草莓(Fragaria×ananassa Duch.)为蔷薇科(Rosaceae)草莓属(Fragaria)多年生草本植物,是重要的园艺作物。草莓色泽红艳,肉质多汁,风味酸甜且芳香浓郁,营养价值高[1],素有“水果皇后”之称[2]。近年来,中国草莓栽培面积持续扩大,国内新品种选育成效显著,产业规模快速增长,经济效益和社会效益同步提升[3]。然而,草莓基因组高度杂合,传统育种方法存在周期长、性状改良效率低等技术瓶颈。转基因技术与基因编辑技术(如CRISPR/Cas9)因操作精准、育种周期短、遗传修饰效率高等优势,为草莓种质创新和新品种选育提供了新的解决方案[4]。因此,建立高效稳定的草莓遗传转化体系,对推进草莓分子设计育种、加速优良品种选育进程具有重要意义[5]。
植物的遗传转化主要利用根癌农杆菌(Agrobacterium tumefaciens)与发根农杆菌(Agrobacterium rhizogenes)[6]。目前,基于根癌农杆菌的叶盘转化法是草莓遗传转化的常规方法。该方法以叶片为外植体,经农杆菌浸染、抗生素筛选及器官再生途径获得转基因植株[7-8]。然而,该体系转化周期长、转化效率低、影响因素较多[9]。相较而言,发根农杆菌介导法(A. rhizogenes-mediated,ARM)具有成本低、效能高、基因沉默率低及宿主适应性强等优势,是一种高效的植物遗传转化方法。它能够快速将自身含有的Ri质粒上T-DNA片段转移至植物细胞中,诱导植物产生毛状根,从而实现遗传物质的交流[10-11],其转化周期通常比根癌农杆菌的叶盘转化法更短。发根农杆菌介导法能够快速获得大量的转基因材料,为后续的基因功能验证、次生代谢产物研究等提供充足的试验样本,极大提高了研究效率。 此外,发根农杆菌具有较强的宿主适应性,不仅能够感染双子叶植物,对部分单子叶植物、裸子植物以及非植物生物如真菌等也具有一定的感染能力,突破了根癌农杆菌宿主范围的限制,极大地拓宽了植物遗传转化的研究范围,为不同种类植物的遗传改良提供了可能。舒铫等[12]在甜瓜中利用发根农杆菌介导法建立了甜瓜转化体系,并利用该系统实现了对CRISPR/Cas9靶点有效性的快速检测。邬婷婷等[13]在蓝莓中也建立了简单、快速的发根农杆菌介导的蓝美1 号遗传转化体系,为蓝莓的基因功能研究和良种选育提供了技术支持。在草莓中,Yan 等[14]利用发根农杆菌在林地草莓中探索出一种有效的侵染方法,该方法可用于草莓根系特异性启动子的研究,并可作为分析草莓根系基因功能的工具。此外,Hao 等[15]在草莓中利用发根农杆菌体内注射(IVI)方法,得到转基因毛状根,并用转基因毛状根系证实了FaNRT1.1的硝酸盐转运功能。
在草莓遗传转化研究中,现有技术主要依赖组织培养[14]和体内注射 [15]获取转基因毛状根。然而,这些方法存在明显局限:IVI法虽规避了组织培养步骤,但操作流程复杂、转化效率低且阳性根获取周期长。此外,现有体系普遍采用GFP 荧光标记筛选,依赖荧光显微镜观察,增加了技术门槛和设备成本。针对以上问题,笔者创新性地引入RUBY 报告系统,进行阳性毛状根筛选,无需使用特殊设备或化学处理,肉眼即可清楚观察[16]。在模式植物拟南芥、水稻[17]以及园艺植物辣椒[18]、矮牵牛[19]、柠檬[20]中均已验证了RUBY 报告系统能够有效发挥预期的功能。因此,通过在草莓中建立以RUBY 报告基因筛选的发根农杆菌介导的遗传转化体系,可简化操作并有望克服GFP 荧光标签的弊端,为提高基因功能研究的准确性和遗传转化过程中的筛选效率奠定基础。
1 材料和方法
1.1 试验材料
发根农杆菌菌株Ar.1193、Ar.Qual、C58C1、K599(表1),所用菌种由上海交通大学农业与生物学院刘振华老师、温海帆老师、李彭丽老师提供;35S::RUBY 质粒购于广西唯创生物科技有限公司;草莓试验材料来自本实验室。
表1 发根农杆菌菌株抗性
Table 1 Resistance of Agrobacterium rhizogenes strains

菌株Strains Ar.1193 Ar.Qual C58C1 K599抗性Resistance羧苄青霉素、链霉素、利福平 Carb, Strep, Rif链霉素、氯霉素 Strep, Cam链霉素、利福平 Strep, Rif链霉素Strep
1.2 外植体获取
匍匐茎子株划伤法:挑选八倍体栽培草莓粉玉健康无病虫害的匍匐茎子株,因其子株部位生根能力较强,故选取此部位作为试验材料。
短缩茎穿刺法:挑选1 月龄无病虫害的二倍体森林草莓rg40 幼苗短缩茎,因其短缩茎部位生根能力较强,并且rg40 叶柄呈绿色,无花青素积累,可避免色素干扰,故选取此部位作为该方法的试验材料。
下胚轴侵染法:挑选二倍体森林草莓Ruegen(简称RG)成熟种子,经表面消毒处理。置于2 mL离心管中,经75%乙醇振荡消毒1 min,吸出乙醇。随后,用10%的次氯酸钠溶液消毒8 min 至种皮脱色,无菌水漂洗3~5 次,播于MS+2%蔗糖的固体培养基中,置于光照培养箱(24 ℃,光照16 h/黑暗8 h)中培养。待幼苗发育至2 片子叶期时,在无菌条件下将其下胚轴与根部连接处切断。因其下胚轴生根能力强[14],故选用携带子叶的下胚轴作为该方法的试验材料。
1.3 发根农杆菌转化
将-80 ℃冰箱中的发根农杆菌感受态置于冰上,待其融化后置于冰盒。每100 µL感受态加入1 µg 35S::RUBY质粒,依次于冰上静置5 min,液氮5 min,37 ℃水浴5 min,冰浴5 min,随后加入700 µL无抗生素的YEP 液体培养基,置于28 ℃摇床上220 r·min-1振荡培养2~3 h。离心后弃掉部分上清液,剩余100 µL 上清液重悬菌体沉淀后涂布在含有对应抗生素的YEP平板上,28 ℃倒置培养2~3 d,以获得单克隆。挑取单克隆置于含相应抗生素的YEP 液体培养基中振荡培养过夜,第2 天进行菌液PCR 扩增验证。根据载体序列合成RUBY检测引物:RUBY检测-F(5'-3')GACAGCCGAGACAGACCAAA 和RUBY 检测-R(5'-3')TCCCAGGAGGAGTGGATCAG。PCR 产物经琼脂糖凝胶电泳检测,目的条带为441 bp,条带大小正确的为阳性克隆,用于后续试验。
1.4 转基因根的鉴定
采用CTAB方法提取DNA[21]。将获得的毛状根置于1.5 mL 离心管中,向离心管中加入2 颗钢珠于液氮中速冻,快速置于组织破碎仪中破碎;随后,加入600 μL CTAB裂解液(含4%的β-巯基乙醇),涡旋混匀,60 ℃烘箱中放置30 min;加入600 μL 氯仿和异戊醇的混合液(氯仿和异戊醇的体积比为24∶1),振荡混匀,在13 400 g条件下离心10 min,吸出600 μL上清液,转移到新的离心管中;随后,加入600 μL 氯仿和异戊醇的混合液,振荡混匀,在13 400 g条件下离心10 min,吸出450 μL上清液,并加入在-20 ℃预冷的无水乙醇900 μL,颠倒混匀,于-20 ℃冰箱放置1 h;随后,在13 400 g 条件下离心10 min,去除上清液,置于通风橱下吹干;最后,加入30 μL TE 缓冲液(pH=8.0,每1.5 mL TE 缓冲液含100 μL 核糖核酸酶A溶液),置于-20 ℃冰箱保存待用。
采用双重PCR 体系进行转基因毛状根的分子鉴定。使用RUBY 特异性引物RUBY 检测-F 和RUBY 检测-R 扩增载体35S::RUBY 的序列,目的条带大小为441 bp;同时,利用引物FvACTIN-F(5'-3')GAGGCTCCATCTTAGCATCC 和引物FvACTIN-R(5'-3')ACAATTGAAGGGCCTGATTC 扩增草莓内参基因,目标条带大小为184 bp,经琼脂糖凝胶电泳检测,同时呈现441 bp(RUBY)和184 bp(ACTIN)特异性条带的样本,可确认为转基因阳性毛状根。
1.5 数据统计与分析
使用Excel统计数据,计算公式如下:

使用Adobe Illustrator 2020软件作图。
2 结果与分析
2.1 草莓发根遗传转化体系的构建
2.1.1 匍匐茎子株划伤法转化流程 将含有35S::RUBY 的农杆菌在YEP 固体培养基上划线,在28 ℃恒温培养箱中培养2~3 d(图1-A)。选取八倍体栽培草莓粉玉健康匍匐茎子株,去除子株上原有根系,使用灭菌刀片在原生根部位轻划5~6 道5~8 mm 长的伤口(图1-B)。用灭菌枪头轻刮固体菌落,涂抹在子株伤口表面(图1-C~D,箭头标注为涂抹部位)。采用一次性透明塑料杯作为培养容器,用镊子将杯底部戳孔以吸水(图1-E),将灭菌蛭石(粒径1~2 mm)装入杯中作为基质(图1-F)。随后,将涂抹菌体的子株垂直扦插于透明塑料杯中,确保接种部位与基质充分接触(图1-G)。杯体外周包裹锡箔纸,模拟根部的暗生长环境(图1-H),将锡箔纸取下后仍可通过透明杯壁观察毛状根生长动态。用无菌水重悬平板上剩余菌体,吸取菌液补充浇灌接种部位(图1-I),幼苗用透明保湿罩覆盖(图1-J),以确保高湿度环境,并使用黑布进行过夜暗处理,黑暗处理16 h 后转入正常光周期(光照16 h/黑暗8 h)。扦插20 d后进行统一观察。

图1 匍匐茎子株划伤法转化流程图
Fig. 1 Flowchart of transformation for the scratching stolon method
2.1.2 短缩茎穿刺法转化流程 将含有35S::RUBY的农杆菌在YEP固体培养基上划线、28 ℃恒温培养箱中培养2~3 d(图2-A)。选取1 月龄森林草莓苗rg40 作为试验材料(图2-B),用注射器针头挑取平板上的单克隆,对准幼苗短缩茎部位进行4~6 次垂直穿刺(图2-C,箭头标注为短缩茎部位),用注射器针管盖刮取平板上的菌体,均匀涂抹至短缩茎创伤部位,随后将幼苗种植于基质中(图2-D~F,箭头标注为短缩茎部位)。鉴于二倍体幼苗幼嫩易损,为避免早期取样损伤,定于45 d后进行统一观察。

图2 短缩茎穿刺法转化流程图
Fig. 2 Flowchart of transformation for the puncturing shortened stem method
2.1.3 下胚轴侵染法转化流程 将含有35S::RUBY的农杆菌在YEP固体培养基上划线、28 ℃恒温培养箱培养2~3 d,待其长出单克隆。选择阳性单克隆接种于含有75 mg·L-1壮观霉素和50 mg·L-1链霉素的YEP液体培养基中,在28 ℃、220 r·min-1摇床中培养16 h。第2天按照1∶100的比例接种于50 mL含有壮观霉素和链霉素的YEP 液体培养基中,并在28 ℃、220 r·min-1 摇床中培养直至OD600 达到0.6。接着3220 g离心10 min,收集菌液沉淀,用含有100 µmol·L-1乙酰丁香酮(AS)的1/2MS 液体培养基进行重悬,使其OD600达到0.8。在28 ℃、100 r·min-1条件下活化菌液1 h。当RG 出现2 片子叶时,切断下胚轴与根部连接处,选用携带子叶的下胚轴作为外植体材料(图3-A)。将外植体浸没在农杆菌溶液中,在28 ℃、100 r·min-1条件下培养侵染15 min(图3-B),随后超声5 s(KQ-500DE 型数控超声波清洗器,功率的90%)(图3-C)。将侵染后的外植体转移到用1/2MS+2%蔗糖(含有100 µmol·L-1 AS)润湿的滤纸上,23 ℃黑暗共培养4 d(图3-D)。共培养后,将感染的外植体用无菌水洗涤并转移到含250 mg·mL-1头孢噻肟钠(Cef)的1/2MS+2%蔗糖培养基上,脱菌培养以诱导毛状根产生(图3-E)。侵染处理30 d 后进行统一观察。
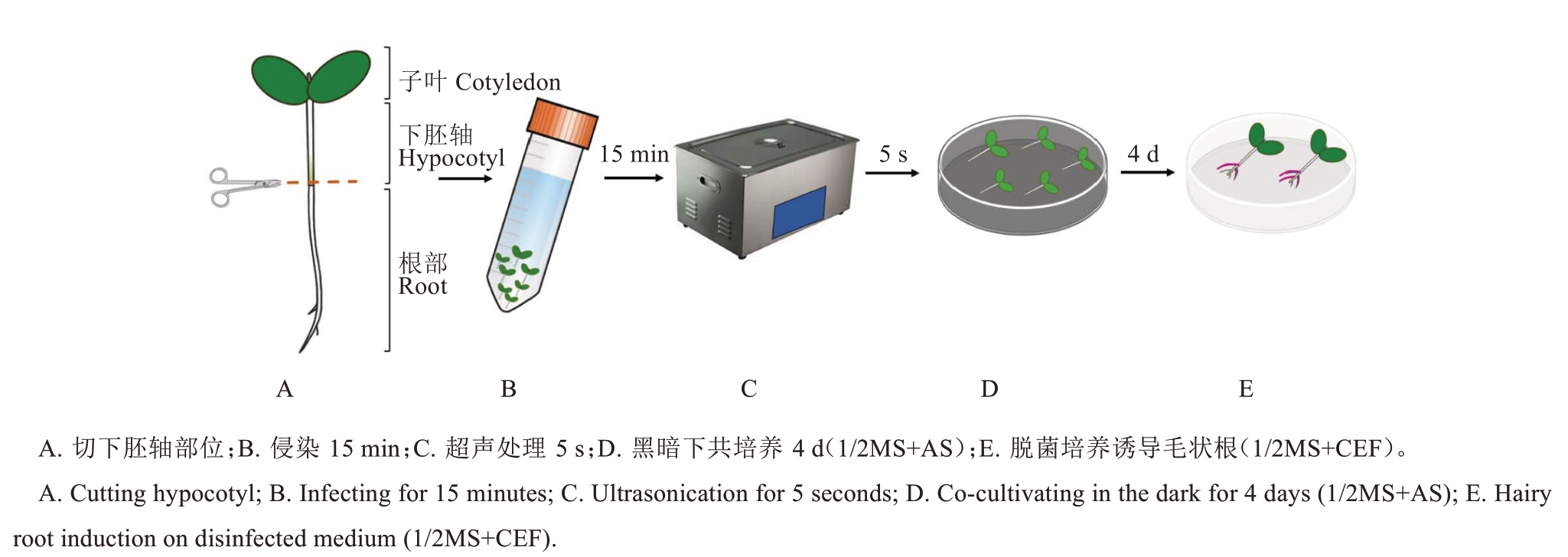
图3 下胚轴侵染法转化流程图
Fig. 3 Flowchart of transformation for the hypocotyl infection method
2.2 毛状根表型观察及阳性鉴定
2.2.1 匍匐茎子株划伤法根系表型观察与鉴定 试验组(RUBY-OE)采用携带35S::RUBY 质粒的4 种菌株(Ar.1193、Ar.Qual、C58C1、K599),对照组(CK)采用对应不携带任何质粒的4 种菌株。在接种后9 d时,观察到试验组根部出现红色表型。接种后20 d时,对透明杯壁观察的结果显示,对照组只产生白色根系(图4-A),而试验组则出现明显的红色根系(图4-B)。随后,将所有材料拔出清洗,进一步观察根部表型。结果同样表明,对照组均产生白色根系,而试验组均能诱导出具有显著甜菜红素积累的红色毛状根(图5)。

图4 匍匐茎子株划伤法杯身表型观察
Fig. 4 Observation from the cup of the scratching stolon method

图5 匍匐茎子株划伤法根系表型观察
Fig. 5 Phenotype of hairy roots of the scratching stolon method
对该方法获得的根系进行随机取样,根据经典CTAB 方法提取DNA。采用双重PCR 体系进行转基因毛状根的分子鉴定,利用引物RUBY 检测-F 和RUBY 检测-R 扩增载体35S::RUBY 的序列,目的条带大小为441 bp;利用引物FvACTIN-F 和FvACTINR 扩增草莓内参基因,目标条带大小为184 bp。同时呈现441 bp(RUBY)和184 bp(ACTIN)特异性条带的样本,可在DNA水平上确认为转基因阳性毛状根。PCR 结果显示,对照组白色根系均未扩增出RUBY 条带,而所取红色根系均可扩增出RUBY 条带,RUBY条带大小与35S::RUBY质粒扩增的大小一致(图6-A),并且红色根中草莓内参基因ACTIN 均存在(图6-B),证实这些红色毛状根为转基因阳性根。
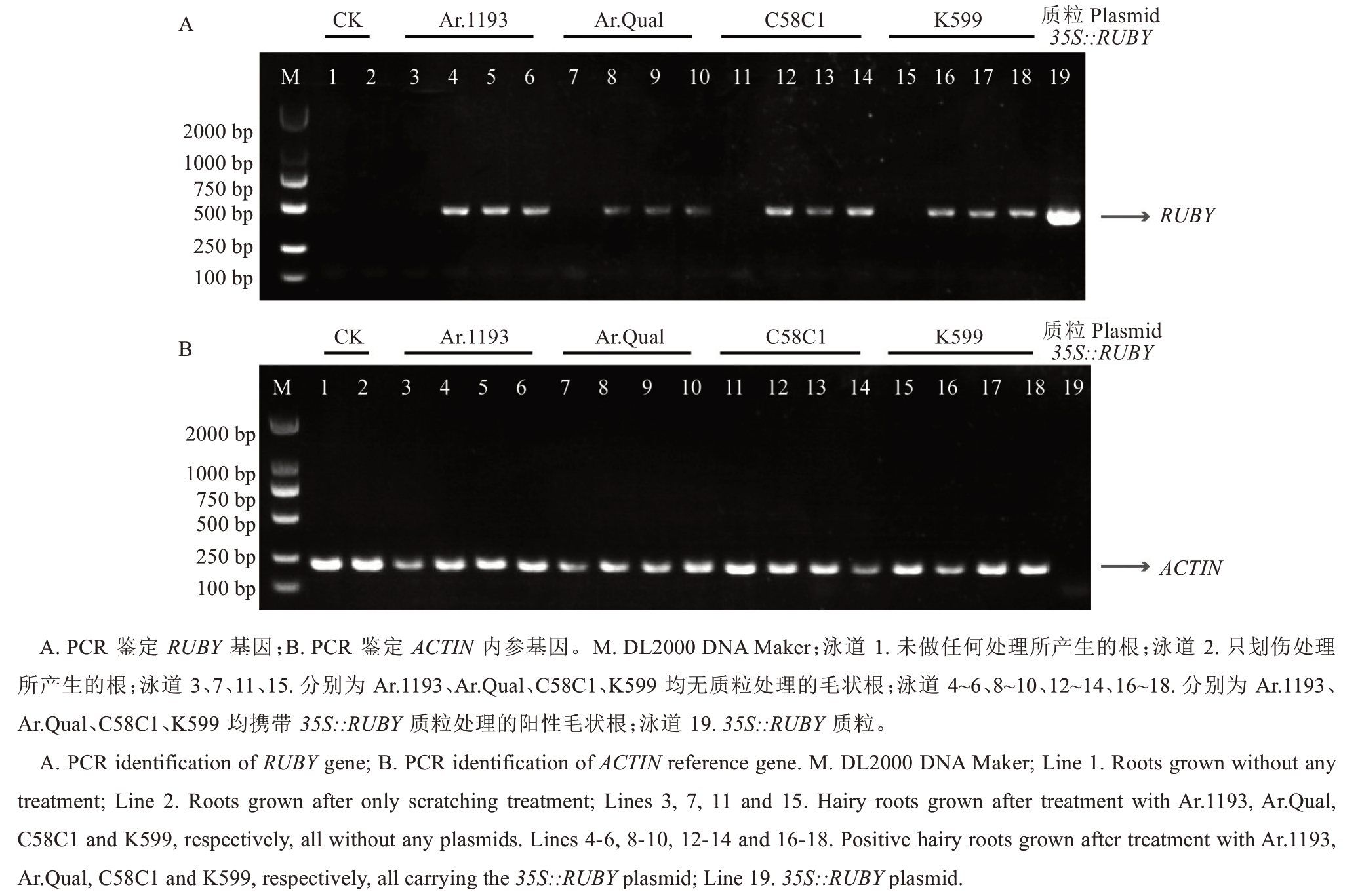
图6 匍匐茎子株划伤法阳性根鉴定
Fig. 6 Identification of positive hairy roots of the scratching stolon method
2.2.2 短缩茎穿刺法根系表型观察与鉴定 由于二倍体小苗较为幼嫩,在较早时期不便于拔起观察,选取在45 d 进行表型观察。试验组(RUBY-OE)采用携带35S::RUBY 质粒的4 种菌株(Ar.1193、Ar.Qual、C58C1、K599),对照组(CK)采用对应不携带任何质粒的4 种菌株。观察结果表明,左侧对照组的新生根为白色,右侧携带35S::RUBY 质粒的试验组产生红色毛状根(图7)。

图7 短缩茎穿刺法根系表型观察
Fig. 7 Phenotype of hairy roots of the puncturing shortened stem method
同样,对该方法获得的根系进行随机取样并提取DNA。PCR 结果显示,对照组白色根均未扩增出RUBY 条带,而所取红色根均可扩增出RUBY 条带,条带大小与35S::RUBY 质粒扩增的大小一致(图8-A),并且红色根中草莓内参基因ACTIN 均存在(图8-B),进一步在DNA水平确认红色根为转基因阳性根。

图8 短缩茎穿刺法阳性根鉴定
Fig. 8 Identification of positive hairy roots of the puncturing shortened stem method
2.2.3 下胚轴侵染法愈伤及根系表型观察与鉴定以携带35S::RUBY质粒的4种菌株作为试验组(RUBY-OE),以不携带任何质粒的对应4 种菌株分别作为对照组(CK)。将带子叶的下胚轴脱菌培养(含250 mg·mL-1 Cef 的1/2 MS+2%蔗糖培养基)7~12 d时,可以观察到下胚轴伤口部位有毛状根出现。侵染30 d后进行统计,观察到对照组产生白色根(图9-A),试验组有紫红色根的产生,并且产生了红色愈伤组织(图9-B)。对获得的根进行随机取样并提取DNA,PCR 结果表明,对照组的白色根均未扩增出RUBY条带,而试验组所取紫红色根均可扩增出RUBY条带,并且条带大小正确(图9-C)。此外,紫红色根中草莓内参基因ACTIN 均存在(图9-D),进一步在DNA水平确认紫红色根为转基因阳性根。

图9 下胚轴侵染法根系及愈伤组织表型观察与阳性根鉴定
Fig. 9 Phenotype of hairy roots and callus and identification of positive hairy roots of the hypocotyl infection method
2.3 4种发根农杆菌菌株在不同方法下的转化效率
对4 种发根农杆菌菌株在匍匐茎子株划伤法、短缩茎穿刺法、下胚轴侵染法3 种方法下的转化情况进行统计。
2.3.1 在匍匐茎子株划伤法中对毛状根转化的影响 将未携带任何质粒的4 种发根农杆菌菌株侵染的划伤匍匐茎子株作为对照组,携带35S::RUBY 质粒的4 种菌株侵染的划伤匍匐茎子株作为试验组。在20 d时进行观察统计,在该方法下,4种携带35S::RUBY 质粒的发根农杆菌菌株的阳性根率均超过58%。其中Ar.1193 的阳性根率最高,可达79.41%;Ar.Qual 与K599 的阳性率分别为67.65%与65.71%(表2)。因此在该方法下,最佳菌株为Ar.1193。
表2 4 种发根农杆菌菌株在匍匐茎子株划伤法中对毛状根转化的影响
Table 2 Effects of four A. rhizogenes strains on hairy root transformation in the scratching stolon method

注:1 对照组. 不携带任何质粒的4 种菌株划伤的匍匐茎子株;2 试验组. 携带35S::RUBY 质粒的4 种菌株划伤的匍匐茎子株。
Note:1 Control groups. Stolon-derived plantlets were scratched by four strains that did not carry any plasmids. 2 Experimental groups. Stolon-derived plantlets were scratched by four strains that carry 35S::RUBY plasmid.
阳性根率Frequency of positive roots/%0.00 0.00 0.00 0.00 79.41 67.65 58.82 65.71菌株Strains Ar.11931 Ar.Qual1 C58C11 K5991 Ar.1193-RUBY2 Ar.Qual-RUBY2 C58C1-RUBY2 K599-RUBY2外植体数Number of explants 12 12 12 12 36 36 37 38毛状根的外植体数Number of explants with hairy roots 11 12 10 11 34 34 34 35阳性根的外植体数Number of explants with positive roots 0 0 0 0 27 23 20 23再生根率Regeneration rate of roots/%91.67 100.00 83.33 91.67 94.44 94.44 91.89 92.11
2.3.2 在短缩茎穿刺法中对毛状根转化的影响 将未携带任何质粒的4 种菌株穿刺的短缩茎作为对照组,携带35S::RUBY 质粒的4 种菌株穿刺的短缩茎作为试验组。在45 d时进行观察统计,在该方法下,有3 种携带35S::RUBY 质粒的菌株阳性根率超过50%。其中,Ar.1193 处理效果最好,阳性根率可达73.33%;其次是K599,阳性根率达到58.06%;Ar.Qual 的阳性根率为50.00%(表3)。因此在该方法下,最佳菌株为Ar.1193。
表3 4 种发根农杆菌菌株在短缩茎穿刺法中对毛状根转化的影响
Table 3 Effects of four A. rhizogenes strains on hairy root transformation in the puncturing shortened stem method
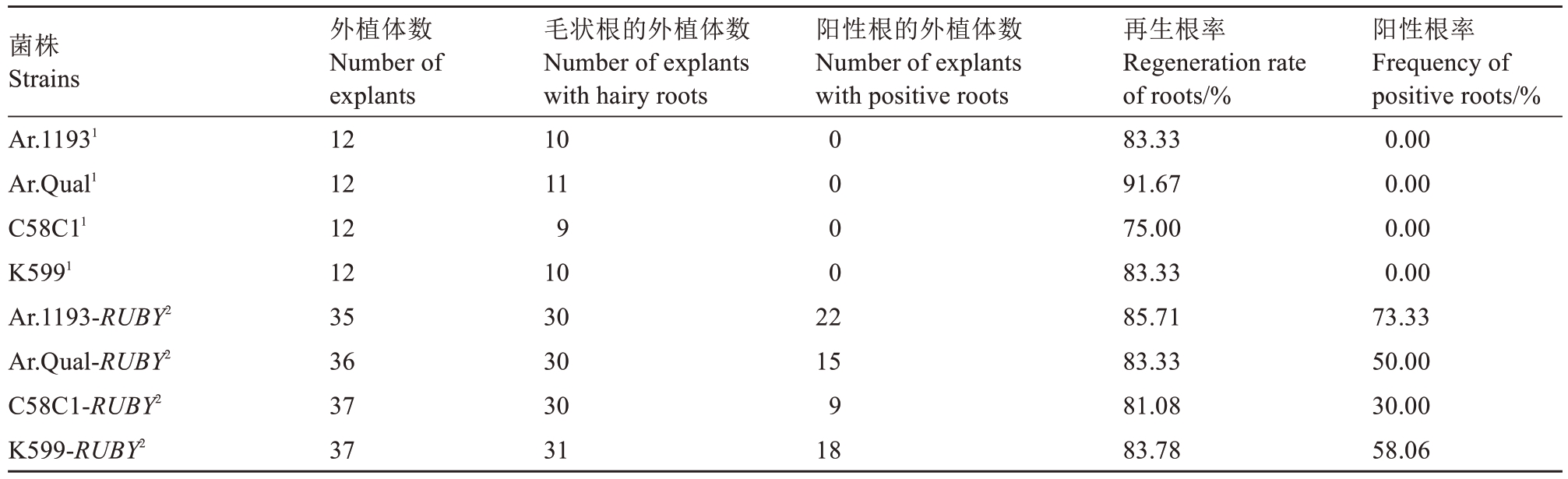
注:1 对照组. 不携带任何质粒的4 种菌株穿刺的短缩茎;2 试验组. 携带35S::RUBY 质粒的4 种菌株穿刺的短缩茎。
Note:1 Control groups. Shortened stem punctured by four strains that did not carry any plasmids. 2 Experimental groups. Shortened stem punctured by four strains that carry 35S::RUBY plasmid.
阳性根率Frequency of positive roots/%0.00 0.00 0.00 0.00 73.33 50.00 30.00 58.06菌株Strains Ar.11931 Ar.Qual1 C58C11 K5991 Ar.1193-RUBY2 Ar.Qual-RUBY2 C58C1-RUBY2 K599-RUBY2外植体数Number of explants 12 12 12 12 35 36 37 37毛状根的外植体数Number of explants with hairy roots 10 11 9 10 30 30 30 31阳性根的外植体数Number of explants with positive roots 0 0 0 0 22 15 9 18再生根率Regeneration rate of roots/%83.33 91.67 75.00 83.33 85.71 83.33 81.08 83.78
2.3.3 在下胚轴侵染法中对毛状根转化的影响 在30 d 时,统计各项数值。由表4 可知,Ar.1193 与Ar.Qual两种菌株在携带或不携带35S::RUBY质粒时诱导的生根率较高,均超过60%。在阳性根率方面,Ar.1193 菌株达到14.38% ,Ar.Qual 菌株达到12.50%。此外,笔者发现,在诱导毛状根的同时也伴随着愈伤组织的产生。菌株Ar.1193与Ar.Qual在诱导愈伤组织方面表现突出,不携带质粒的2 种菌株侵染胚轴后愈伤率分别为90.74%和82.69%,携带35S::RUBY 质粒的2 种菌株的愈伤率分别为94.55%和80.54%,2 种菌株诱导产生的愈伤组织均较大且较为明显。K599 菌株诱导产生的愈伤组织较小且较少;Ar.Qual 菌株诱导所产生的红色愈伤组织较多,红色愈伤率高达88.59%;Ar.1193 菌株的红色愈伤率达到73.82%。
表4 4 种发根农杆菌菌株在下胚轴侵染法中对毛状根转化的影响
Table 4 Effects of four A. rhizogenes strains on hairy root transformation in hypocotyl infection method
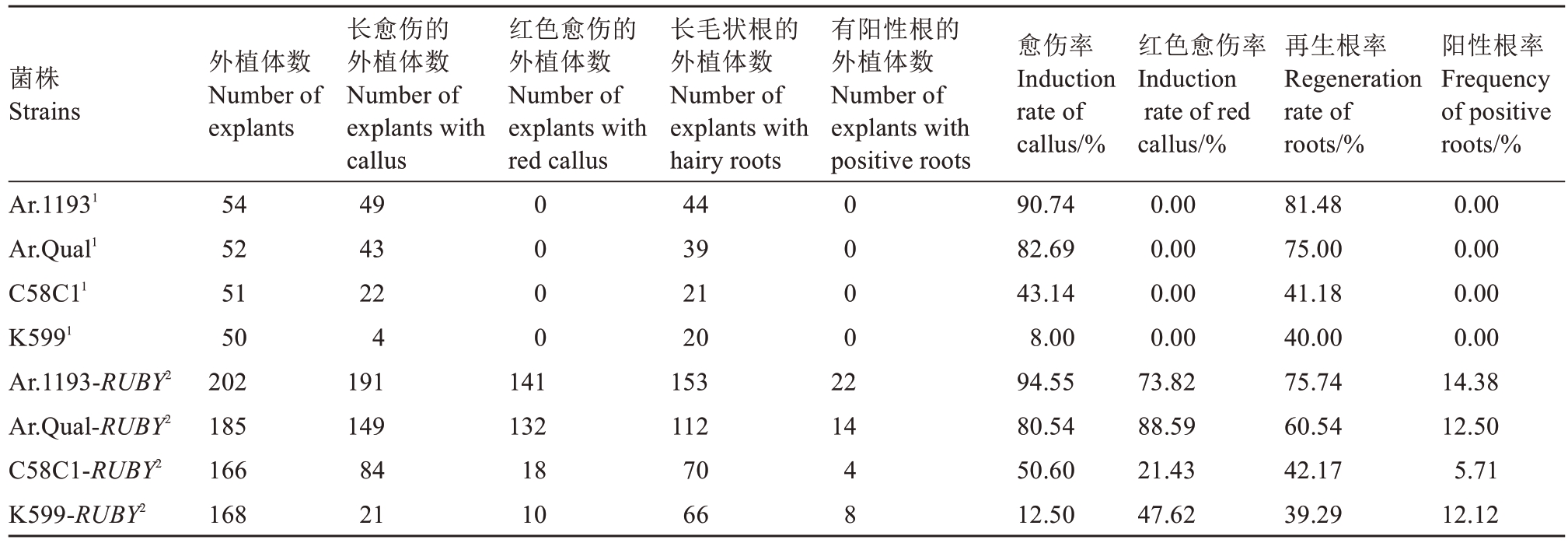
注:1 对照组. 不携带任何质粒的4 种菌株侵染的下胚轴;2 试验组. 携带35S::RUBY 质粒的4 种菌株侵染的下胚轴。
Note:1 Control groups. Hypocotyl infected by four strains that did not carry any plasmids. 2 Experimental groups. Hypocotyl infected by four strains that carry 35S::RUBY plasmid.
阳性根率Frequency of positive roots/%0.00 0.00 0.00 0.00 14.38 12.50 5.71 12.12菌株Strains外植体数Number of explants红色愈伤的外植体数Number of explants with red callus有阳性根的外植体数Number of explants with positive roots红色愈伤率Induction rate of red callus/%Ar.11931 Ar.Qual1 C58C11 K5991 Ar.1193-RUBY2 Ar.Qual-RUBY2 C58C1-RUBY2 K599-RUBY2 54 52 51 50 202 185 166 168长愈伤的外植体数Number of explants with callus 49 43 22 4 191 149 84 21 0 0 0 0 0 0 0 0 141 132 18 10长毛状根的外植体数Number of explants with hairy roots 44 39 21 20 153 112 70 66 22 14 4 8愈伤率Induction rate of callus/%90.74 82.69 43.14 8.00 94.55 80.54 50.60 12.50 0.00 0.00 0.00 0.00 73.82 88.59 21.43 47.62再生根率Regeneration rate of roots/%81.48 75.00 41.18 40.00 75.74 60.54 42.17 39.29
3 讨 论
在非组织培养方法层面,针对现有草莓毛状根诱导技术存在的问题,采取了系统化解决对策。与已报道的草莓体内注射诱导毛状根方法[15]相比,传统方法在匍匐茎茎部注射后40 d,毛状根在注射部位附近开始发育,在注射后60 d 毛状根大量增殖。此方法耗时较长,操作繁琐,茎段易折断,且只获得了50%的表皮隆起率,发根效率较低。此外,仅测试了K599 菌株,菌株单一。为此,笔者创新性地开发了基于匍匐茎子株划伤法的快速转化体系,在划伤部位涂抹菌体,进行扦插培养。该方法将培养周期缩短至20 d,效率提升了2~3 倍。此外,结合RUBY视觉报告基因,无需使用特殊设备即可肉眼观察,简化操作环节、节省成本。并且,经对4 种发根农杆菌(Ar.1193、Ar.Qual、C58C1、K599)进行系统比较,确定Ar.1193 为最优菌株,其转化效率高达79.41%,显著优于传统方法。
在组织培养方法层面,笔者利用发根农杆菌侵染下胚轴,相较于传统根癌农杆菌介导的叶盘法,该方法展现出显著优势:下胚轴再生根率最高可达75.74%,30 d即可获得转基因材料,相比于传统叶盘法周期缩短50%以上,转化效率提升4~5 倍[22]。此外,研究发现下胚轴侵染法可同时诱导产生转基因愈伤组织,这些呈现红色表型的愈伤组织证实了35S::RUBY 报告基因的成功整合与表达。值得注意的是,与根癌农杆菌介导的叶盘法相比,发根农杆菌诱导的愈伤组织形成周期显著缩短[7],表现出更高的转化效率,红色愈伤率最高可达88.59%,为草莓及其他蔷薇科作物的遗传转化和功能基因组研究提供了高效、普适的新技术体系。这不仅解决了传统叶盘法转化率低、周期长的问题,更为难转化植物的基因编辑和分子育种奠定了基础。
此外,众多研究表明,当植物受到创伤与植物生长调节剂刺激时,发育调节因子(developmental regulators,DRs)可以改变植物细胞发育进程,重新诱导分生组织形成,以此得到转基因幼芽进而获得转基因植株[23]。利用WUS、IPT、BBM、PLT、GRF、GIF 和STM 等DRs 可极大地改善植物体细胞胚胎的发生速度和再生能力,显著提高遗传转化效率[24]。目前,已有利用发根农杆菌携带发育调节因子侵染植物成功获得转基因植株的研究[25]。Wang 等[26]在白菜中已实现利用发根农杆菌共表达发育调节因子Zm-WUS2-IPT-AtPLT5复合体,获得了转基因植株,在白菜物种中打破了基因型的限制条件,建立了高效便捷的遗传转化系统。Yi等[27]在萝卜中通过发根农杆菌介导法,利用发育调节因子与RUBY 视觉报告基因,在不添加任何外源激素的情况下成功获得了转基因植株。对于发根农杆菌介导的草莓转基因体系的建立,可参考利用发育调节因子的方式。
4 结 论
通过匍匐茎子株划伤法、短缩茎穿刺法和下胚轴侵染法3种方法,结合35S::RUBY视觉报告基因,成功诱导二倍体森林草莓以及八倍体栽培草莓产生转基因阳性毛状根。研究结果表明,4种发根农杆菌(Ar.Qual、Ar.1193、C58C1、K599)在不同处理方法下均能诱导草莓外植体形成毛状根。其中Ar.1193菌株的诱导效果最为显著,毛状根阳性率最高可达79.41%。笔者筛选出最适宜的菌株,并建立了草莓高效、经济、快速的发根农杆菌转化体系,为草莓遗传转化研究提供了有力的技术支持,也为草莓的根系发育、CRISPR/Cas9基因编辑效率的快速验证奠定了基础。
[1] 罗学兵,贺良明. 草莓的营养价值与保健功能[J]. 中国食物与营养,2011,17(4):74-76.LUO Xuebing,HE Liangming. Nutritional value and health function of strawberry[J]. Food and Nutrition in China,2011,17(4):74-76.
[2] LIU Z C,LIANG T,KANG C Y. Molecular bases of strawberry fruit quality traits:Advances,challenges,and opportunities[J].Plant Physiology,2023,193(2):900-914.
[3] 武冲,姜莉莉,宗晓娟,王晓芳,田中一久. 中国草莓育种研究进展[J]. 落叶果树,2022,54(2):28-30.WU Chong,JIANG Lili,ZONG Xiaojuan,WANG Xiaofang,TIAN Zhongyijiu. Advances in strawberry breeding in China[J].Deciduous Fruits,2022,54(2):28-30.
[4] 邢思年. 草莓CRISPR/Cas9 基因组编辑技术体系建立和FaMAPK12 基因功能分析[D]. 北京:中国农业大学,2018.XING Sinian. Establishment of strawberry CRISPR/Cas9 genome editing technology system and analysis of FaMAPK12 gene function[D]. Beijing:China Agricultural University,2018.
[5] 乔瑞琪,金万梅,沈元月. 二倍体草莓‘Ruegen’再生体系的建立[J]. 北京农学院学报,2018,33(3):35-39.QIAO Ruiqi,JIN Wanmei,SHEN Yuanyue. Establishment of regeneration system using the leaves of diploid strawberry ‘Ruegen’[J]. Journal of Beijing University of Agriculture,2018,33(3):35-39.
[6] 陈赢男,胡传景,诸葛强,胡建军,尹佟明. 杨树农杆菌遗传转化研究30 年[J]. 林业科学,2022,58(12):114-129.CHEN Yingnan,HU Chuanjing,ZHUGE Qiang,HU Jianjun,YIN Tongming. Thirty years of Agrobacterium-mediated genetic transformation of Populus[J]. Scientia Silvae Sinicae,2022,58(12):114-129.
[7] 吴雪梅,汤浩茹. 草莓叶片培养研究进展[J]. 果树学报,2004,21(6):598-602.WU Xuemei,TANG Haoru. Advances on in vitro culture of strawberry (Fragaria ananassa) leaves[J]. Journal of Fruit Science,2004,21(6):598-602.
[8] 王翠华,刘荇,杜小云,曾弓剑,谭雅慧,周超,刘开鸿,沈祥陵.红颜草莓叶盘再生及遗传转化体系的优化[J]. 生物资源,2020,42(2):234-242.WANG Cuihua,LIU Xing,DU Xiaoyun,ZENG Gongjian,TAN Yahui,ZHOU Chao,LIU Kaihong,SHEN Xiangling. Optimization of leaf disc regeneration and genetic transformation system in Benihope strawberry[J]. Biotic Resources,2020,42(2):234-242.
[9] 李鹏举,宋鹏慧,段亚东,杨光,周双,张鹍,房磊. 草莓遗传育种中的生物技术的应用[J]. 现代农业研究,2021,27(8):121-122.LI Pengju,SONG Penghui,DUAN Yadong,YANG Guang,ZHOU Shuang,ZHANG Kun,FANG Lei. Application of biotechnology in strawberry genetics and breeding[J]. Modern Agriculture Research,2021,27(8):121-122.
[10] 汪虞骅,黄思远,郝雪婷,金朝辉,张清虹,赵荣. 植物毛状根研究与应用进展[J]. 现代农业科技,2024(22):138-142.WANG Yuhua,HUANG Siyuan,HAO Xueting,JIN Zhaohui,ZHANG Qinghong,ZHAO Rong. Research and application progress on plant hairy roots[J]. Modern Agricultural Science and Technology,2024(22):138-142.
[11] 曹冬梅,韩振海,许雪峰. 发根农杆菌Ri 质粒研究进展[J]. 中国生物工程杂志,2003,23(2):74-78.CAO Dongmei,HAN Zhenhai,XU Xuefeng. Research progress of Agrobacterium rhizogenes[J]. Journal of Chinese Biotechnology,2003,23(2):74-78.
[12] 舒铫,杨嵩涵,孔可心,吕若涵,吕桂云,张春秋,宋士清. 发根农杆菌介导的甜瓜转化体系的建立及对CRISPR/Cas9 靶位点的快速检测[J]. 农业生物技术学报,2025,33(4):924-932.SHU Yao,YANG Songhan,KONG Kexin,LÜ Ruohan,LÜ Guiyun,ZHANG Chunqiu,SONG Shiqing. Establishment of Agrobacterium rhizogenes-mediated transformation system in melon (Cucumis melo) and rapid detection of CRISPR/Cas9 target sites[J].Journal of Agricultural Biotechnology,2025,33(4):924-932.
[13] 邬婷婷,刘凉琴,姜燕琴,葛春峰,田亮亮,刘梦溪,曾其龙,於虹. 发根农杆菌介导的蓝莓遗传转化体系的建立[J/OL]. 果树学报,2025:1-12(2025-04-27). https://doi.org/10.13925/j.cnki.gsxb.20250077.WU Tingting,LIU Liangqin,JIANG Yanqin,GE Chunfeng,TIAN Liangliang,LIU Mengxi,ZENG Qilong,YU Hong. Construction of genetic transformation system mediated by Agrobacterium rhizogenes in Vaccinium corymbosum[J/OL]. Journal of Fruit Science,2025:1-12(2025-04-27). https://doi.org/10.13925/j.cnki.gsxb.20250077.
[14] YAN H Q,MA D D,YI P P,SUN G L,CHEN X Y,YI Y,HUANG X L. Highly efficient Agrobacterium rhizogenes-mediated transformation for functional analysis in woodland strawberry[J]. Plant Methods,2023,19(1):99.
[15] HAO F L,WU S X,SHEN Z Y,TANG M T,GE X J,WU M Q,SUN Q H,FANG C B. Establishment of an in vivo transgenic hairy root system in strawberry for verifying the nitrate-transport activity of FaNRT1.1[J/OL]. Horticultural Plant Journal,2024(2024-12-18). https://doi.org/10.1016/j.hpj.2024.11.001.
[16] 李雯静,李旭彤,辛同旭,柴森,王帅,杨学勇. RUBY 无创筛选系统在黄瓜遗传转化中的应用[J]. 中国蔬菜,2025(3):46-54.LI Wenjing,LI Xutong,XIN Tongxu,CHAI Sen,WANG Shuai,YANG Xueyong. Application of RUBY noninvasively selection system in cucumber genetic transformation[J]. China Vegetables,2025(3):46-54.
[17] HE Y B,ZHANG T,SUN H,ZHAN H D,ZHAO Y D. A reporter for noninvasively monitoring gene expression and plant transformation[J]. Horticulture Research,2020,7:152.
[18] 王中一,刘熠,胡博文,朱凡,刘峰,杨莎,熊程,欧立军,戴雄泽,邹学校. 基于RUBY 及CaREF1 的辣椒高效遗传转化体系构建[J]. 园艺学报,2025,52(4):1093-1104.WANG Zhongyi,LIU Yi,HU Bowen,ZHU Fan,LIU Feng,YANG Sha,XIONG Cheng,OU Lijun,DAI Xiongze,ZOU Xuexiao. Construction of a high-efficiency genetic transformation system in pepper leveraging RUBY and CaREF1[J]. Acta Horticulturae Sinica,2025,52(4):1093-1104.
[19] 胡晓. 矮牵牛PMADS18 和PMADS9 基因的定点敲除及RUBY 标记基因在矮牵牛中的应用[D]. 重庆:西南大学,2022.HU Xiao. Site-directed knockout of PMADS18 and PMADS9 genes and application of RUBY marker gene in Petunia hybrida[D]. Chongqing:Southwest University,2022.
[20] 苑忠杰. RNAi 技术在柠檬抗CYVCV 育种中的应用及柠檬遗传转化体系的建立[D]. 重庆:西南大学,2024.YUAN Zhongjie. Application of RNAi technology in breeding of lemon resistant to CYVCV and establishment of genetic transformation system of lemon[D]. Chongqing:Southwest University,2024.
[21] ROGERS S O,BENDICH A J. Extraction of DNA from milligram amounts of fresh,herbarium and mummified plant tissues[J]. Plant Molecular Biology,1985,5(2):69-76.
[22] FOLTA K M,DAVIS T M. Strawberry genes and genomics[J].Critical Reviews in Plant Sciences,2006,25(5):399-415.
[23] 马曦,张金睿,庄红梅,陈巍,张蜜,袁晓琪,易小芳,王聪聪,王海云,王燕. 发根农杆菌介导的芜菁高效遗传转化体系建立[J/OL]. 园艺学报,2025:1-10(2025-05-07). https://doi.org/10.16420/j.issn.0513-353x.2025-0140.MA Xi,ZHANG Jinrui,ZHUANG Hongmei,CHEN Wei,ZHANG Mi,YUAN Xiaoqi,YIXiaofang ,WANG Congcong,WANG Haiyun,WANG Yan. Establishment of an efficient genetic transformation system mediated by Agrobacterium rhizogenes in turnip[J/OL]. Acta Horticulturae Sinica,2025:1-10(2025-05-07). https://doi.org/10.16420/j.issn.0513-353x.2025-0140.
[24] 谢盈盈,王克剑,饶玉春,黄勇. 发育调节因子助力作物遗传转化效率提升[J/OL]. 遗传,2025:1-21(2025-04-21). http://kns.cnki.net/kcms/detail/11.1913.r.20250418.1608.002.html.XIE Yingying,WANG Kejian,RAO Yuchun,HUANG Yong.Developmental regulatory factors promote the efficiency of cropgenetic transformation[J/OL]. Hereditas (Beijing),2025:1- 21(2025- 04- 21). http://kns.cnki.net/kcms/detail/11.1913.r.20250418.1608.002.html.
[25] 王盈润,郑仲辰,JUDITH M,何轶洋,李杰勤,郑甲成. 发育调控因子提高植物遗传转化效率的研究进展[J]. 河南科技学院学报(自然科学版),2025,53(1):23-30.WANG Yingrun,ZHENG Zhongchen,JUDITH M,HE Yiyang,LI Jieqin,ZHENG Jiacheng. Improvement progress of plant genetic transformation efficiency by the developmental regulatory factors[J]. Journal of Henan Institute of Science and Technology(Natural Science Edition),2025,53(1):23-30.
[26] WANG Y L,YANG X D,WANG W L,WANG Y,CHEN X S,WU H,GAO Z Y,XU H H,LIU T K,LI Y,XIAO D,LIU W S,HOU X L,ZHANG C W. Efficient genetic transformation and gene editing of Chinese cabbage using Agrobacterium rhizogenes[J].Plant Physiology,2025,197(2):kiae543.
[27] YI X F,WANG C C,YUAN X Q,ZHANG M,ZHANG C W,QIN T J,WANG H Y,XU L,LIU L W,WANG Y. Exploring an economic and highly efficient genetic transformation and genome-editing system for radish through developmental regulators and visible reporter[J]. The Plant Journal,2024,120(4):1682-1692.