土壤盐碱化是全球农业可持续发展面临的重大挑战。研究表明,全球约13.81 亿万hm2土地受到盐碱化影响。中国盐碱地面积约为1亿万hm2,主要分布在西北内陆和东部沿海地区[1]。盐碱胁迫通过离子毒害(Na+/K+失衡)、渗透胁迫(水分吸收受限)与氧化损伤(活性氧积累)等多重机制抑制植物生长[2-3]。其中,高浓度Na+不仅破坏细胞膜完整性,干扰K+吸收,导致离子稳态失衡,还会诱导活性氧(ROS)过量积累,引发脂质过氧化与蛋白质损伤[4-5]。苹果(Malus domestica)作为我国主要的经济果树,其在盐碱胁迫下常表现为根系发育受阻、生物量降低及离子失衡(Na+过度积累、K+外流)[6-7]。苹果砧木M9-T337 因具有矮化特性广泛用于集约化栽培,但该品种对盐碱胁迫耐受性较弱,叶片黄化、光合效率下降及氧化损伤等问题严重制约盐碱地苹果建园的整齐性以及后续的生产力[8]。
丛枝菌根真菌(arbuscular mycorrhizal fungi,AMF)作为与植物共生的关键微生物,其菌丝网络可帮助宿主植物扩大养分吸收范围,并通过调控离子转运蛋白(如SOS1、HKT1)表达,减少Na+向地上部运输,同时激活SOD、POD、CAT 等抗氧化酶系统以清除ROS[9-11]。研究表明,AMF 在单一盐或碱胁迫下通过增强渗透调节能力、提高抗氧化酶活性、诱导AsA-GSH 循环相关基因表达等方式提高植物的抗逆性[12-13]。例如,在辣椒(Capsicum annuum L.)中,AMF 处理可以增加K+的积累,减少Na+和Cl-的积累,从而减轻离子失衡[14]。在杨树(Populus spp.)中,AMF 可以提高蒸腾速率,有效缓解植株水分流失,显著增加幼苗生物量。同时,AMF 能够通过增强抗氧化酶(SOD、POD、CAT)系统活性、提高活性氧清除效率,从而抑制脯氨酸(Pro)与丙二醛(MDA)的过度积累,进而维持细胞膜的结构完整性与功能稳定性[15]。AMF在不同逆境下的应用均有相关报道,且多见于彩叶草[16]、棉花[17]、番茄[18]等植物,而在苹果砧木M9-T337 中的应用却鲜有报道,亟须研究其在苹果砧木中是否也具有相同的作用机制。
基于此,以苹果砧木M9-T337为试材,通过对比分析植株生长表型、Na+/K+分布及抗氧化系统动态变化,从离子平衡(Na+/K+吸收)与氧化平衡(抗氧化酶系统及AsA-GSH 循环)的双视角,系统解析AMF缓解盐碱胁迫的生理机制。笔者首次提出并验证以下科学假设:AMF 通过协同调控SOS 信号通路(促进Na+外排)和AsA-GSH 循环(增强ROS 清除能力),打破盐碱胁迫下“离子毒害-氧化损伤”恶性循环,从而增强苹果砧木耐盐碱性。该研究结果将为AMF 增强果树盐碱抗性的生理机制提供理论依据,并为盐碱地区苹果园生物改良技术开发奠定基础。
1 材料和方法
1.1 材料
1 年生苹果砧木M9-T337 幼苗购自山东惠农园艺有限公司,统一放置于甘肃农业大学温室进行管理,后续进行试验处理。
丛枝菌根真菌幼套近明球囊霉(Claroideoglomus etunicatum,Ce,BGC G203C)购自中国丛枝菌根种质资源库(BGC)。2023 年11 月以白三叶草(Trifolium repens L.)作为宿主植物进行扩繁。培养3 个月后,收获盆中所有培养物(孢子、菌丝、根段和栽培基质)作为菌剂,平均活性孢子含量为40个·g-1。
1.2 试验设计
试验采用单因素完全随机区组设计。共设置对照(清水灌溉,CK)、盐碱胁迫(200 mmol·L-1 NaCl∶NaHCO2=1∶1,pH=8.36,电导率为0.42 mS·cm-1,SA)和在盐碱胁迫基础上接种丛枝菌根真菌(Ce,SA+AMF)3 个处理。每个处理设置3 次生物学重复,每个重复10株幼苗,共90株幼苗。试验所用M9-T337幼苗均在直径25 cm、深度38 cm 的花盆(移栽前喷洒75%乙醇消毒)中栽培,每盆移栽1 株。每盆基质为2.50 kg,基质由土壤、珍珠岩与蛭石按体积比3∶1∶1 混合配制,121 ℃高压灭菌2 h。接种AMF 处理的接种量为30 g 菌剂。菌剂的具体接种过程为:将灭菌后的基质填充至花盆约1/3 深度后,将30 g 菌剂均匀铺撒于表面,在菌剂表面均匀铺撒一层基质,然后将幼苗移栽至花盆中,继续填充基质至花盆约3/4深度。接种AMF 45 d后进行盐碱胁迫处理。每6 d浇灌一次盐碱混合液(300 mL),共浇灌3次,其余时间则按相同量浇水以保持浓度一致。对照组则浇灌相同量清水。为了保证盐碱胁迫环境的稳定性,盆下配有托盘,以便将渗出的盐碱溶液倒回花盆内。自盐碱胁迫处理时开始计时,第30天采集幼苗中上部分的功能叶片。一部分鲜样用锡纸包裹后液氮冷冻,放置于-80 ℃超低温冰箱,用于后续生理指标测定;另一部分称量地上部及地下部鲜质量,然后装入信封袋,105 ℃杀青30 min,75 ℃烘干至恒质量,再称量记录各部分的干质量,粉碎待用。
1.3 测定指标与方法
1.3.1 表型观察 于胁迫后第30 天,每个处理组随机选取1 株生长状况相似度达80%的幼苗进行表型拍照。
1.3.2 生物量测定 于胁迫后第30 天,每个处理组随机选择3株幼苗,先用清水将根系冲洗干净,再将地上部分和地下部分完整分开并称量其鲜质量,然后置于105 ℃烘箱杀青30 min,75 ℃烘干至恒质量,再称量记录各部分的干质量。最后根据得到的数据计算出各处理的根冠比(R/S)和叶片相对含水量(WC)。
1.3.3 生理指标测定 超氧化物歧化酶(SOD)活性测定采用NBT 法[19];过氧化物酶(POD)活性测定采用愈创木酚法[20];过氧化氢酶(CAT)活性测定采用过氧化氢氧化法[21];过氧化氢(H2O2)含量、超氧阴离子(O2-·)产生速率、还原型抗坏血酸(AsA)含量、脱氢抗坏血酸(DHA)含量、谷胱甘肽(GSH)含量、还原氧化型谷胱甘肽(GSSG)含量、抗坏血酸氧化酶(AAO)活性、谷胱甘肽还原酶(GR)活性、坏血酸过氧化物酶(APX)活性、氢抗坏血酸还原酶(MDHAR)活性均采用试剂盒(苏州科铭生物技术有限公司)测定。
1.3.4 Na+和K+含量测定 Na+和K+含量测定参考Kamble 等[22]的方法。首先,将叶片干燥并研磨,准确称取5 g 粉末干样品,用H2SO4-H2O2进行消煮,然后用火焰光度计测定Na+和K+含量。
1.3.5 盐碱胁迫相关响应基因的实时荧光定量PCR(qRT-PCR)分析 基于前期研究报道的苹果盐碱胁迫响应机制[23-24],选取钠钾平衡及抗氧化系统相关基因进行qRT-PCR 分析。针对钠钾平衡,测定Md-SOS2 和MdSOS3(盐过敏感基因,调控Na+外排)、MdGORK1(高亲和性K+转运蛋白,介导K+吸收与Na+/K+选择性运输)及MdNHX2(液泡膜Na+/H+逆向转运蛋白,促进Na+液泡区隔化)的表达水平;针对抗氧化系统,测定MdAREBI 和MdSOD1(ROS 清除)的基因表达水平。选取3 组处理植株的叶片,采用TRIzol 法(Invitrogen,USA)提取总RNA,经Prime-Script RT试剂盒(TaKaRa,Japan)反转录为cDNA,通过NCBI Primer-BLAST 设计特异性引物(表1),以MdActin为内参[23],按照AG试剂的说明书进行操作。
表1 qRT-PCR 引物
Table 1 qRT-PCR primer
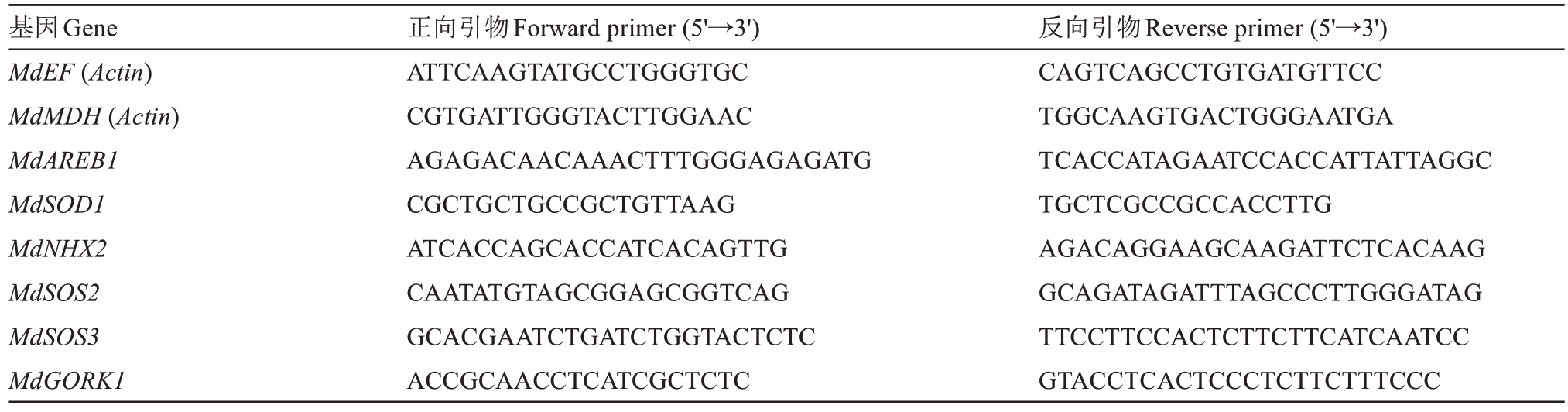
基因Gene MdEF (Actin)MdMDH (Actin)MdAREB1 MdSOD1 MdNHX2 MdSOS2 MdSOS3 MdGORK1正向引物Forward primer (5'→3')ATTCAAGTATGCCTGGGTGC CGTGATTGGGTACTTGGAAC AGAGACAACAAACTTTGGGAGAGATG CGCTGCTGCCGCTGTTAAG ATCACCAGCACCATCACAGTTG CAATATGTAGCGGAGCGGTCAG GCACGAATCTGATCTGGTACTCTC ACCGCAACCTCATCGCTCTC反向引物Reverse primer (5'→3')CAGTCAGCCTGTGATGTTCC TGGCAAGTGACTGGGAATGA TCACCATAGAATCCACCATTATTAGGC TGCTCGCCGCCACCTTG AGACAGGAAGCAAGATTCTCACAAG GCAGATAGATTTAGCCCTTGGGATAG TTCCTTCCACTCTTCTTCATCAATCC GTACCTCACTCCCTCTTCTTTCCC
1.4 数据处理
采用Microsoft Office Excel 2019处理数据,使用Origin 2022绘制图表。统计分析使用IBM SPSS Statistics 25。采用方差分析(ANOVA)比较各处理组间的均值,并用邓肯检验确定差异显著性(P<0.05)。
2 结果与分析
2.1 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗生长表型的影响
由图1可知,在盐碱胁迫30 d后,单一胁迫(SA)处理的M9-T337 幼苗的生长状况明显差于CK,具体表现为植株中部叶片边缘出现黄化及枯萎现象。相反,SA+AMF 处理的M9-T337 幼苗仅底部叶片出现黄化,且未出现边缘枯萎现象。

图1 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗生长表型的影响
Fig. 1 Effects of arbuscular mycorrhizal fungi on the growth of apple rootstock M9-T337 seedlings under saline-alkali stress
2.2 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗生物量的影响
由图2 可知,盐碱胁迫30 d 后,SA 处理的M9-T337 幼苗的地上和地下部分的干、鲜质量均较CK显著下降,干质量降幅分别为25.45%和39.15%,鲜质量降幅分别为35.80%和44.37%。而SA+AMF 处理的植株地上和地下部分的干、鲜质量均显著高于SA处理,干质量较SA处理分别升高10.78%和38.91%,鲜质量较SA处理分别升高17.13%和30.06%。
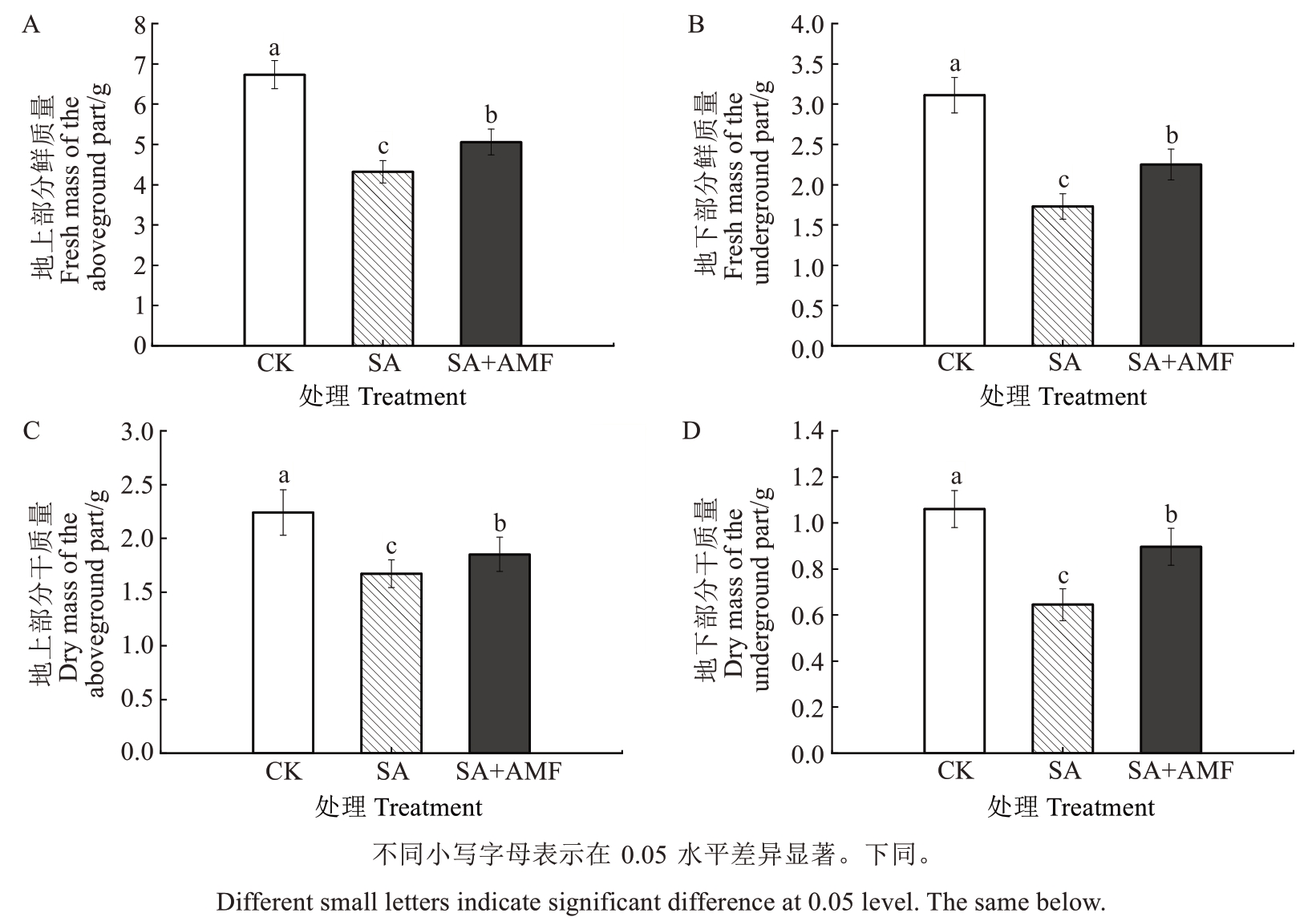
图2 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗生物量的影响
Fig. 2 Effects of arbuscular mycorrhizal fungi on biomass of apple rootstock M9-T337 seedlings under saline-alkali stress
2.3 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗地上部生长指标的影响
由图3 可知,SA 处理显著抑制了M9-T337 幼苗的生长。具体表现为茎粗、叶面积和株高增长量及叶片相对含水量均显著低于CK,降幅分别为66.67%、66.84%、51.65%和35.00%。值得注意的是,SA+AMF 处理的茎粗、叶面积和株高增长量及叶片相对含水量均显著高于SA 处理。其中SA+AMF 处理的株高增长量的增幅最大,为86.38%。

图3 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗生长指标的影响
Fig. 3 Effects of arbuscular mycorrhizal fungi on growth index of apple rootstock M9-T337
seedlings under saline-alkali stress
2.4 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗叶片抗氧化酶活性的影响
由图4 可知,SA 处理的M9-T337 幼苗叶片的SOD、POD和CAT活性均较CK显著升高,增幅分别为12.85%、19.21%和17.89%。值得注意的是,SA+AMF 处理叶片的SOD、POD 和CAT 表现出更高的活性,与SA 处理相比,分别显著提高30.45%、38.87%和19.35%。
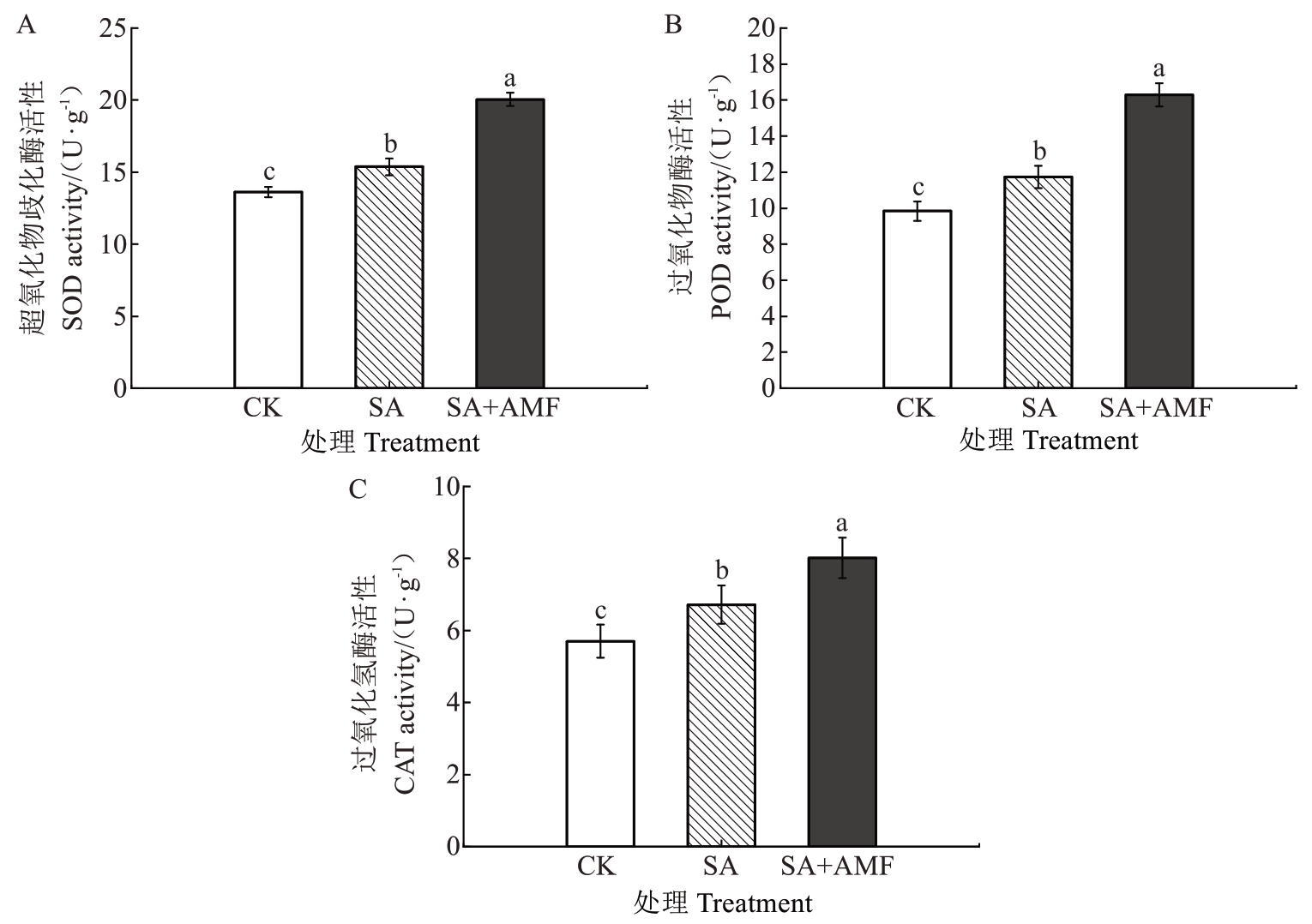
图4 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗叶片抗氧化酶活性的影响
Fig. 4 Effects of arbuscular mycorrhizal fungi on antioxidant enzyme activities in leaves of apple rootstock M9-T337 seedlings under saline-alkali stress
2.5 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗叶片ROS含量的影响
由图5 可知,SA 处理M9-T337 幼苗叶片的H2O2含量和O2-·产生速率均较CK 显著升高,分别为CK的2.03 倍和2.39 倍。SA+AMF 处理的叶片H2O2含量和O2-·产生速率均较SA 处理呈现不同程度的下降,但仍显著高于CK,分别较SA 处理显著下降18.09%和27.03%。
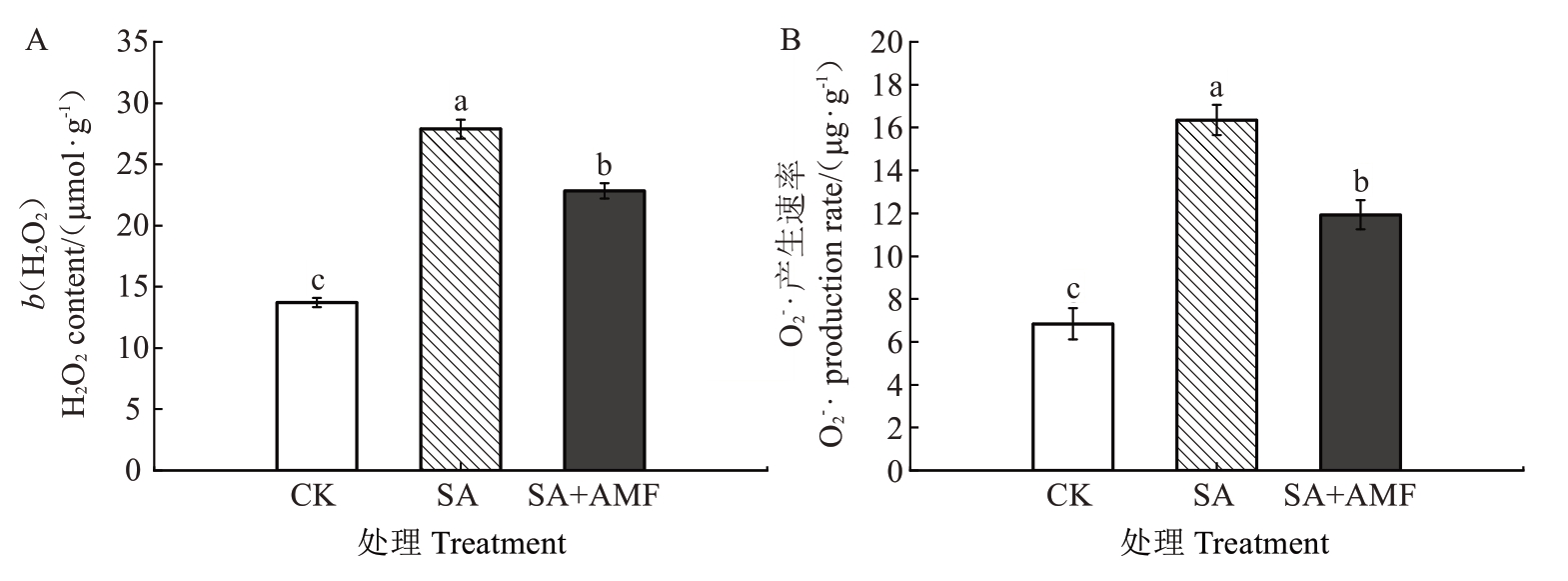
图5 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗叶片ROS 含量的影响
Fig. 5 Effects of arbuscular mycorrhizal fungi on ROS content in leaves of apple rootstock M9-T337 seedlings under saline-alkali stress
2.6 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗叶片AsA-GSH循环的影响
2.6.1 AsA-GSH 循环相关酶活性 由图6 可知,SA处理的M9-T337 幼苗叶片的APX、GR 和MDHAR活性均较CK 呈现不同程度的升高,其中GR 活性增幅最大,为142.11%。而SA+AMF处理的APX、GR和MDHAR 活性较SA 处理显著升高,分别较SA 处理升高48.89%、21.38%和70.27%。值得注意的是,SA处理的M9-T337幼苗叶片的AAO活性较CK显著降低,降幅为70.83%,而SA+AMF 处理的叶片AAO 活性显著高于SA处理,但较CK显著降低26.39%。
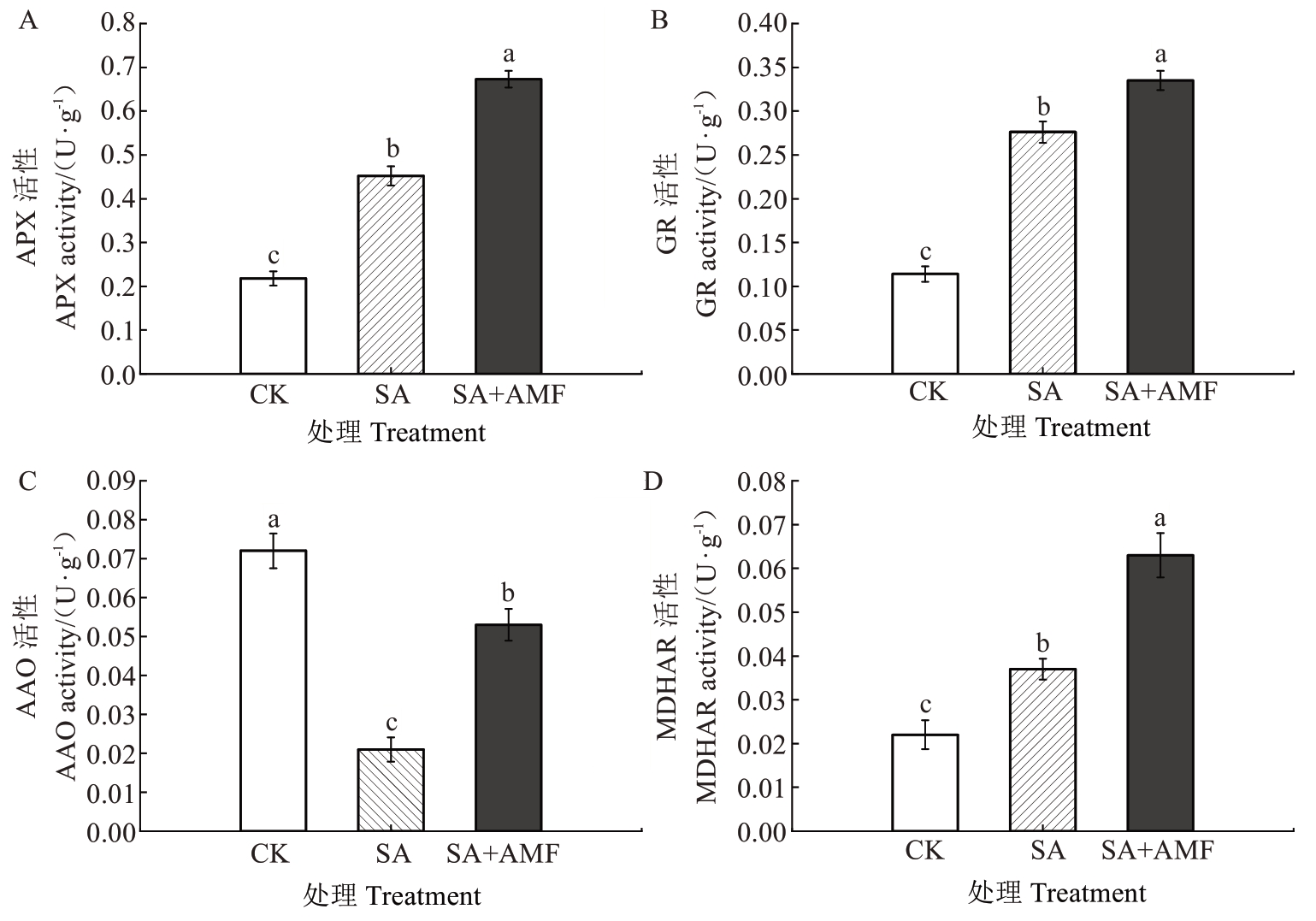
图6 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗叶片AsA-GSH 循环相关酶活性的影响
Fig. 6 Effects of arbuscular mycorrhizal fungi on AsA-GSH cycle-related enzyme activities in leaves of apple
rootstock M9-T337 seedlings under saline-alkali stress
2.6.2 AsA-GSH 循环相关物质含量 由图7 可知,SA 处理的M9-T337 幼苗叶片的AsA 和GSH 含量均下降,较CK 分别显著降低29.09%和45.71%。SA+AMF 处理叶片的AsA 和GSH 含量显著低于CK,但显著高于SA 处理,分别比CK 显著下降29.09%和9.29%。与此相反的是,SA 处理叶片的DHA 和GSSG 含量均较CK 显著升高,分别为CK 的1.69 倍和1.68倍,而SA+AMF处理叶片的DHA和GSSG含量均显著低于SA 处理,分别较SA 处理显著下降10.40%和12.12%。
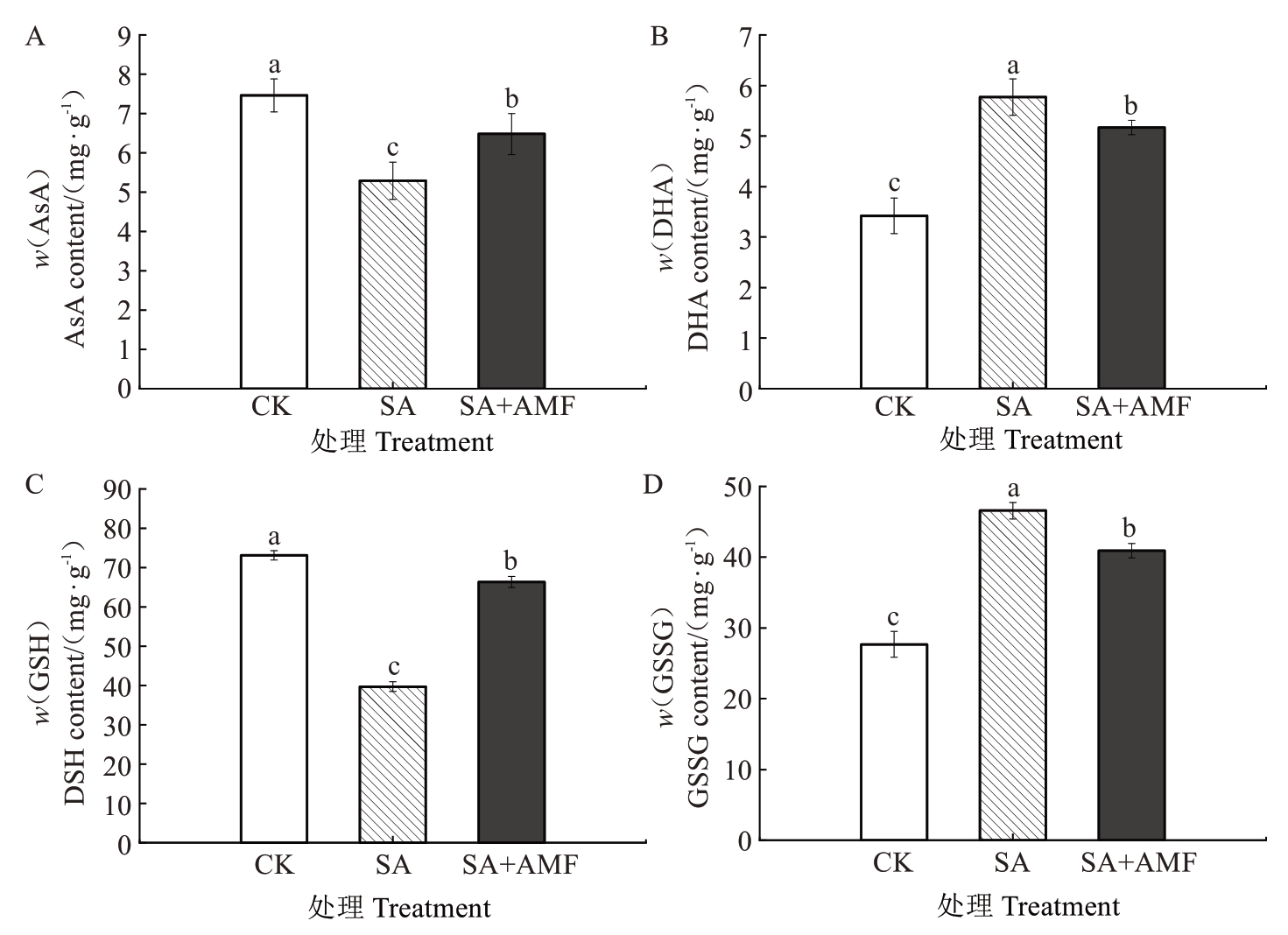
图7 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337 幼苗叶片AsA-GSH 循环相关物质含量的影响
Fig. 7 Effects of arbuscular mycorrhizal fungi on the content of AsA-GSH cycle-related substances in leaves of apple rootstock M9-T337 seedlings under saline-alkali stress
2.7 丛枝菌根真菌对盐碱胁迫下苹果砧木M9-T337幼苗叶片Na+和K+含量的影响
由图8 可知,SA 处理显著降低了M9-T337 幼苗叶片的K+含量,较CK 下降40.05%。相比之下,SA+AMF 处理叶片的K+含量较SA 显著升高,增幅为15.40%。值得注意的是,SA 处理叶片的Na+和Na+/K+均较CK 呈现不同程度的升高,增幅分别为90.41%和217.59%。与SA处理相比,SA+AMF处理显著降低了叶片的Na+和Na+/K+,降幅分别为18.02%和28.96%。
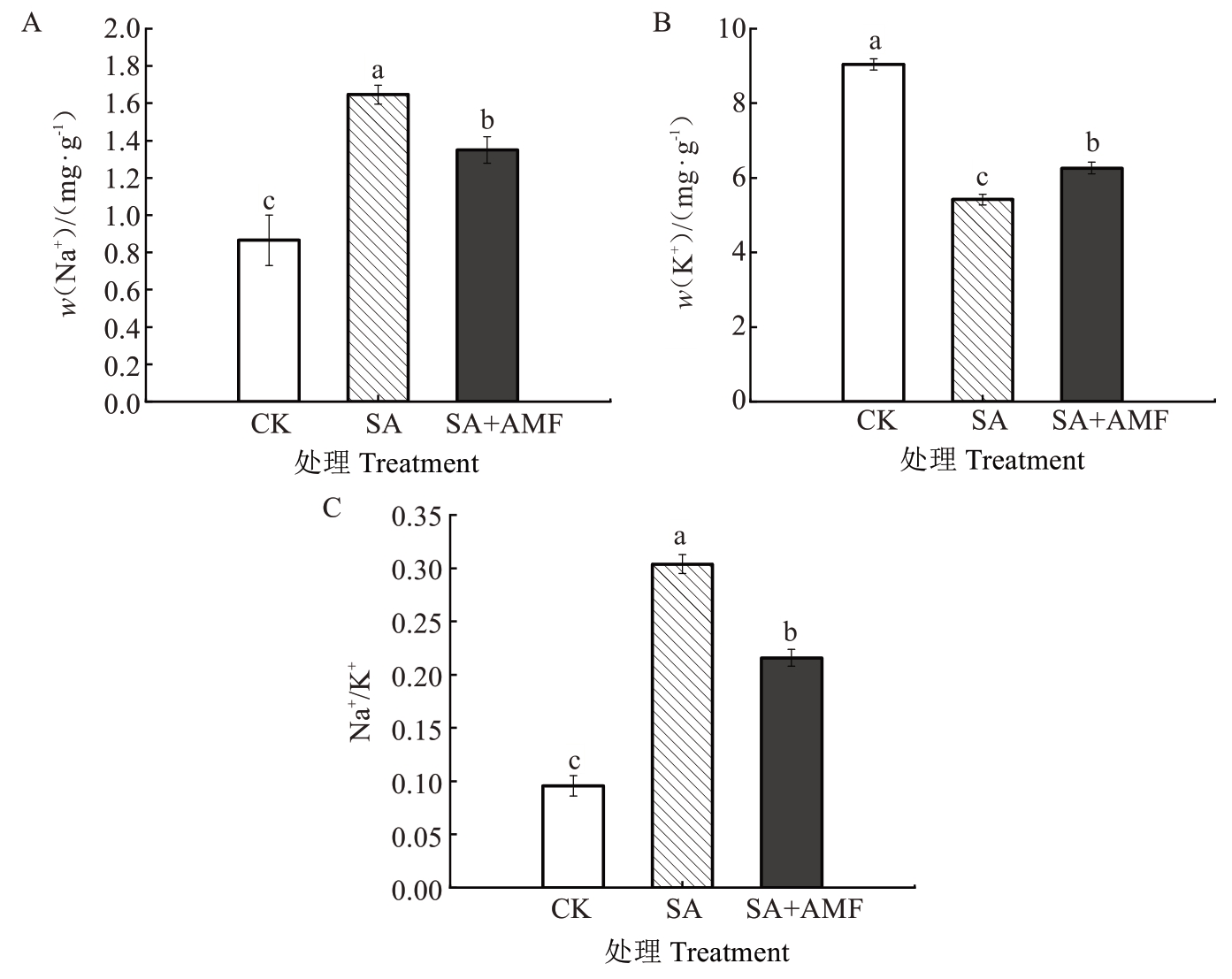
图8 丛枝菌根真菌对盐碱胁下苹果砧木M9-T337 幼苗叶片Na+和K+含量的影响
Fig. 8 Effects of arbuscular mycorrhizal fungi on Na + and K + content in leaves of apple rootstock M9-T337 seedlings under saline-alkali stress
2.8 丛枝菌根真菌对苹果砧木M9-T337 幼苗盐碱胁迫应答基因表达的影响
由图9 可知,盐碱胁迫对钠钾平衡以及ROS 清除相关基因的表达产生显著影响。与CK 相比,SA处理下MdAREB1、MdNHX2、MdSOS2、MdSOS3 和MdGORK1的表达量显著升高,分别为CK的1.65倍、6.43 倍、2.31 倍、3.27 倍和8.82 倍。相比之下,SA+AMF处理下MdAREB1、MdNHX2、MdSOS2、MdSOS3和MdGORK1 的表达量均较SA 处理显著升高,分别为SA 处理的1.38 倍、1.22 倍、1.15 倍、1.49 倍和1.23倍。值得注意的是,MdSOD1 的表达量呈现出相反的变化趋势,SA 处理下MdSOD1 表达量仅为CK 的45.00%,而在AMF 预处理下MdSOD1 表达量较SA处理显著升高,增幅为73.33%,但仍显著低于CK。
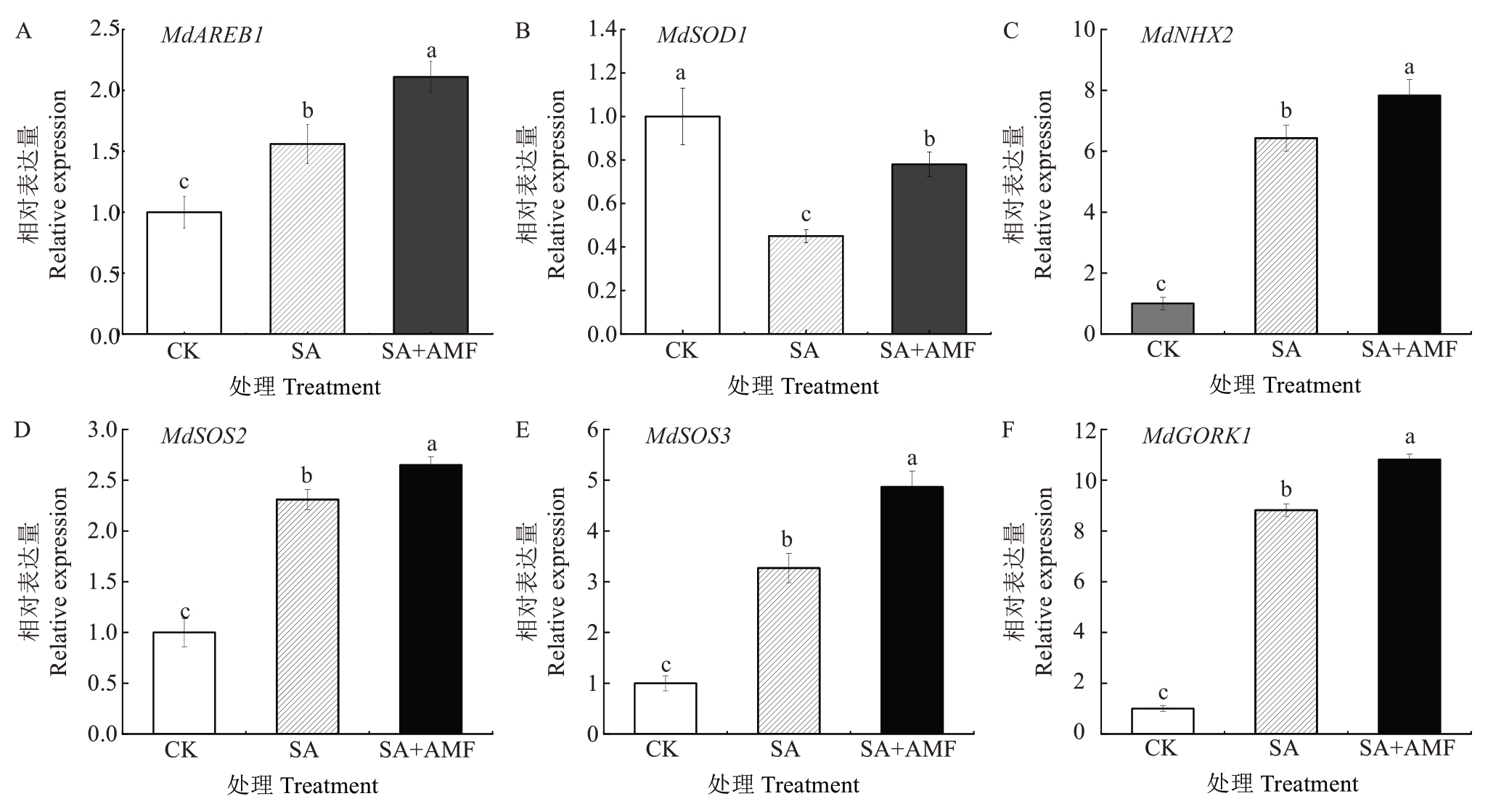
图9 丛枝菌根真菌对苹果砧木M9-T337 幼苗盐碱胁迫应答基因表达的影响
Fig. 9 Effects of arbuscular mycorrhizal fungi on the expression of saline-alkali stress response genes in apple rootstock M9-T337 seedlings
2.9 AMF 对盐碱胁迫下苹果砧木M9-T337 幼苗生理效应的综合评价
将处理后M9-T337 幼苗叶片的27 个生理指标进行相关性分析(图10)。结果表明,M9-T337 幼苗叶片的K+含量与SDI呈极显著正相关(P<0.01),与LAI呈显著正相关(P<0.05),与DHA 和GSSG 含量呈显著负相关(P<0.05);Na+含量与H2O2含量呈显著正相关;GR 活性与MdNHX2、MdSOS2 和Md-GOPK1的表达水平呈显著正相关(P<0.05)。
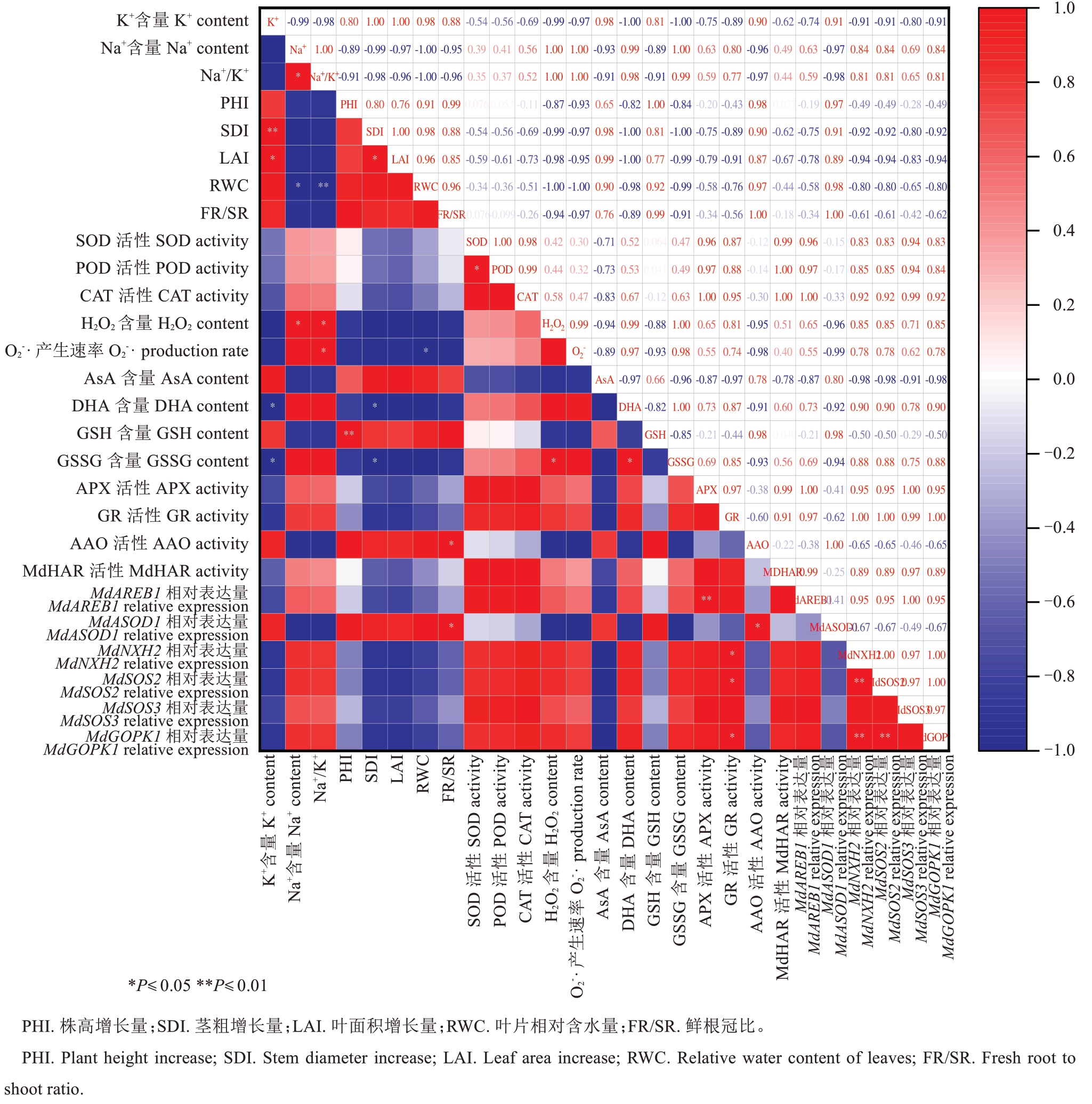
图10 相关性分析
Fig. 10 Correlation analysis
为综合评价盐碱胁迫下M9-T337 幼苗对AMF的生理响应特性,将27个指标进行主成分分析。提取特征值大于1 的2 个主成分,累计方差贡献率达98.688%,达到分析要求(表2)。将提取的2 个主成分的得分值代入综合评价函数F=F1×78.556%+F2×20.132%,计算每个处理的综合得分(表3)。其中,SA+AMF 处理综合得分最高,为0.590 7。因此,AMF 对盐碱胁迫下M9-T337 幼苗具有较好的缓解效应。
表2 主成分及方差贡献率
Table 2 Principal component and variance contribution rate
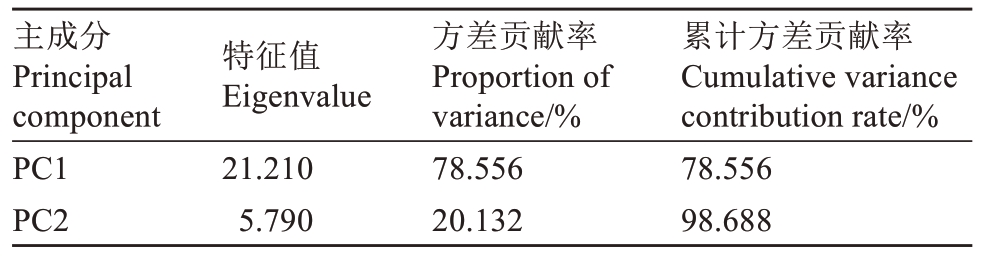
主成分Principal component PC1 PC2特征值Eigenvalue 21.210 5.790方差贡献率Proportion of variance/%78.556 20.132累计方差贡献率Cumulative variance contribution rate/%78.556 98.688
表3 综合得分排名
Table 3 Comprehensive score ranking
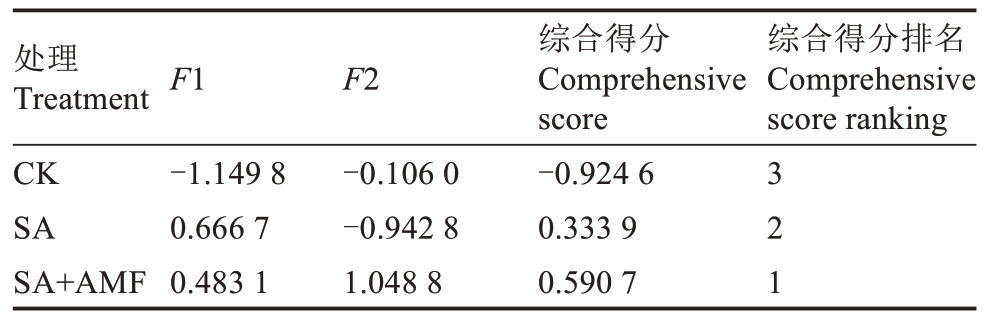
处理Treatment CK SA SA+AMF F1 F2综合得分排名Comprehensive score ranking-1.149 8 0.666 7 0.483 1-0.106 0-0.942 8 1.048 8综合得分Comprehensive score-0.924 6 0.333 9 0.590 7 3 2 1
3 讨 论
植物生物量、根冠比及光合效率是衡量抗盐碱性的核心表型指标,直接反映植物对胁迫的适应能力[25]。生物量的维持表明碳同化与分配的协调性,根冠比的优化有助于平衡水分吸收与地上部生长,而光合效率的稳定性则是逆境下能量供应的保障[26]。研究结果表明,SA 处理的M9-T337幼苗的地上部和根系生物量相较CK 显著下降,而AMF 预处理显著提升了M9-T337 幼苗的地上部和根系生物量,与Han 等[15]提出的“菌根依赖性生物量补偿效应”理论相符。SA处理的M9-T337幼苗中部叶片边缘出现黄化及枯萎现象,而SA+AMF 处理的幼苗仅有底部叶片黄化,无边缘枯萎现象,这可能是菌根共生通过调控叶绿体发育相关基因的表达实现光合修复[27],从而改善了盐碱胁迫下M9-T337 幼苗的生长状况。值得注意的是,SA+AMF 处理幼苗的根冠比较SA 处理显著提升(19.3%),表明AMF 可能通过调节碳素的分配格局,在一定程度上增强了根部作为同化物“库”的能力,同时降低了叶片作为“源”的负担,从而维持了植物源-库之间的动态平衡,促进了盐碱胁迫下M9-T337幼苗的生长发育[27]。进一步分析生长指标发现,SA处理显著抑制了M9-T337幼苗的生长,其茎粗、叶面积和株高增长量及根冠比、叶片相对含水量均显著低于CK,而SA+AMF 处理下的这些生长指标均显著高于SA 处理。这些结果充分表明,AMF 通过多方面的调节作用,显著增强苹果砧木M9-T337 幼苗在盐碱胁迫下的生长性能和耐逆性。
抗氧化酶(SOD、POD、CAT)活性与AsA-GSH循环是植物清除ROS 的关键防线,其协同效率决定了膜脂过氧化(MDA 含量)和氧化损伤的严重程度[28]。酶促系统直接中和ROS,而非酶促系统通过维持抗坏血酸-谷胱甘肽的动态平衡修复氧化损伤[29]。笔者在本研究中发现,AMF 显著增强M9-T337 幼苗的抗氧化酶活性。具体而言,AMF 显著提升了SOD、POD 和CAT 活性,这些酶作为植物体内过量ROS的清除剂,能够迅速将ROS转化为无害物质,从而减轻了膜脂过氧化引发的氧化损伤。进一步分析发现,AMF 可能通过提升AsA-GSH 循环中APX、GR 和MDHAR 等关键酶活性,加快AsA 和GSH 的再生与循环,增强非酶抗氧化机制的效率,协同清除多余的ROS。
Na+/K+稳态是植物耐盐碱的核心生理指标,直接决定细胞的渗透调节、酶活性及膜电位的稳定性[30]。笔者发现,SA 处理相较CK 显著降低了M9-T337 幼苗叶片的K+含量,同时使Na+和Na+/K+显著升高,表明盐碱胁迫严重破坏了M9-T337 幼苗的离子平衡。而接种AMF(SA+AMF 处理)能够显著缓解离子失衡状况,使叶片K+含量显著回升,同时降低Na+含量和Na+/K+。这一现象可能与AMF 增强根际离子选择性吸收能力、调节Na+外排与K+保持过程有关,从而提升了植株对离子胁迫的耐受性[31]。AMF 处理能够上调苹果中MdSOS2 与MdNHX2 等与Na+转运相关的基因表达,这可能通过增强液泡膜上Na+/H+逆向转运活性,促进Na+向液泡的区隔,从而有效降低细胞质中Na+浓度,缓解其毒性累积[32]。
在复合盐碱胁迫下,GORK1介导的K+选择性吸收与AREB1主导的ABA信号通路协同激活,是区别于单一胁迫的核心抗性特征,直接决定了离子选择性与胁迫信号转导的耦合效率[26]。AMF 预处理显著上调了MdGORK1 的表达,表明AMF 可能通过提高根系对K+的主动吸收能力,部分缓解了由H+泵抑制造成的K+流失[33]。同时,AMF 预处理也显著诱导了MdAREB1 的高表达,表明AMF 可能通过ABA 信号通路增强胁迫响应能力,与付海奇等[34]提出的激素-代谢物互作模型一致。这些发现揭示了复合胁迫下AMF调控网络的复杂性。
植物对盐碱胁迫的响应是一个极其复杂的生理生化过程,单一指标不足以全面评价植物的抗盐碱能力。为了全面评估AMF 对盐碱胁迫下M9-T337 幼苗的调控作用,采用主成分分析法对3 个处理下的27 个指标进行了综合评价。结果显示两个主成分(PC1 和PC2)的贡献率分别为78.556%和20.132%,累计贡献率为98.688%。主成分分析结果表明,AMF可以有效缓解盐碱胁迫对M9-T337幼苗的损害。
4 结 论
笔者系统揭示了丛枝菌根真菌(AMF)在缓解苹果砧木M9-T337 幼苗盐碱胁迫中的多层调控机制。AMF 通过协同调节离子稳态(上调MdSOS2、MdNHX2、MdGORK1 等基因表达)、提高K+吸收及Na+区隔化效率,有效缓解了离子毒害。同时,AMF还通过激活AsA-GSH循环与抗氧化酶系统,提高了氧自由基清除能力,减轻了膜脂过氧化损伤,从而维持了细胞膜稳定性。此外,AMF 还可能通过改善源-库关系、提高根冠比与光合效率,全面改善幼苗在盐碱逆境下的生理活性与生长表现。研究结果不仅丰富了对AMF 在果树砧木耐逆调控中作用机制的理解,也为盐碱地苹果建园提供了理论依据与技术支撑。
[1] LIU L L,WANG B S. Protection of halophytes and their uses for cultivation of saline-alkali soil in China[J]. Biology,2021,10(5):353.
[2] 关秀玲,申健. 植物响应盐碱胁迫的生理和分子机制研究进展[J]. 江苏农业科学,2024,52(21):10-16.GUAN Xiuling,SHEN Jian. Research progress on physiological and molecular mechanisms of plant in response to salt alkali stress[J]. Jiangsu Agricultural Sciences,2024,52(21):10-16.
[3] HASANUZZAMAN M,BHUYAN M H M B,PARVIN K,BHUIYAN T F,ANEE T I,NAHAR K,HOSSEN M S,ZULFIQAR F,ALAM M M,FUJITA M. Regulation of ROS metabolism in plants under environmental stress:A review of recent experimental evidence[J]. International Journal of Molecular Sciences,2020,21(22):8695.
[4] TEBINI M,CHIEB M,LUU D T,DAILLY H,LUTTS S,AHMED H B,CHALH A. Assessment of salt stress effects on antioxidant levels and membrane transport protein in Amaranthus caudatus[J]. Journal of Plant Growth Regulation,2025,44(6):3318-3329.
[5] 仪泽会,毛丽萍,赵婧. 嫁接对复合盐碱胁迫下青椒幼苗生长、抗氧化能力及渗透调节能力的影响[J]. 植物生理学报,2020,56(9):1943-1954.YI Zehui,MAO Liping,ZHAO Jing. Effects of grafting on growth,antioxidant capacity and osmotic adjustment capacity of green pepper seedlings under mixed salt-alkali stress[J]. Plant Physiology Journal,2020,56(9):1943-1954.
[6] XIAN X L,ZHANG Z X,WANG S C,CHENG J,GAO Y L,MA N Y,LI C L,WANG Y X. Exogenous melatonin strengthens saline-alkali stress tolerance in apple rootstock M9-T337 seedlings by initiating a variety of physiological and biochemical pathways[J]. Chemical and Biological Technologies in Agriculture,2024,11(1):58.
[7] JIA X M,ZHU Y F,ZHANG R,ZHU Z L,ZHAO T,CHENG L,GAO L Y,LIU B,ZHANG X Y,WANG Y X. Ionomic and metabolomic analyses reveal the resistance response mechanism to saline-alkali stress in Malus halliana seedlings[J]. Plant Physiology and Biochemistry,2020,147:77-90.
[8] ZHAI J T,XIAN X L,ZHANG Z X,WANG Y X. Nano-zinc oxide can enhance the tolerance of apple rootstock M9-T337 seedlings to saline alkali stress by initiating a variety of physiological and biochemical pathways[J]. Plants,2025,14(2):233.
[9] EVELIN H,DEVI T S,GUPTA S,KAPOOR R. Mitigation of salinity stress in plants by arbuscular mycorrhizal symbiosis:Current understanding and new challenges[J]. Frontiers in Plant Science,2019,10:470.
[10] 段海霞,罗崇亮,师茜,康生萍,赵玲,熊友才. 丛枝菌根真菌对植物-土壤系统的影响研究进展[J]. 生态学报,2025,45(1):475-491.DUAN Haixia,LUO Chongliang,SHI Qian,KANG Shengping,ZHAO Ling,XIONG Youcai. Research progress in the effects of arbuscular mycorrhizal fungi on plant-soil systems[J]. Acta Ecologica Sinica,2025,45(1):475-491.
[11] 苑海鹏,叶云舒,司皓,纪秋研,张玉红. 丛枝菌根真菌对植物逆境胁迫抗性及次生代谢产物合成的影响[J]. 生物技术通报,2024,40(6):45-56.YUAN Haipeng,YE Yunshu,SI Hao,JI Qiuyan,ZHANG Yuhong. Effects of arbuscular mycorrhizal fungi on plant stress resistance and secondary metabolite synthesis[J]. Biotechnology Bulletin,2024,40(6):45-56.
[12] 王子贺,刘雅洁,金蕴,卢晓瑜,李进华,杨春雪. 盐胁迫下丛枝菌根真菌接种和虎尾草伴生对羊草生长和生理特性的影响[J].中国草地学报,2024,46(6):22-35.WANG Zihe,LIU Yajie,JIN Yun,LU Xiaoyu,LI Jinhua,YANG Chunxue. Effects of arbuscular mycorrhizal fungi inoculation and association with Chloris virgata on growth and physiological characteristics of Leymus chinensis under salt stress[J]. Chinese Journal of Grassland,2024,46(6):22-35.
[13] 谭英,尹豪. 盐胁迫下根施AMF 和褪黑素对紫花苜蓿生长、光合特征以及抗氧化系统的影响[J]. 草业学报,2024,33(6):64-75.TAN Ying,YIN Hao. Effects of root application of an arbuscular mycorrhizal fungus and melatonin on the growth,photosynthetic characteristics,and antioxidant system of Medicago sativa under salt stresss[J]. Acta Prataculturae Sinica,2024,33(6):64-75.
[14] DERE S. Mitigating the adverse effects of salt stress on pepper plants through arbuscular mycorrhizal fungi (AMF) and beneficial bacterial (PGPR) inoculation[J]. Horticulturae,2024,10(11):1150.
[15] HAN S,CHENG Y,WU G Q,HE X W,ZHAO G Z. Enhancing salt tolerance in poplar seedlings through arbuscular mycorrhizal fungi symbiosis[J]. Plants,2024,13(2):233.
[16] 侯亚男,江帆,周舒扬,陈丁尹,朱怡杰,缪怡宁,岑锎,王怡芳,吴敏,刘鹏. 镉胁迫下丛枝菌根真菌对彩叶草生理代谢和根系损伤的影响[J]. 生物工程学报,2025,41(2):680-692.HOU Yanan,JIANG Fan,ZHOU Shuyang,CHEN Dingyin,ZHU Yijie,MIAO Yining,CEN Kai,WANG Yifang,WU Min,LIU Peng. Arbuscular mycorrhizal fungi improve physiological metabolism and ameliorate root damage of Coleus scutellarioides under cadmium stress[J]. Chinese Journal of Biotechnology,2025,41(2):680-692.
[17] 彭梓程,杜洪力,王铭,张凤华,杨海昌. 丛枝菌根真菌调控盐碱胁迫下棉花生长及离子平衡的研究[J]. 中国农业科技导报(中英文),2025,27(2):33-41.PENG Zicheng,DU Hongli,WANG Ming,ZHANG Fenghua,YANG Haichang. Research on AMF regulation of cotton growth and ion balance under salt alkali stress[J]. Journal of Agricultural Science and Technology,2025,27(2):33-41.
[18] CHANDRASEKARAN M,BOOPATHI T,MANIVANNAN P.Comprehensive assessment of ameliorative effects of AMF in alleviating abiotic stress in tomato plants[J]. Journal of Fungi,2021,7(4):303.
[19] 谢芳,任利明,丁飞,张硕新. 褪黑素对干旱胁迫下花椒影响的生理和转录组分析[J]. 西北林学院学报,2023,38(4):1-10.XIE Fang,REN Liming,DING Fei,ZHANG Shuoxin. Physiological and transcriptomic analyses of the effects of melatonin on Zanthoxylum bungeanum under drought stress[J]. Journal of Northwest Forestry University,2023,38(4):1-10.
[20] 山雨思,代欢欢,何潇,辛正琦,吴能表. 外源茉莉酸甲酯和水杨酸对盐胁迫下颠茄生理特性和次生代谢的影响[J]. 植物生理学报,2019,55(9):1335-1346.SHAN Yusi,DAI Huanhuan,HE Xiao,XIN Zhengqi,WU Nengbiao. Effects of exogenous methyl jasmonate and salicylic acid on physiological characteristics and secondary metabolism of Atropa belladonna under NaCl stress[J]. Plant Physiology Journal,2019,55(9):1335-1346.
[21] 邹琦. 植物生理学实验指导[M]. 北京:高等教育出版社,2003.ZOU Qi. Experimental guidance for plant physiology[M]. Beijing:Higher Education Press,2003.
[22] KAMBLE D,CHAVAN P,JONDHALE V. Study of potassium and sodium content of Mahad-Raigad tertiary soil by flame photometry[J]. Asian Journal of Research in Chemistry,2021:417-420.
[23] 张瑞. 垂丝海棠响应盐碱胁迫的生理特性及基于转录组学分析的MhPR1 功能基因验证[D]. 兰州:甘肃农业大学,2021.ZHANG Rui. Response of Malus halliana to saline- alkali stress and verification of MhPR1 functional based on transcriptomic[D]. Lanzhou:Gansu Agricultural University,2021.
[24] 孙志娟,刘文杰,郑晓东,袭祥利,马长青,刘晓丽,王彩虹,田义轲. 褪黑素对盐碱胁迫下平邑甜茶幼苗生长的影响及其机制[J]. 园艺学报,2023,50(8):1697-1710.SUN Zhijuan,LIU Wenjie,ZHENG Xiaodong,XI Xiangli,MA Changqing,LIU Xiaoli,WANG Caihong,TIAN Yike. Effects and functional mechanism of melatonin on the growth of Malus hupehensis seedlings under saline-alkali stress[J]. Acta Horticulturae Sinica,2023,50(8):1697-1710.
[25] WEN Y Q,SHI F,ZHANG B,LI K,CHANG W,FAN X X,DAI C L,SONG F Q. Rhizophagus irregularis and biochar can synergistically improve the physiological characteristics of saline-alkali resistance of switchgrass[J]. Physiologia Plantarum,2024,176(3):e14367.
[26] ZHU J K. Abiotic stress signaling and responses in plants[J].Cell,2016,167(2):313-324.
[27] SHINDE S,NAIK D,CUMMING J R. Carbon allocation and partitioning in Populus tremuloides are modulated by ectomycorrhizal fungi under phosphorus limitation[J]. Tree Physiology,2018,38(1):52-65.
[28] HASANUZZAMAN M,BHUYAN M H M B,ZULFIQAR F,RAZA A,MOHSIN S M,AL MAHMUD J,FUJITA M,FOTOPOULOS V. Reactive oxygen species and antioxidant defense in plants under abiotic stress:Revisiting the crucial role of a universal defense regulator[J]. Antioxidants,2020,9(8):681.
[29] HE Y,ZHOU J X,HU Y F,FANG C Y,YU Y J,YANG J,ZHU B,RUAN Y L,ZHU Z J. Overexpression of sly-miR398b increased salt sensitivity likely via regulating antioxidant system and photosynthesis in tomato[J]. Environmental and Experimental Botany,2021,181:104273.
[30] AMANIFAR S,TOGHRANEGAR Z. The efficiency of arbuscular mycorrhiza for improving tolerance of Valeriana officinalis L. and enhancing valerenic acid accumulation under salinity stress[J]. Industrial Crops and Products,2020,147:112234.
[31] PORCEL R,AROCA R,AZCON R,RUIZ-LOZANO J M. Regulation of cation transporter genes by the arbuscular mycorrhizal symbiosis in rice plants subjected to salinity suggests improved salt tolerance due to reduced Na+ root-to-shoot distribution[J].Mycorrhiza,2016,26(7):673-684.
[32] KHALID M,HASSANI D,LIAO J L,XIONG X,BILAL M,HUANG D F. An endosymbiont Piriformospora indica reduces adverse effects of salinity by regulating cation transporter genes,phytohormones,and antioxidants in Brassica campestris ssp.chinensis[J]. Environmental and Experimental Botany,2018,153:89-99.
[33] CHENG H Q,ZOU Y N,WU Q S,KUČA K. Arbuscular mycorrhizal fungi alleviate drought stress in trifoliate orange by regulating H+-ATPase activity and gene expression[J]. Frontiers in Plant Science,2021,12:659694.
[34] 付海奇,刘晓,宋姝,吕婉嘉,杨永青. 次生代谢物调控植物抵抗盐碱胁迫的机制[J]. 植物生理学报,2023,59(4):727-740.FU Haiqi,LIU Xiao,SONG Shu,LÜ Wanjia,YANG Yongqing.Mechanisms of secondary metabolites regulating plant resistance to salinity and alkali stress[J]. Plant Physiology Journal,2023,59(4):727-740.