原始数据,先使用Analvsis Base Fileconverter 工具将质谱原始数据转成abf 格式,再使用MSDIAL 软件进行数据预处理,具体包括噪音滤除、背景扣除、保留时间、质荷比的校正、峰识别、峰对齐、平滑处理等;然后利用小分子代谢物质谱二级碎片数据库mzCloud、MassBanK、METLIN 等进行搜索匹配,以及查找文献。根据代谢物的裂解规律结合二级质谱信息进行定性定量分析,得到代谢物鉴定信息。
将采集的数据UV(unit variance scaling)标准化处理后进行PCA(principal component analysis)分析;同时,将数据进行Log 转换和Mean-Centered 尺度化处理,再利用R 包Meta boAnalyst 进行OPLSDA 分析,得到变量投影重要性分析值(VIP)、差异倍数(fold change,FC)以及Student’s t-test 的P-value 综合筛选差异代谢物[18];利用TBtools-Ⅱ和R 包ClusterProfiler对代谢物数据进行聚类分析和KEGG(kyoto encyclopedia of genes and genomes)富集分析[19]。
2 结果与分析
2.1 UPLC-MS/MS非靶向数据PCA分析
基于UPLC-MS/MS 检测平台和自建数据库,从泰国红宝石柚、越南青柚、红玉香柚、三红蜜柚、星露比葡萄柚、暹罗红柚6 组共18 个样品中检测到1019种代谢物。其中脂质和类脂质分子246 种,占比24.14%;苯丙酮和多酮类化合物215 种,占比21.10%;有机酸及其衍生物127 种,占比12.46%;有机杂环化合物119种,占比11.68%;有机含氧化合物97种,占比9.52%。此外,还检测到有机含氮化合物、核苷及其类似物、香豆素和木质素、苯类化合物、生物碱及其衍生物等(表1)。为探究6个柚类品种果肉间代谢物的整体差异,对以上检测到的代谢物进行PCA 分析。如图2 所示,第一主成分PC1、第二主成分PC2分别包含了原来信息量的23.33%和19.02%。QC 样本密集分布,表明代谢数据稳定、质量高。在PC1方向上,泰国红宝石柚、暹罗红柚和星露比葡萄柚与其他3个品种明显分离,越南青柚、三红蜜柚、红玉香柚分布较集中,泰国红宝石柚、暹罗红柚、越南青柚、三红蜜柚、红玉香柚位于正轴,而星露比葡萄柚位于负轴;在PC2 方向上,泰国红宝石柚、暹罗红柚和星露比葡萄柚也与其他3个品种明显分离。
表1 UPLC-MS/MS 非靶向代谢组学检测的代谢物种类和数量
Table 1 The classification and quantity of metabolites detected by untargeted metabolomics using UPLC-MS/MS

种类Classification生物碱及其衍生物Alkaloids and derivatives苯类化合物Benzenoids香豆素和木质素Coumarins and lignans黄酮类化合物Flavonoids脂质和类脂质分子Lipids and lipid-like molecules核苷、核苷酸类似物Nucleosides, nucleotides analogues有机酸及其衍生物Organic acids and derivatives有机含氮化合物Organic nitrogen compounds有机含氧化合物Organic oxygen compounds有机杂环化合物Organoheterocyclic compounds苯丙酮和多酮类化合物Phenylpropanoids and polyketides其他Others数量Quantity 12占比Proportion/%1.18 84 17 8.24 1.67 12 246 1.18 24.14 25 2.45 127 12.46 26 2.55 97 9.52 119 11.68 215 21.10 39 3.83
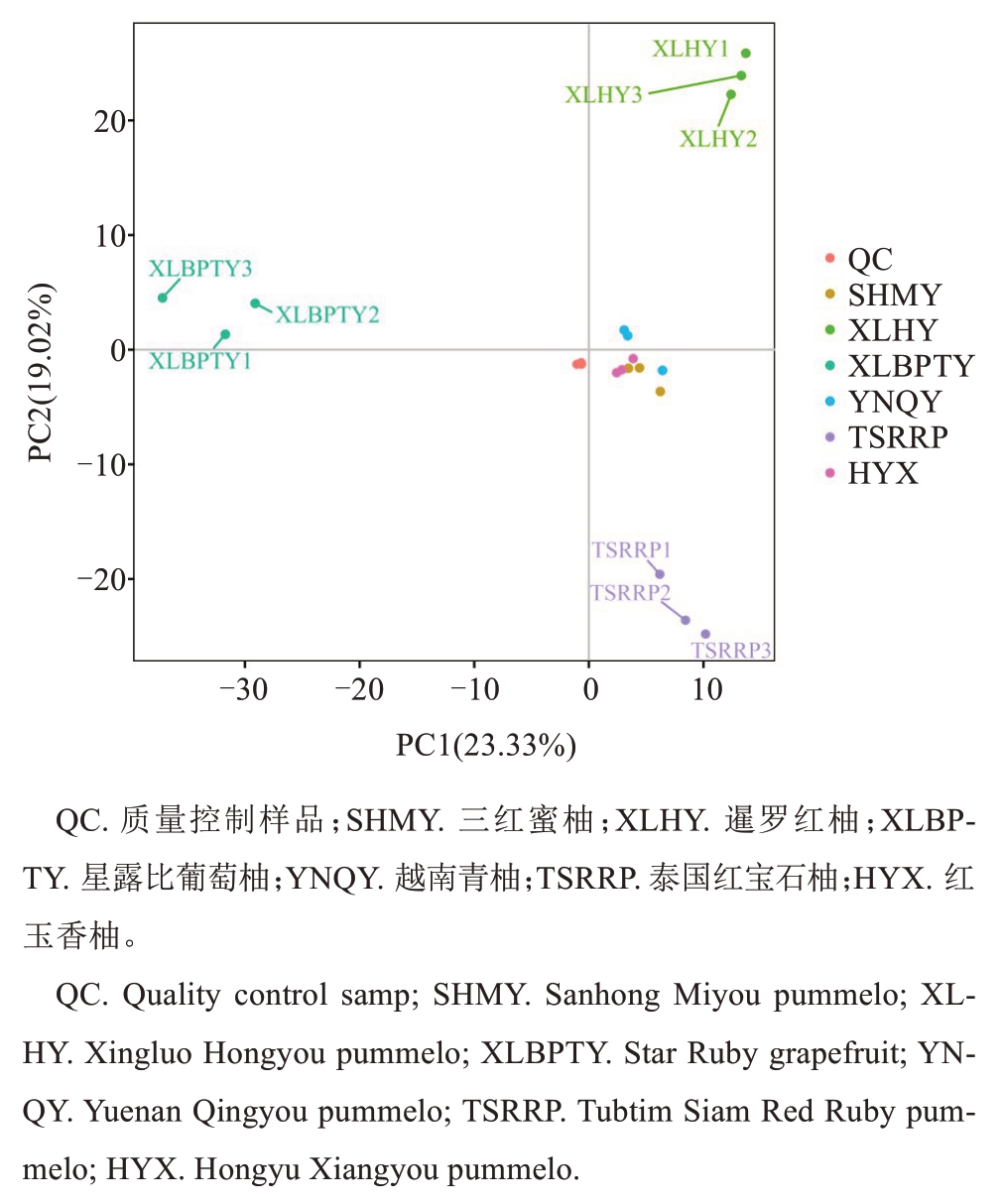
图2 UPLC-MS/MS 检测的代谢物主成分分析
Fig. 2 Principal component analysis (PCA) of metabolites detected by UPLC-MS/MS
2.2 泰国红宝石柚果肉代谢数据OPLS-DA分析
OPLS-DA 评价模型的预测参数为R2X、R2Y 和Q2,其中R2X和R2Y分别表示所建模型对XY矩阵的解释率,Q2 表示模型的预测能力,这三个指标越接近1 时,表明模型越稳定可靠。Q2>0.5 时被认为是有效的模型,Q2>0.9 时为出色的模型。经过200 次随机排列组合试验(图3-A),5个比较组合泰国红宝石柚vs越南青柚(TSRRP vs YNQY)、泰国红宝石柚vs红玉香柚(TSRRP vs HYX)、泰国红宝石柚vs 三红蜜柚(TSRRP vs SHMY)、泰国红宝石柚vs星露比葡萄柚(TSRRP vs XLBPTY)、泰国红宝石柚vs暹罗红柚(TSRRP vs XLHY,后同)的OPLS-DA 模型中R2X、R2Y、Q2值均大于0.5,且Q2>0.9,表明所建模型具有较强的预测能力。以OPLS-DA 模型中变量投影重要性分析值VIP≥1 作为标准筛选差异代谢物,对分析结果用S-plots进行可视化分析,横坐标p[1]为负载向量在主成分1 上的协方差,纵坐标p(corr)[1]为负载向量与主成分1 的相关性。其中TSRRP vs YNQY 筛选出461 种代谢物(图3-B),TSRRP vsHYX为448种,TSRRP vs SHMY为417种,TSRRP vs XLBPTY为504种,TSRRP vs XLHY为513种。
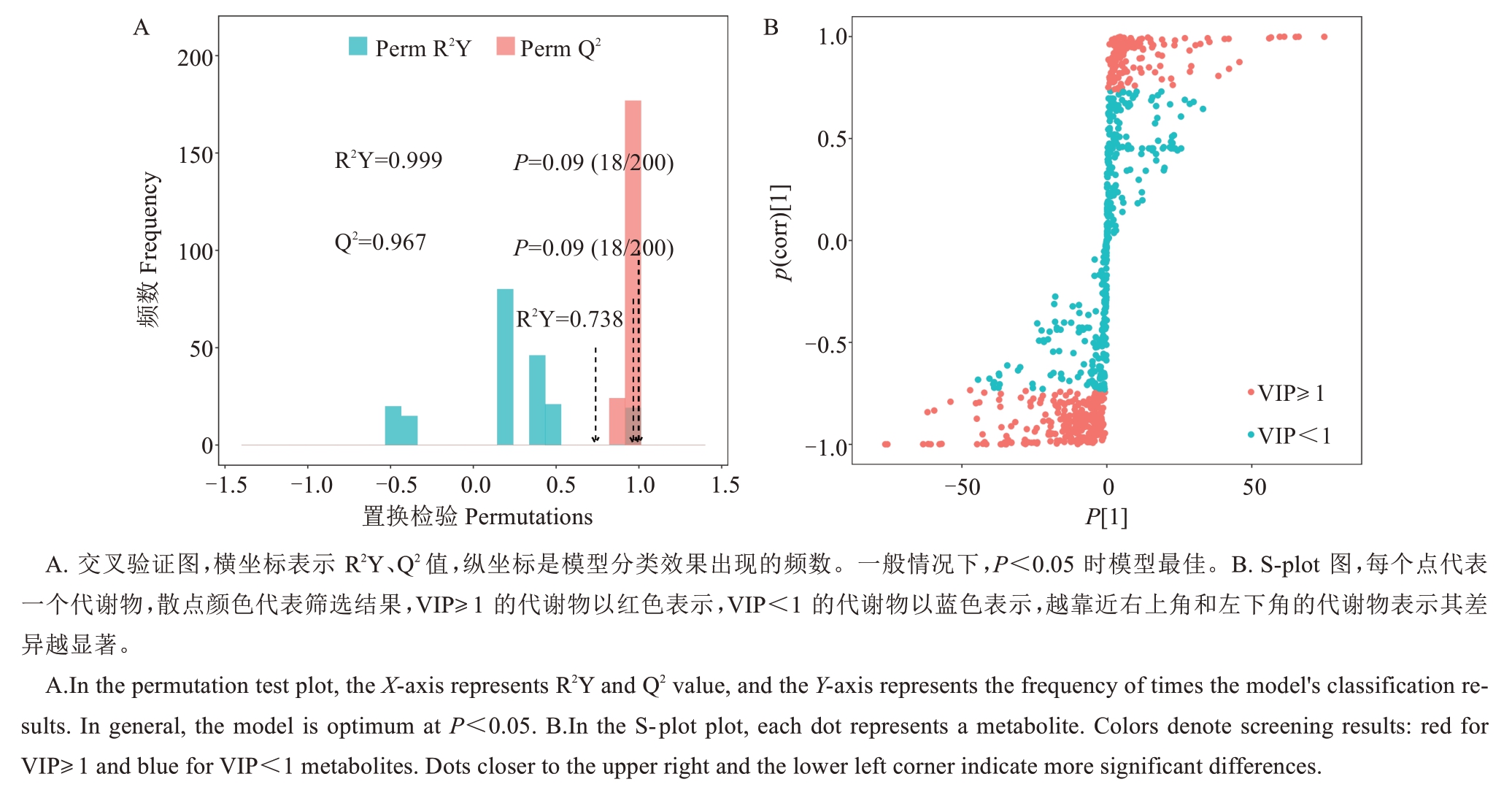
图3 泰国红宝石柚 vs 越南青柚果肉代谢成分OPLS-DA 分析的模型交叉验证结果(A)和S-plot 图(B)
Fig. 3 The results of permutation test(A) and S-plot(B) for OPLS-DA analysis of Tubtim Siam Red Ruby pummelo vs Yuenan Qingyou pummelo fruit pulp metabolites
2.3 泰国红宝石柚果肉差异代谢物筛选
以VIP≥1 为筛选标准获得大量差异代谢物,为进一步筛选不同品种间的标志性代谢物,以FC>2或者FC<0.5、P<0.05 标准执行,筛选结果以Log2FoldChange和-Log10 P-value分别为横纵坐标绘制火山图,对数据进行可视化分析。从TSRRP vs YNQY 中筛选出213 种代谢物,其中138 种上调,75种下调(图4);从TSRRP vs HYX 中筛选出237 种代谢物,其中165 种上调,72 种下调;从TSRRP vs SHMY 中筛选出264 种代谢物,其中196 种上调,68种下调;从TSRRP vs XLBPTY 中筛选出386 种代谢物,其中193 种上调,193 种下调;从TSRRP vs XLHY 中筛选出390 种代谢物,其中259 种上调,131 种下调。以上结果表明,泰国红宝石柚差异代谢物上调的种类数目远高于下调。根据各分组鉴定到的606种差异代谢物绘制upset图(图5),在5个比较组合中同时具有差异的代谢物有55 种。其中,在TSRRP vs XLHY 中具有差异的代谢物有76 种,TSRRP vs XLBPTY 有74 种,TSRRP vs SHMY 有28种。在TSRRP vs YNQY 中具有差异的代谢物有9种,分别为曲克芦丁、槲皮素-3-芸香糖苷-7-葡萄糖苷、紫草素衍生物、槲皮素-3-O-葡萄糖苷衍生物、野漆树苷、黄芩苷衍生物、非美诺司他、脱镁叶绿酸a甲酯、西伯利亚远志糖A5。在TSRRP vs HYX 中具有差异的代谢物有6种,分别为N-三甲基赖氨酸、十六酰胺乙醇、2’,4’-二羟基-6’-甲氧基苯乙酮、升麻素-β-D-葡萄糖苷、溶血磷脂酰胆碱、阿糖腺苷一水合物。
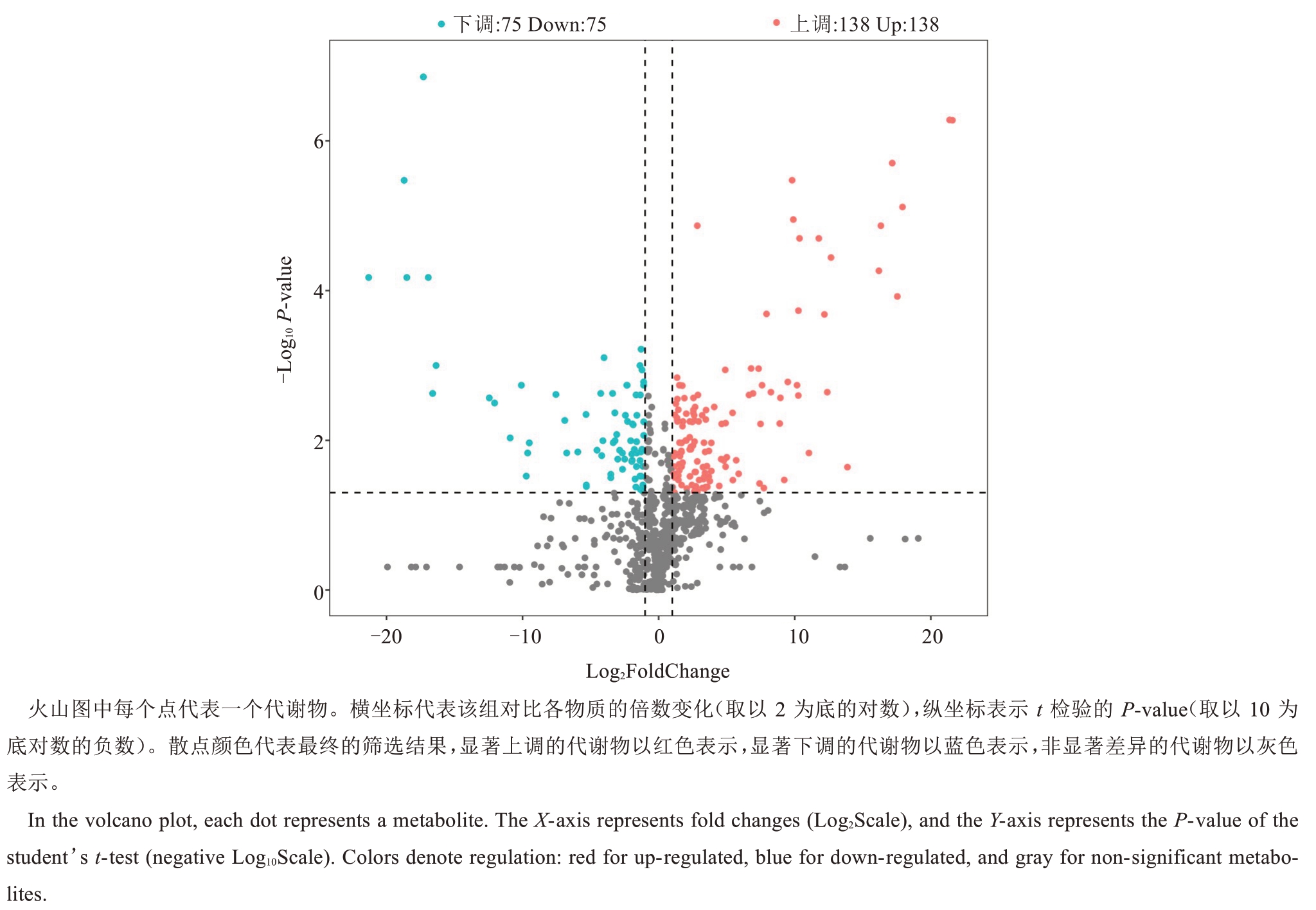
图4 泰国红宝石柚 vs 越南青柚果肉代谢物火山图
Fig. 4 Volcano plot of Tubtim Siam Red Ruby pummelo vs Yuenan Qingyou pummelo fruit pulp metabolites
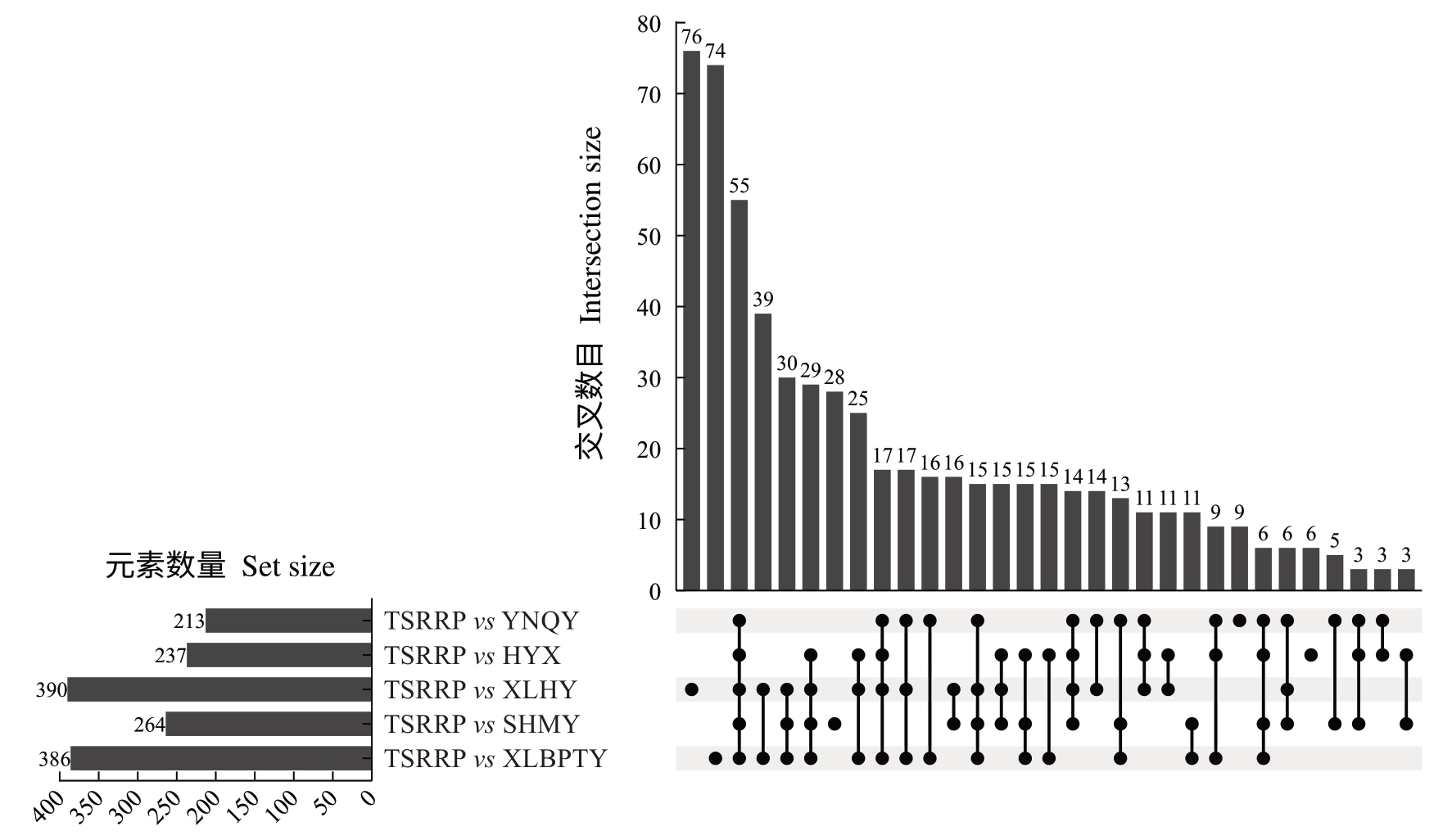
图5 差异代谢成分upset 图
Fig. 5 The upset plot of differential metabolites
2.4 泰国红宝石柚果肉差异代谢物聚类热图分析
对5个比较组合中同时具有差异的55种代谢物进行统计分析,结果显示,其主要包括苯丙酮和多酮类化合物(12种),脂质和类脂质分子(12种),有机酸及其衍生物(10 种),有机杂环化合物(8 种),苯类化合物(5 种),氨基酸及其衍生物(2 种),核苷、核苷酸类似物(2 种),有机含氧化合物(2 种),木脂素、新木脂素和相关化合物(1 种),香豆素和木质素(1 种)。苯丙酮和多酮类化合物、脂质和类脂质分子占比最高均为21.82%,其次为有机酸及其衍生物(18.18%)、有机杂环化合物(14.55%)、苯类化合物(9.09%)。为直观展示品种间的代谢物差异,对55 种显著差异代谢物表达量进行欧式聚类分析。结果表明(图6),在泰国红宝石柚中11-羟基-7-烯-6-酮、8-羟基-7(11)-佛术烯-12,8-内酯、双酚Z、α-香附酮、3-氨基-4-甲基戊酸、L-缬氨酸、DL-异亮氨酸、D-酪氨酸、L-酪氨酸、DL-酪氨酸、DL-间酪氨酸代谢物含量显著积累,而替米沙坦、东莨菪内酯代谢物含量较低。此外,基于样品欧式距离的聚类分析显示,泰国红宝石柚明显区别于其他5 个品种,其中越南青柚与星露比葡萄柚聚为一支,暹罗红柚、三红蜜柚、红玉香柚聚为一支。

图6 6 个红肉柚品种果肉差异代谢物热图
Fig. 6 The heatmap of differential metabolites in the six red-flesh pummelo cultivars
2.5 泰国红宝石柚果肉差异代谢物KEGG 通路富集分析
为进一步探究泰国红宝石柚成熟期果肉代谢物的生物学功能,对每个组合的差异代谢物进行KEGG代谢通路注释分析。注释结果分为环境信息处理、遗传信息处理、新陈代谢3类(表2)。如图7所示,差异代谢产物涉及多种代谢途径。其中次生代谢物的生物合成途径占比最大,其次是氨基酸代谢途径。此外,还包括脂质代谢相关通路,如甘油酯和甘油磷脂代谢;辅助因子和维生素代谢相关通路,如泛酸、辅酶A、硫胺素及类胡萝卜素的生物合成;碳水化合物代谢相关通路,如糖酵解、糖原生成及葡萄糖苷酸生物合成。这些通路均与果实内源物质转化和能量供应密切相关,直接影响果实发育和品质形成。
表2 5 个比较组合的差异代谢物KEGG 通路富集元素
Table 2 Differential metabolite KEGG pathway enrichment elements for the five comparison combinations
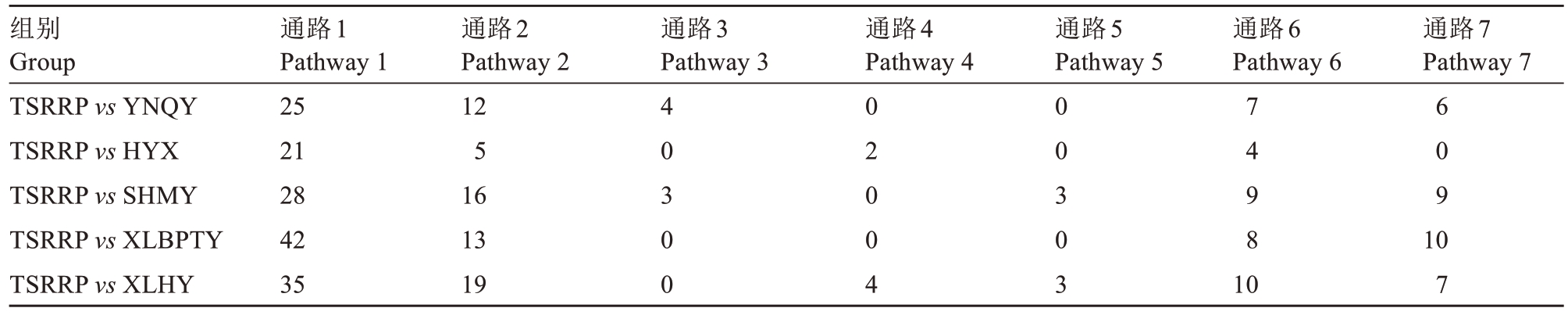
注:通路1 表示次生代谢物的生物合成;通路2 表示氨基酸代谢;通路3 表示脂质代谢;通路4 表示辅助因子和维生素代谢;通路5 表示碳水化合物代谢;通路6 表示翻译;通路7 表示膜转运。
Note:Pathway 1 denotes biosynthesis of secondary metabolites; pathway 2 denotes amino acid metabolism; pathway 3 denotes lipid metabolism;pathway 4 denotes metabolism of cofactors and vitamins; pathway 5 denotes carbohydrate metabolism; pathway 6 denotes translation; pathway 7 denotes membrane transport.
组别Group TSRRP vs YNQY TSRRP vs HYX TSRRP vs SHMY TSRRP vs XLBPTY TSRRP vs XLHY通路1 Pathway 1 25 21 28 42 35通路2 Pathway 2 12 5 16 13 19通路3 Pathway 3通路4 Pathway 4通路5 Pathway 5通路6 Pathway 6通路7 Pathway 7 4 0 3 0 0 0 2 0 0 4 0 0 3 0 3 7 4 9 8 6 0 9 10 7 10
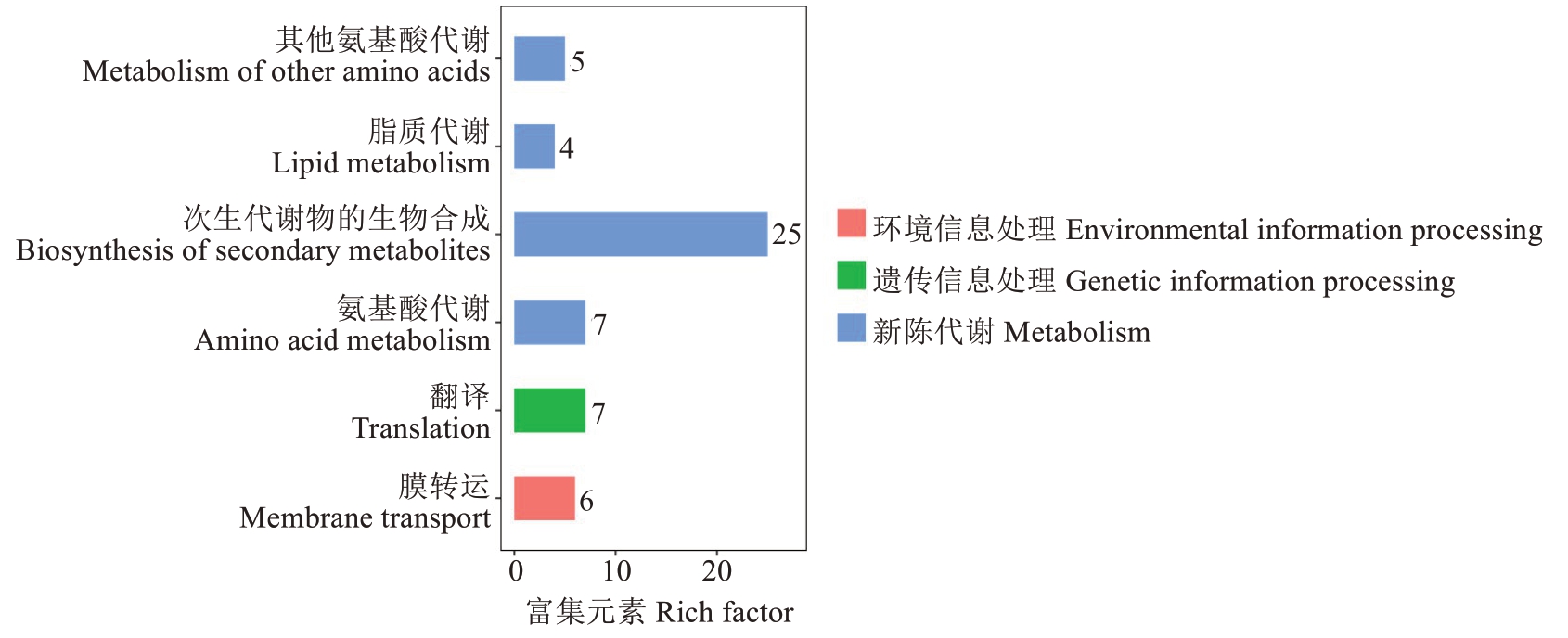
图7 泰国红宝石柚 vs 越南青柚差异代谢物KEGG 分类
Fig. 7 KEGG-classification plot of Tubtim Siam Red Ruby pummelo vs Yuenan Qingyou pummelo differential metabolites
基于差异代谢物的KEGG 差异丰度得分(图8)可知,显著富集的代谢通路为苯丙烷类生物合成、硫代葡萄糖苷生物合成、氨基酰-tRNA生物合成、氰氨酸生物合成途径。在苯丙烷类生物合成途径中,芥子酸、1-O-没食子酰基-β-D-葡萄糖含量上调;在硫代葡萄糖苷、氨基酰-tRNA和氰氨酸生物合成途径中,L-缬氨酸、L-酪氨酸、DL-异亮氨酸含量同时上调;在色氨酸代谢通路中,吲哚-3-乙醛含量上调;在甘油磷酸酯代谢通路中,1-戊二酰基-2-羟基-sn-甘油-3-磷酸胆碱含量上调;在其他次生代谢物生物合成途径中,滨蒿内酯、花椒醇含量上调,东莨菪内酯含量下调。
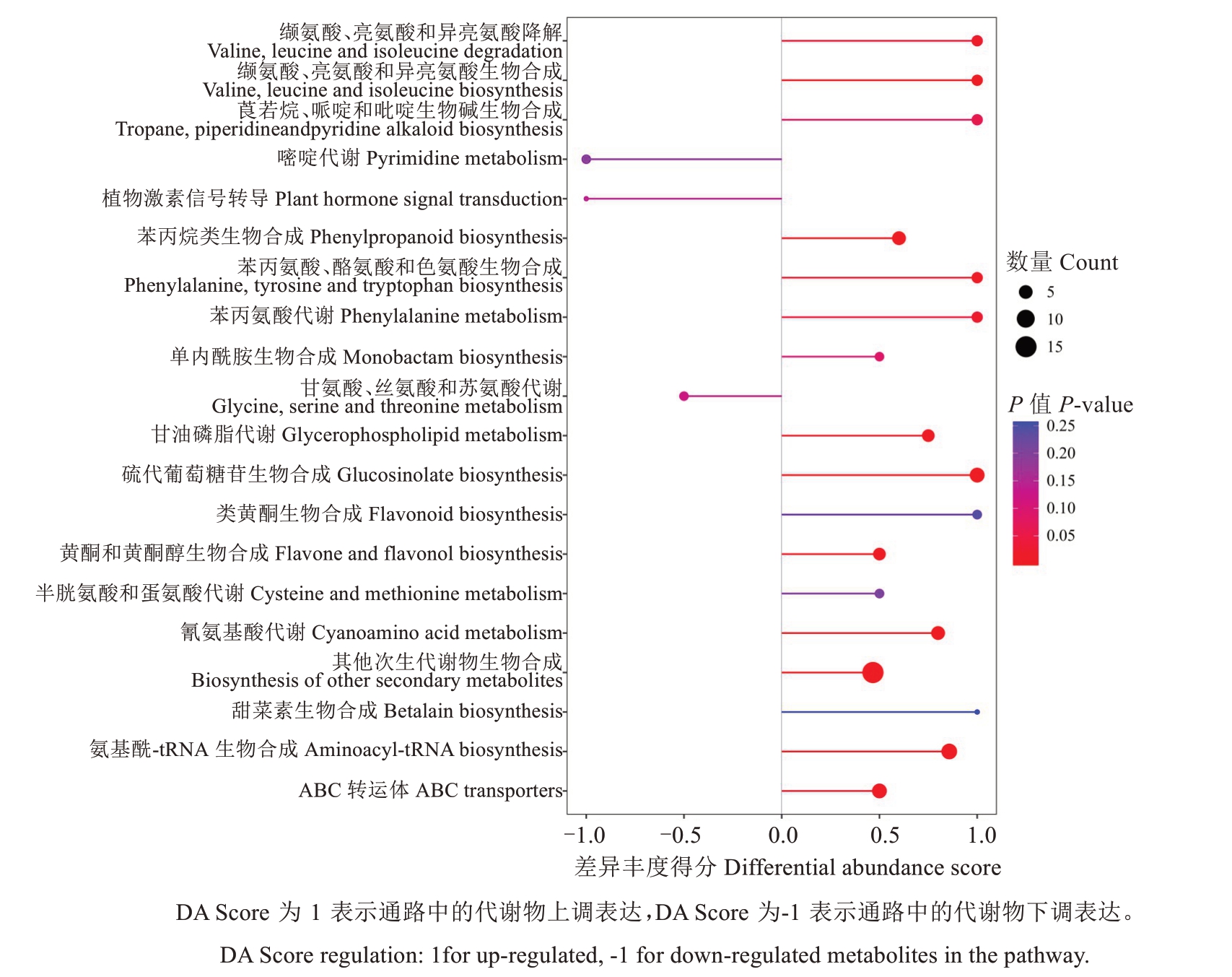
图8 泰国红宝石柚 vs 越南青柚差异代谢物KEGG 差异丰度得分图Fig. 8 KEGG differential abundance score(DA score) plot of Tubtim Siam Red Ruby pummelo vs Yuenan Qingyou pummelo differential metabolites
3 讨 论
在泰国红宝石柚、越南青柚、红玉香柚、三红蜜柚、星露比葡萄柚、暹罗红柚果肉中共检测到1019种代谢物,依据代谢成分化学特性及生物学功能将其分为24 个类别。其中,脂质和类脂质分子占比最高,其次是苯丙酮和多酮类化合物、有机酸及其衍生物、有机杂环化合物等。PCA 是一种无监督模式识别的多维数据统计分析方法,可初步了解各组样本之间的总体代谢差异、组内样本之间的变异度及组间代谢组分离趋势[20-22]。笔者对6 个品种果肉代谢物数据进行PCA 分析,结果显示PC1 和PC2 的累计方差贡献率为42.35%,泰国红宝石柚与其他5 个品种明显分离。与PCA相比,OPLS-DA可以使组间区分最大化,更利于寻找差异代谢物[23-25]。以VIP≥1、FC>2或者FC<0.5、P<0.05为标准,共筛选出差异代谢物606 种。在TSRRP vs XLHY 中差异代谢物有76 种,TSRRP vs XLBPTY 有74 种,TSRRP vs SHMY 有28 种,TSRRP vs YNQY 有9 种,TSRRP vs HYX 有6 种,上述差异代谢物可作为泰国红石柚区别于其他品种的潜在标志性代谢成分。
次生代谢是初生代谢的补充和发展,次生代谢物参与植物的营养生长和生殖生长以及抵御恶劣的生存环境,在植物的生长发育中扮演着十分重要的角色[15]。笔者利用UPLC-MS/MS 进行非靶向代谢组学分析,从6 个品种果肉中筛选出55 种差异代谢物。在泰国红宝石柚中11-羟基-7-烯-6-酮、8-羟基-7(11)-佛术烯-12,8-内酯、α-香附酮、3-氨基-4-甲基戊酸、L-缬氨酸、L-酪氨酸、DL-异亮氨酸等11 种代谢物含量高于其他品种。其中L-缬氨酸、L-酪氨酸为人体所必需的氨基酸,在食品、医药等领域广泛应用[26-27];3-氨基-4-甲基戊酸是一种β 氨基酸,是L-亮氨酸的异构体,在蛋白质合成和修复、免疫调节等方面具有重要作用[28];α-香附酮通过抑制BV-2 细胞中STING 信号通路的激活,下调炎症因子IL-6与TNFα的表达,从而降低炎症反应[29]。
此外,笔者对55 种差异代谢物进行KEGG[30-32]通路富集分析,发现上调的差异代谢物种类远高于下调。其中,L-缬氨酸、L-酪氨酸、DL-异亮氨酸在硫代葡萄糖苷、氨基酰-tRNA 和氰氨酸生物合成途径中同时上调。在苯丙烷类生物合成途径中,上调的芥子酸和1-O-没食子酰基-β-D-葡萄糖均具有一定的抗氧化、抗肿瘤功能。其中,芥子酸作为酚类酸的中间产物,通过苯丙烷类代谢途径和莽草酸途径合成,还可进一步转化为黄酮、类黄酮、花色苷类等多酚物质[10]。在其他次生代谢物生物合成途径中,上调的滨蒿内酯、花椒醇为香豆素类化合物。大量研究显示,柑橘香豆素具有抗炎、抗凝血以及保护心脑血管等多种生物学功能[33-34]。在甘油磷酸酯代谢通路中,上调的1-戊二酰基-2-羟基-sn-甘油-3-磷酸胆碱是细胞膜的主要组成成分之一。在色氨酸代谢通路中,上调的吲哚-3-乙醛具有抗肿瘤、抗菌、调节腺苷受体等多种生物活性功能,在药物化学领域广泛应用[35]。
4 结 论
通过系统比较泰国红宝石柚与5 个红肉柚品种果肉中代谢物的差异,共筛选出55 种差异代谢物,其显著富集的代谢通路主要为苯丙烷类和硫代葡萄糖苷等生物合成途径。进一步筛选出10 种主要代谢物,分别为芥子酸、1-O-没食子酰基-β-D-葡萄糖、L-缬氨酸、L-酪氨酸、DL-异亮氨酸、吲哚-3-乙醛、1-戊二酰基-2-羟基-sn-甘油-3-磷酸胆碱、滨蒿内酯、花椒醇、东莨菪内酯。泰国红宝石柚中有效成分的含量显著高于其他品种,表明其可能具有更高的药用和食用价值,为开发功能性产品及药物提供了一定的理论基础。
[1] 邓秀新. 中国柑橘育种60 年回顾与展望[J]. 园艺学报,2022,49(10):2063-2074.DENG Xiuxin. A review and perspective for citrus breeding in China during the last six decades[J]. Acta Horticulturae Sinica,2022,49(10):2063-2074.
[2] WU G A,TEROL J,IBANEZ V,LÓPEZ-GARCÍA A,PÉREZROMÁN E,BORREDÁ C,DOMINGO C,TADEO F R,CARBONELL-CABALLERO J,ALONSO R,CURK F,DU D L,OLLITRAULT P,ROOSE M L,DOPAZO J,GMITTER F G,ROKHSAR D S,TALON M. Genomics of the origin and evolution of citrus[J]. Nature,2018,554(7692):311-316.
[3] 刘承浪,冯迪,曹宗洪,闫素云,张永青,徐祥增,高世德,叶俊丽,柴利军,谢宗周,邓秀新. 东试早柚与沙田柚和水晶柚遗传背景比较[J]. 园艺学报,2023,50(11):2337-2349.LIU Chenglang,FENG Di,CAO Zonghong,YAN Suyun,ZHANG Yongqing,XU Xiangzeng,GAO Shide,YE Junli,CHAI Lijun,XIE Zongzhou,DENG Xiuxin. Comparative analysis of genetic background of ‘Dongshi Zaoyou’,‘Shatianyou’and ‘Shuijingyou’ pummelos[J]. Acta Horticulturae Sinica,2023,50(11):2337-2349.
[4] 胡宇,曹宗洪,孙怡,刘聪,文豪,刘慧敏,郑鑫,徐梦梦,蒋祥东,张友和,李光浩,叶俊丽,邓秀新,谢宗周,柴利军. ‘贡水白柚’无核成因探究[J]. 园艺学报,2025,52(1):1-12.HU Yu,CAO Zonghong,SUN Yi,LIU Cong,WEN Hao,LIU Huimin,ZHENG Xin,XU Mengmeng,JIANG Xiangdong,ZHANG Youhe,LI Guanghao,YE Junli,DENG Xiuxin,XIE Zongzhou,CHAI Lijun. Investigation of the causes of seedlessness in ‘Gongshui baiyou’ pummelo[J]. Acta Horticulturae Sinica,2025,52(1):1-12.
[5] 刘勇. 柚类资源分子系统学及其核心种质构建研究[D]. 武汉:华中农业大学,2005.LIU Yong. Molecular phylogenetic analysis and core collection construction using SSR and AFLP markers in pummelo[D]. Wuhan:Huazhong Agricultural University,2005.
[6] 郑雄杰. 柑橘果皮红色性状形成的生化基础及遗传机制[D].武汉:华中农业大学,2019.ZHENG Xiongjie. The biochemical and genetic mechanism underlying red color trait of citrus peel[D]. Wuhan:Huazhong Agricultural University,2019.
[7] 徐娟,邓秀新. 柑橘类果实汁胞的红色现象及其呈色色素[J].果树学报,2002,19(5):307-313.XU Juan,DENG Xiuxin. Red juice sac of citrus and its main pigments[J]. Journal of Fruit Science,2002,19(5):307-313.
[8] LU S W,ZHANG Y,ZHU K J,YANG W,YE J L,CHAI L J,XU Q,DENG X X. The citrus transcription factor CsMADS6 modulates carotenoid metabolism by directly regulating carotenogenic genes[J]. Plant Physiology,2018,176(4):2657-2676.
[9] ASAI T,MATSUKAWA T,KAJIYAMA S. Metabolomic analysis of primary metabolites in citrus leaf during defense responses[J].Journal of Bioscience and Bioengineering,2017,123(3):376-381.
[10] 曹小汉,毛惠敏,任莉萍. 代谢组学技术在植物次生代谢产物研究中的应用[J]. 农业与技术,2022,42(11):1-3.CAO Xiaohan,MAO Huimin,REN Liping. Application of metabolomics techniques in the study of plant secondary metabolites[J]. Agriculture and Technology,2022,42(11):1-3.
[11] 王梦迪,雍旭红,印敏,王奇志. 代谢组学技术在植物次生代谢调控研究中的应用[J]. 植物科学学报,2023,41(2):269-278.WANG Mengdi,YONG Xuhong,YIN Min,WANG Qizhi. Application of metabonomics in regulation study of plant secondary metabolites[J]. Plant Science Journal,2023,41(2):269-278.
[12] 赵小娜,田祥涛,梁清青,王燕君,朱姝昱,鲁周民. 基于非靶向代谢组学分析狮头柑生长过程主要滋味物质变化[J]. 食品科学,2025,46(8):198-210.ZHAO Xiaona,TIAN Xiangtao,LIANG Qingqing,WANG Yanjun,ZHU Shuyu,LU Zhoumin. Untargeted metabolomic analysis of changes in key taste substances during the growth process of shitougan (Citrus reticulata Blanco cv. Manau Gan)[J]. Food Science,2025,46(8):198-210.
[13] 邵淑贤,徐梦婷,林燕萍,陈潇敏,方德音,蔡捷英,王金焕,金珊,叶乃兴. 基于电子鼻与HS-SPME-GC-MS 技术对不同产地黄观音乌龙茶香气差异分析[J]. 食品科学,2023,44(4):232-239.SHAO Shuxian,XU Mengting,LIN Yanping,CHEN Xiaomin,FANG Deyin,CAI Jieying,WANG Jinhuan,JIN Shan,YE Naixing. Differential analysis of aroma components of Huangguanyin oolong tea from different geographical origins using electronic nose and headspace solid-phase microextraction-gas chromatography-mass spectrometry[J]. Food Science,2023,44(4):232-239.
[14] CACERES-CORTES J,FALK B,MUELLER L,DHAR T G M.Perspectives on nuclear magnetic resonance spectroscopy in drug discovery research[J]. Journal of Medicinal Chemistry,2024,67(3):1701-1733.
[15] 淡墨,高先富,谢国祥,刘忠,赵爱华,贾伟. 代谢组学在植物代谢研究中的应用[J]. 中国中药杂志,2007,32(22):2337-2341.DAN Mo,GAO Xianfu,XIE Guoxiang,LIU Zhong,ZHAO Aihua,JIA Wei. Application of metabolomics in research of plant metabolites[J]. China Journal of Chinese Materia Medica,2007,32(22):2337-2341.
[16] 明家琪. 红肉柚与普通柚成熟果实中品质相关代谢物质含量的分析[D]. 武汉:华中农业大学,2015.MING Jiaqi. Analysis of metabolites content related to quality of mature red/pale flesh pomelos[D]. Wuhan:Huazhong Agricultural University,2015.
[17] 陈婉冰,毛根林,钟玉娟,曾继吾. 基于代谢组学分析不同品系化州柚花间差异代谢物[J]. 果树学报,2024,41(10):1990-2001.CHEN Wanbing,MAO Genlin,ZHONG Yujuan,ZENG Jiwu.Metabolomic analysis of differential metabolites among different cultivars of Citrus grandis ‘Tomentosa’ flowers[J]. Journal of Fruit Science,2024,41(10):1990-2001.
[18] FAN R Y,ZHU C Y,QIU D Y,ZENG J W. Comparison of the bioactive chemical components and antioxidant activities in three tissues of six varieties of Citrus grandis ‘Tomentosa’fruits[J]. International Journal of Food Properties,2019,22(1):1848-1862.
[19] CHEN C J,CHEN H,ZHANG Y,THOMAS H R,FRANK M H,HE Y H,XIA R. TBtools:An integrative toolkit developed for interactive analyses of big biological data[J]. Molecular Plant,2020,13(8):1194-1202.
[20] 牟蛟琳,卢杨,张哲惠,叶俊丽,邓秀新. 柑橘果实成熟过程中氨基酸、维生素E 和脂质的动态分析[J]. 华中农业大学学报,2024,43(1):115-123.MOU Jiaolin,LU Yang,ZHANG Zhehui,YE Junli,DENG Xiuxin. Analyzing dynamics of amino acid,vitamin E and lipids during fruit ripening in citrus[J]. Journal of Huazhong Agricultural University,2024,43(1):115-123.
[21] 段敏仙,张碧蓉,杨帆,李雪佳,史文斌,闫素云,唐少平,赵俊,周先艳. 云南7 个澳洲坚果主栽品种果仁脂肪酸组分和含量分析[J]. 果树学报,2025,42(4):828-839.DUAN Minxian,ZHANG Birong,YANG Fan,LI Xuejia,SHI Wenbin,YAN Suyun,TANG Shaoping,ZHAO Jun,ZHOU Xianyan. Analysis of fatty acid compositions and concentrations in the kernel of seven main macadamia cultivars in Yunnan[J].Journal of Fruit Science,2025,42(4):828-839.
[22] 程玉娇,李贵节,欧阳祝,谈安群,吴厚玖,梁国鲁,王华,陈炜铃,王震寰. 基于气相色谱-质谱/脉冲火焰检测器和主成分分析对不同品种柚汁挥发性风味组分的分析[J]. 食品与发酵工业,2022,48(10):255-263.CHENG Yujiao,LI Guijie,OUYANG Zhu,TAN Anqun,WU Houjiu,LIANG Guolu,WANG Hua,CHEN Weiling,WANG Zhenhuan. Analysis of aroma compounds in different varieties of pomelo juice based on gas chromatography-mass spectrometry/pulsed flame photometric detector and principal component analysis method[J]. Food and Fermentation Industries,2022,48(10):255-263.
[23] KANG C D,ZHANG Y Y,ZHANG M Y,QI J,ZHAO W T,GU J,GUO W P,LI Y Y. Screening of specific quantitative peptides of beef by LC-MS/MS coupled with OPLS-DA[J]. Food Chemistry,2022,387:132932.
[24] HOW M S,HAMID N,LIU Y,KANTONO K,OEY I,WANG M F. Using OPLS-DA to fingerprint key free amino and fatty acids in understanding the influence of high pressure processing in New Zealand clams[J]. Foods,2023,12(6):1162.
[25] 吴雅诺. 基于感官评价及代谢组学比较不同产地柑橘果实品质差异[D]. 武汉:华中农业大学,2023.WU Yanuo. Intergrated sensory evaluation with metabolomics to compare the quality differences of citrus fruits from different regions[D]. Wuhan:Huazhong Agricultural University,2023.
[26] 柳亚迪. 乙酰羟酸合酶的进化及其在L-缬氨酸合成中关键作用的研究[D]. 无锡:江南大学,2019.LIU Yadi. Studies on the evolution of acetohydroxyacid synthase and its key role on the biosynthesis of L-valine[D]. Wuxi:Jiangnan University,2019.
[27] 杨菲,王玥,石选平,尤甲甲,邵明龙,徐美娟,饶志明. 多酶级联全细胞转化合成酪氨酸[J/OL]. 生物工程学报,2025:1-20.(2025-05-08). https://link.cnki.net/doi/10.13345/j.cjb.250175.YANG Fei,WANG Yue,SHI Xuanping,YOU Jiajia,SHAO Minglong,XU Meijuan,RAO Zhiming. Whole-cell transformation for the synthesis of tyrosine by a multi-enzyme cascade[J/OL]. Chinese Journal of Biotechnology,2025:1-20. (2025-05-08). https://link.cnki.net/doi/10.13345/j.cjb.250175.
[28] LYNCH C J,FOX H L,VARY T C,JEFFERSON L S,KIMBALL S R. Regulation of amino acid–sensitive TOR signaling by leucine analogues in adipocytes[J]. Journal of Cellular Biochemistry,2000,77(2):234-251.
[29] 高鹏. 肿瘤源性DNA 对小胶质细胞STING 信号通路的作用机制及α-香附酮的调节作用[D]. 大连:大连医科大学,2021.GAO Peng. α- cyperone inhibitory effects on tumor- derived DNA trigger microglia by STING pathway[D]. Dalian:Dalian Medical University,2021.
[30] KANEHISA M,GOTO S. KEGG kyoto encyclopedia of genes and genomes[J]. Nucleic Acids Research,2000,28(1):27-30.
[31] 李港秋. 无核椪柑‘华柑4 号’果实发育研究[D]. 武汉:华中农业大学,2023.LI Gangqiu. Study on the fruit development of seedless ponkan‘Huagan No. 4’[D]. Wuhan:Huazhong Agricultural University,2023.
[32] 杨梦宇,杜洁,王来平,王贵平,陈汝,薛晓敏. 基于代谢组学分析ATS 对威海金苹果的疏花作用机制[J]. 果树学报,2025,42(6):1137-1147.YANG Mengyu,DU Jie,WANG Laiping,WANG Guiping,CHEN Ru,XUE Xiaomin. Mechanism of ATS-induced flower thinning in Weihai Gold apple revealed by metabolomics analysis[J]. Journal of Fruit Science,2025,42(6):1137-1147.
[33] MEDINA F G,MARRERO J G,MACÍAS- ALONSO M,GONZÁLEZ M C,CÓRDOVA- GUERRERO I,TEISSIER GARCÍA A G,OSEGUEDA-ROBLES S. Coumarin heterocyclic derivatives:Chemical synthesis and biological activity[J].Natural Product Reports,2015,32(10):1472-1507.
[34] PATIL S B. Medicinal significance of novel coumarin analogs:Recent studies[J]. Results in Chemistry,2022,4:100313.
[35] 胡鸿雨,王飞,赵胜贤,方美娟. 吲哚-3-乙醛酰胺类化合物的合成及生物活性研究进展[J]. 合成化学,2017,25(2):178-184.HU Hongyu,WANG Fei,ZHAO Shengxian,FANG Meijuan.Research progress on the synthesis and bioactivities of indole-3-glyoxylamide derivatives[J]. Chinese Journal of Synthetic Chemistry,2017,25(2):178-184.